Optimizing Cephalosporin Side Chain Production via Safe Triphosgene Chlorination Technology
Optimizing Cephalosporin Side Chain Production via Safe Triphosgene Chlorination Technology
The pharmaceutical industry's relentless pursuit of safer and more efficient antibiotic production has led to significant innovations in the synthesis of key intermediates. Patent CN101805311A details a breakthrough synthetic method for 2-(2-amino-4-thiazolyl)-2-(Z)-methoxyimino acetic acid, a critical side chain active ester used extensively in the manufacturing of third-generation cephalosporins such as Cefixime and Cefpodoxime. This compound is pivotal because it imparts broad-spectrum activity and enhanced stability against beta-lactamases to the final antibiotic drug. The structural integrity and purity of this thiazole derivative are paramount, as any impurities can compromise the efficacy and safety profile of the life-saving medications derived from it. 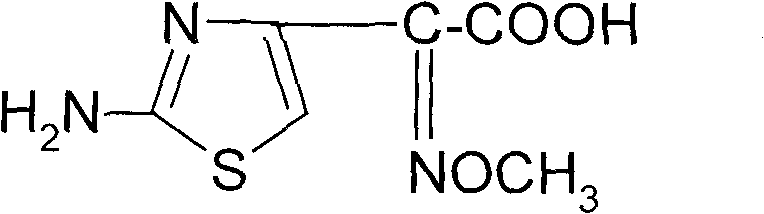 . By transitioning from hazardous gaseous reagents to safer solid alternatives and optimizing reaction homogeneity, this technology addresses long-standing bottlenecks in pharmaceutical intermediate manufacturing, offering a robust pathway for reliable pharmaceutical intermediates supplier networks globally.
. By transitioning from hazardous gaseous reagents to safer solid alternatives and optimizing reaction homogeneity, this technology addresses long-standing bottlenecks in pharmaceutical intermediate manufacturing, offering a robust pathway for reliable pharmaceutical intermediates supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of ainothiazoly loximate has been plagued by significant safety hazards and process inefficiencies inherent to traditional synthetic routes. Conventional methods typically rely on the use of chlorine gas (Cl2) for the crucial chlorination step, which presents severe occupational health risks due to its high toxicity and potential for leakage, posing a constant threat to operator safety and environmental compliance. Furthermore, the traditional nitrosation reaction often employs a sulfuric acid system that results in a heterogeneous reaction mixture, leading to poor mass transfer, sluggish reaction kinetics, and extended production cycles that strain manufacturing capacity. These inefficiencies not only result in lower overall yields but also generate complex waste streams that are difficult and costly to treat, thereby inflating the operational expenditure for cost reduction in pharmaceutical intermediates manufacturing. The unpredictability of gas-liquid reactions also complicates process control, making it challenging to maintain consistent batch-to-batch quality required for GMP compliance.
The Novel Approach
The innovative methodology outlined in the patent data introduces a paradigm shift by replacing hazardous gaseous chlorine with solid triphosgene, a crystalline compound that serves as a safe and controllable source of phosgene equivalents. This substitution fundamentally alters the risk profile of the plant, eliminating the need for specialized gas handling infrastructure and drastically reducing the potential for catastrophic accidental releases. Additionally, the implementation of a homogeneous oximation system using sodium nitrite and glacial acetic acid ensures that the reactants are fully dissolved, facilitating rapid and uniform interaction that significantly shortens reaction times. This dual approach of enhancing safety through reagent substitution and improving efficiency through phase homogeneity creates a streamlined process that is easier to scale and control. For procurement managers, this translates to a more stable supply chain with reduced downtime risks, while ensuring that the commercial scale-up of complex pharmaceutical intermediates remains economically viable and environmentally sustainable.
Mechanistic Insights into Triphosgene-Mediated Chlorination and Homogeneous Oximation
The core chemical innovation lies in the mechanistic behavior of triphosgene during the chlorination of the methoxyimino intermediate. Unlike molecular chlorine which reacts violently and indiscriminately, triphosgene decomposes in situ under controlled thermal conditions to release phosgene gradually, allowing for a much more regulated electrophilic attack on the enolizable position of the substrate. This controlled release mechanism minimizes side reactions such as over-chlorination or decomposition of the sensitive oxime functionality, which are common pitfalls in traditional gas-phase chlorination. The presence of an organic base, such as DMF or pyridine, further catalyzes this decomposition and scavenges the generated hydrochloric acid, driving the equilibrium forward and protecting the acid-sensitive groups within the molecule. This precise control over the chlorination step is critical for maintaining the stereochemical integrity of the Z-isomer, which is essential for the biological activity of the final cephalosporin antibiotic.
Simultaneously, the shift to a homogeneous oximation system fundamentally changes the kinetics of the initial nitrosation step. In traditional heterogeneous systems, the reaction rate is limited by the diffusion of nitrite ions across the phase boundary into the organic layer. By utilizing glacial acetic acid as a co-solvent with sodium nitrite, the system becomes a single phase where molecular collisions occur with maximum frequency and efficiency. This eliminates the diffusion barrier, allowing the nitrosation to proceed rapidly even at low temperatures (0°C to 5°C), which is crucial for preventing the thermal decomposition of the unstable nitroso intermediate. The result is a cleaner reaction profile with fewer by-products, simplifying the downstream purification burden and ensuring that the high-purity pharmaceutical intermediates required for sensitive beta-lactam coupling are achieved with minimal effort.
How to Synthesize 2-(2-Amino-4-Thiazolyl)-2-(Z)-Methoxyimino Acetic Acid Efficiently
The synthesis protocol described involves a sequential six-step process designed to maximize yield while minimizing hazard exposure. It begins with the preparation of the oxime precursor followed by methylation, then proceeds to the critical triphosgene chlorination, cyclization with thiourea, hydrolysis of the ester, and final recrystallization. Each step is optimized with specific solvent systems and temperature controls to ensure reproducibility. For R&D teams looking to implement this, the detailed standardized synthesis steps are provided below to guide laboratory and pilot plant operations effectively.
- Perform homogeneous oximation of methyl acetoacetate using sodium nitrite and glacial acetic acid at 0-5°C to form 2-hydroximino ethyl acetoacetate.
- Execute methylation using dimethyl sulfate under phase-transfer catalysis at pH 8-10 to yield 2-methoxyimino ethyl acetoacetate.
- Conduct chlorination using solid triphosgene in organic solvent with an organic base at 10-20°C, replacing toxic chlorine gas.
- Complete cyclization with thiourea and sodium acetate, followed by hydrolysis and final recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this triphosgene-based synthesis route offers profound advantages that extend beyond simple chemical yield improvements. The elimination of chlorine gas removes a major regulatory and logistical bottleneck, as facilities no longer require specialized permits for storing large quantities of toxic compressed gases, thereby reducing insurance premiums and compliance overheads. This simplification of the safety infrastructure allows for more flexible manufacturing site selection and reduces the risk of production halts due to safety inspections or minor leaks. Consequently, this leads to substantial cost savings in the long-term operation of the facility, making the final intermediate more price-competitive in the global market without compromising on quality standards.
- Cost Reduction in Manufacturing: The transition to a homogeneous reaction system significantly reduces the energy consumption associated with prolonged stirring and heating required to overcome mass transfer limitations in heterogeneous mixtures. Furthermore, the higher selectivity of the triphosgene chlorination minimizes the formation of difficult-to-remove impurities, which reduces the volume of solvents and adsorbents needed for purification. By streamlining the workflow and reducing the consumption of auxiliary materials, manufacturers can achieve a leaner production model that inherently lowers the cost of goods sold, providing a competitive edge in cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Relying on solid reagents like triphosgene and sodium nitrite rather than hazardous gases ensures a more robust supply chain that is less susceptible to transportation disruptions or strict regulatory shipping constraints. Solid chemicals are easier to store, handle, and inventory, allowing manufacturers to maintain larger safety stocks without the same level of risk associated with pressurized cylinders. This stability ensures consistent production schedules and reliable delivery timelines, which is critical for downstream API manufacturers who depend on just-in-time delivery to maintain their own antibiotic production lines without interruption.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, as the by-products of triphosgene decomposition are easier to neutralize and treat than chlorine-contaminated effluents. This aligns with increasingly stringent global environmental regulations, reducing the liability and cost associated with waste disposal. The inherent safety and controllability of the process also make it highly scalable, allowing for seamless transition from kilogram-scale R&D batches to multi-ton commercial production without the need for disproportionate increases in safety engineering controls.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this critical cephalosporin intermediate. These insights are derived directly from the patented technology to provide clarity on process capabilities and quality assurance measures. Understanding these details helps stakeholders make informed decisions regarding supplier qualification and process integration.
Q: Why is triphosgene preferred over chlorine gas in AE-Oxime synthesis?
A: Triphosgene is a solid crystalline substance that acts as a safe equivalent to phosgene, eliminating the severe safety risks, toxicity, and storage difficulties associated with handling gaseous chlorine. It allows for precise stoichiometric control and smoother reaction kinetics.
Q: How does the homogeneous oximation system improve yield?
A: By utilizing a sodium nitrite and glacial acetic acid system, the reaction mixture remains homogeneous, significantly increasing the contact area between reactants compared to traditional heterogeneous sulfuric acid methods. This accelerates reaction speed and boosts overall conversion rates.
Q: What is the typical purity specification for this intermediate?
A: The optimized process described in patent CN101805311A achieves a finished product purity of not less than 99.06%, with a melting point range of 182.1°C to 183.9°C, meeting stringent requirements for third-generation cephalosporin synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Amino-4-Thiazolyl)-2-(Z)-Methoxyimino Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we understand that the consistency of your antibiotic production depends entirely on the quality of your starting materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the advanced triphosgene-mediated synthesis described in patent CN101805311A can be implemented with the highest standards of safety and efficiency. Our rigorous QC labs and stringent purity specifications guarantee that every batch of AE-Oxime meets the exacting requirements necessary for synthesizing high-performance third-generation cephalosporins, giving you confidence in your supply chain.
We invite you to collaborate with our technical team to explore how this optimized route can benefit your specific production needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and reliability for your organization.
