Advanced One-Pot Synthesis of TES Buffer: Enhancing Purity and Scalability for Biopharma Applications
The biopharmaceutical industry relies heavily on high-performance buffering agents to maintain stable physiological conditions during critical processes such as vaccine production, tissue culture, and protein storage. Among these, 2-[[tris(hydroxymethyl)methyl]amino]ethanesulfonic acid, commonly known as TES, stands out as a vital zwitterionic buffer with exceptional properties for biological systems. Recent advancements in organic synthesis have sought to optimize the production of this key reagent, addressing long-standing issues regarding yield and environmental impact. Specifically, the technical disclosure in patent CN112479937A introduces a groundbreaking preparation method that utilizes a two-step one-pot synthesis strategy. This innovative approach not only streamlines the manufacturing workflow but also significantly enhances the purity profile of the final product, making it an ideal candidate for sensitive diagnostic medicine and clinical chemistry applications. By shifting away from traditional, waste-intensive pathways, this technology offers a robust solution for reliable pharma intermediate supplier networks seeking to upgrade their production capabilities.
![Chemical structure of 2-[[tris(hydroxymethyl)methyl]amino]ethanesulfonic acid showing the sulfonic acid group and tris-hydroxymethyl moiety](/insights/img/tes-buffer-synthesis-copper-catalysis-pharma-supplier-20260308170752-01.png)
The structural integrity of TES is paramount for its function as a biological buffer, and the new synthesis method ensures that the molecular architecture is preserved with minimal degradation. The process begins with readily available starting materials, circumventing the need for specialized and hazardous precursors that have historically plagued the supply chain. For R&D directors focused on impurity profiles, this method represents a significant leap forward, as the controlled reaction conditions minimize the formation of side products that are difficult to remove. The ability to produce high-purity OLED material or pharmaceutical intermediates often hinges on such precise control over reaction parameters, and this patent demonstrates exactly that level of sophistication. As we delve deeper into the technical specifics, it becomes clear that this methodology is not just a laboratory curiosity but a scalable industrial process ready for commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of TES buffer has been fraught with challenges that hindered large-scale commercial viability and cost efficiency. Traditional routes typically relied on the reaction of tris(hydroxymethyl)aminomethane with 2-haloethylsulfonic acid derivatives. While chemically feasible, this approach suffered from severe drawbacks, particularly when using chloride or bromide variants of the sulfonic acid precursor. In many cases, the reaction with chlorides resulted in negligible yields or complete failure to obtain the target product due to the poor leaving group ability and steric hindrance associated with the specific reaction conditions. Alternatively, using bromide derivatives did improve yields to some extent, but at a prohibitive cost and with the generation of substantial amounts of hazardous three wastes. These environmental and economic burdens made the conventional method unsustainable for modern green chemistry standards, creating a bottleneck for cost reduction in electronic chemical manufacturing and broader biotech sectors.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent employs a clever two-step one-pot synthesis that fundamentally alters the reaction landscape. Instead of starting with a pre-functionalized sulfonic acid, the process initiates with the alkylation of tris(hydroxymethyl)aminomethane using inexpensive 1,2-dihaloethane. This intermediate is then directly subjected to sulfonation using sodium sulfite in the presence of a catalyst, all within the same reaction vessel. This telescoping of steps eliminates the need for isolating unstable intermediates and drastically reduces solvent consumption and processing time. The reaction scheme below illustrates this elegant transformation, highlighting the simplicity and atom economy of the new route.
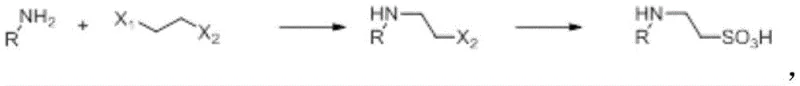
The implementation of this novel approach allows for the use of water as the primary solvent, which is a massive advantage for both safety and cost. By avoiding organic solvents in the main reaction steps, the process inherently reduces the risk of fire and toxicity, aligning perfectly with stringent environmental regulations. Furthermore, the flexibility to use various 1,2-dihaloethanes, including mixed halogen species like 2-chloro-1-bromoethane, provides procurement teams with the agility to source the most cost-effective raw materials available on the global market. This adaptability is crucial for maintaining supply chain continuity, especially when specific halogenated compounds face temporary shortages. The result is a streamlined process that delivers high yields, reportedly up to 87%, with a purity profile that meets the rigorous demands of high-purity pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed Sulfonation
The success of this synthesis lies in the precise mechanistic orchestration of nucleophilic substitution and subsequent sulfonation. In the first stage, the primary amine of tris(hydroxymethyl)aminomethane acts as a nucleophile, attacking the electrophilic carbon of the 1,2-dihaloethane. The maintenance of a pH value greater than or equal to 7.0 through the dropwise addition of sodium hydroxide is critical here; it ensures that the amine remains in its unprotonated, nucleophilic form, driving the formation of the amino-halo intermediate. Without this pH control, the accumulation of hydrogen halide would protonate the amine, effectively quenching the reaction and leading to low conversion rates. This careful regulation of the reaction environment is a hallmark of sophisticated process chemistry, ensuring that the kinetic pathway favors the desired product over potential oligomerization or hydrolysis side reactions.
Following the initial alkylation, the introduction of sodium sulfite and a copper powder catalyst triggers the second critical transformation. The copper catalyst facilitates the nucleophilic displacement of the remaining halogen atom by the sulfite ion, a reaction that typically requires elevated temperatures and prolonged reflux times of 16 to 17 hours. This step converts the halo-amine intermediate into the final zwitterionic sulfonic acid structure. The use of copper powder is particularly advantageous as it is a heterogeneous catalyst that can be easily removed by filtration, preventing metal contamination in the final product. For R&D teams, understanding this mechanism is key to troubleshooting and optimizing the process further, as it highlights the importance of catalyst surface area and mixing efficiency during the long reflux period to maximize the commercial scale-up of complex polymer additives or similar fine chemicals.
How to Synthesize 2-[[tris(hydroxymethyl)methyl]amino]ethanesulfonic acid Efficiently
Executing this synthesis requires strict adherence to the patented parameters to ensure reproducibility and high quality. The process is designed to be robust, utilizing common laboratory and industrial equipment such as reflux condensers and filtration units. Operators must monitor the pH closely during the initial heating phase and ensure that the catalyst is well-dispersed during the sulfonation step to prevent hot spots or incomplete reactions. The detailed standardized synthetic steps provided below outline the exact molar ratios, temperature profiles, and workup procedures necessary to achieve the reported 99% purity. Following these guidelines allows manufacturers to replicate the high yields demonstrated in the patent examples, ensuring a consistent supply of this critical buffer for downstream applications.
- Dissolve tris(hydroxymethyl)aminomethane and 1,2-dihaloethane in water, heat to reflux while maintaining pH ≥ 7.0 with NaOH to form the first intermediate.
- Add sodium sulfite and a copper powder catalyst to the reaction mixture, then heat to reflux for 16-17 hours to complete the sulfonation.
- Cool the mixture, filter off solids, treat with cation exchange resin, remove solvent, and crystallize using ethanol to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound benefits for procurement managers and supply chain heads looking to optimize their operational expenditures. The shift to a water-based, one-pot system eliminates the need for expensive organic solvents and the complex recovery systems associated with them, leading to substantial cost savings in utility and waste disposal. Moreover, the raw materials involved, such as 1,2-dihaloethane and sodium sulfite, are commodity chemicals with stable global supply chains, reducing the risk of production stoppages due to raw material scarcity. This stability is essential for reducing lead time for high-purity pharmaceutical intermediates, allowing companies to respond more agilely to market demands without the burden of holding excessive inventory.
- Cost Reduction in Manufacturing: The elimination of expensive 2-haloethylsulfonic acid precursors and the use of a recyclable copper catalyst significantly lower the direct material costs of production. Additionally, the simplified workup procedure, which involves straightforward filtration and crystallization rather than complex extractions or distillations, reduces labor and energy consumption. These factors combine to create a highly cost-efficient manufacturing process that improves overall profit margins without compromising on the quality of the final buffer product.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals like tris(hydroxymethyl)aminomethane and dihaloethanes, the supply chain becomes far more resilient to regional disruptions. The flexibility to substitute different dihaloethane variants based on availability further strengthens this reliability, ensuring that production schedules can be maintained even if a specific halogenated reagent is temporarily constrained. This robustness is a key strategic advantage for any organization aiming to secure a reliable agrochemical intermediate supplier or pharma partner status.
- Scalability and Environmental Compliance: The aqueous nature of the reaction and the absence of volatile organic compounds (VOCs) make this process inherently safer and easier to scale from pilot plants to multi-ton production facilities. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, minimizing the regulatory burden and potential fines associated with chemical manufacturing. This environmental compliance not only protects the company's reputation but also facilitates smoother permitting processes for capacity expansion, supporting long-term growth strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of TES buffer using this advanced methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and quality assurances associated with this synthesis route. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing manufacturing portfolios or sourcing strategies.
Q: What are the primary advantages of the one-pot synthesis method for TES buffer described in CN112479937A?
A: The primary advantages include a significantly simplified post-treatment process, high reaction yields reaching up to 87%, and excellent product purity of approximately 99%. Furthermore, this method avoids the use of expensive and waste-generating 2-haloethylsulfonic acid raw materials found in conventional routes.
Q: Why is pH control critical during the initial alkylation step of this synthesis?
A: Maintaining a pH value of greater than or equal to 7.0 during the reflux reaction between tris(hydroxymethyl)aminomethane and 1,2-dihaloethane is essential to neutralize the hydrogen halide byproduct. This ensures the amine remains nucleophilic enough to drive the reaction forward efficiently without forming unwanted salt impurities that could complicate downstream purification.
Q: How does the use of cation exchange resin improve the final quality of the TES buffer?
A: The addition of cation exchange resin, such as styrene gel type strong acid cation resin, effectively adsorbs residual metal cations and other ionic impurities from the reaction mixture. This step is crucial for achieving the high purity specifications required for biological applications, ensuring the final buffer is free from trace metals that could interfere with sensitive enzymatic or cell culture processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-[[tris(hydroxymethyl)methyl]amino]ethanesulfonic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality buffers play in the advancement of biotechnology and pharmaceutical research. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of TES buffer we produce meets the highest international standards. Our capability to implement complex synthetic routes like the one described in CN112479937A demonstrates our technical prowess and dedication to delivering superior chemical solutions.
We invite you to collaborate with us to leverage these technological advancements for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, demonstrating how our optimized processes can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us support your journey towards more efficient and sustainable chemical manufacturing.
