Scalable Synthesis of Disulfonated Diphenyl Sulfone Monomers for Advanced Polymer Applications
Scalable Synthesis of Disulfonated Diphenyl Sulfone Monomers for Advanced Polymer Applications
The development of advanced functional materials, particularly for energy conversion and storage systems, relies heavily on the availability of specialized monomers with precise structural attributes. Patent CN1232506C introduces a significant breakthrough in the synthesis of 4,4'-disubstituted diphenyl sulfones containing dual sulfonic acid groups, a class of compounds critical for the next generation of ion exchange membranes and proton exchange membranes. This technology addresses the growing demand for polymers with enhanced solubility and controlled sulfonation degrees, which are essential for optimizing the performance of fuel cells and other electrochemical devices. By utilizing a straightforward yet highly effective two-step sequence involving electrophilic sulfonation followed by oxidative conversion of alkyl groups, this method offers a robust pathway to high-value intermediates that were previously difficult to access with consistent quality. For R&D directors and procurement specialists alike, understanding the nuances of this synthetic route is vital for securing a reliable supply chain for high-performance polymer precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of sulfonated aromatic monomers has been plagued by issues related to regioselectivity and harsh reaction conditions that often lead to complex impurity profiles. Conventional routes frequently involve direct sulfonation of pre-functionalized aromatics, which can result in over-sulfonation or the formation of insoluble cross-linked byproducts that are notoriously difficult to purify. Furthermore, many existing methods rely on protecting group strategies to achieve the desired substitution pattern, adding multiple steps that drastically reduce overall yield and increase waste generation. The inability to precisely control the position of sulfonic acid groups relative to other functional moieties, such as carboxyl groups, often limits the utility of these monomers in creating well-defined copolymers. Consequently, manufacturers face challenges in scaling these processes due to the accumulation of intractable tars and the need for expensive purification techniques like preparative HPLC, which are economically unviable for industrial production.
The Novel Approach
The methodology outlined in the patent data presents a paradigm shift by leveraging the inherent reactivity of 4,4'-dialkyl diphenyl sulfones to achieve high regioselectivity without the need for complex protection schemes. By initiating the synthesis with a readily available dialkyl precursor, the process exploits the directing effects of the sulfone bridge to guide sulfonation specifically to the meta positions, ensuring a clean 3,3'-disulfonated intermediate. This strategic ordering of reactions—sulfonation first, followed by oxidation of the alkyl chains to carboxylic acids—prevents the degradation of sensitive carboxyl groups under the aggressive conditions required for sulfonation. The result is a streamlined workflow that minimizes side reactions and maximizes the recovery of the target disulfonated dicarboxylic acid. This approach not only simplifies the operational complexity but also significantly enhances the purity of the final product, making it ideally suited for the rigorous demands of polymerization into high-performance engineering plastics and membranes.
Mechanistic Insights into Electrophilic Sulfonation and Permanganate Oxidation
The core of this synthetic strategy lies in the precise control of electrophilic aromatic substitution during the sulfonation stage. When 4,4'-dialkyl diphenyl sulfone is treated with oleum, the sulfur trioxide acts as a potent electrophile, attacking the aromatic rings at positions meta to the electron-withdrawing sulfone group and ortho to the electron-donating alkyl groups. However, the strong deactivating nature of the sulfone linkage dominates, directing the incoming sulfonyl groups primarily to the 3 and 3' positions relative to the sulfone bridge. The use of oleum with a controlled concentration of free sulfur trioxide (preferably 3-12 mol per mole of substrate) is critical to prevent polysulfonation while ensuring complete conversion to the disulfonated species. The reaction temperature is carefully maintained between 70°C and 150°C to balance reaction kinetics with selectivity, avoiding the thermal degradation that can occur at higher temperatures. This mechanistic understanding allows for the fine-tuning of reaction parameters to achieve optimal yields of the 3,3'-disulfonic acid-4,4'-dialkyl diphenyl sulfone intermediate.
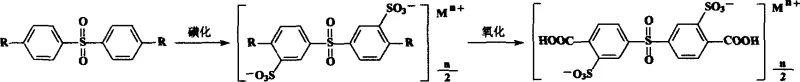
Following the successful introduction of the sulfonic acid groups, the second critical transformation involves the oxidation of the alkyl substituents to carboxylic acids using potassium permanganate. This oxidation step proceeds through a radical mechanism where the permanganate ion abstracts hydrogen atoms from the benzylic positions of the alkyl groups, eventually converting the methyl or higher alkyl chains into carboxyl functionalities. The choice of potassium permanganate is advantageous due to its high oxidation potential and selectivity for benzylic carbons, leaving the aromatic ring and the sulfonic acid groups intact. The reaction is typically conducted in an aqueous medium at temperatures ranging from 60°C to 100°C, ensuring that the oxidant remains soluble and reactive while minimizing the risk of over-oxidation or ring cleavage. The resulting manganese dioxide byproduct is easily removed by hot filtration, simplifying the downstream purification process and contributing to the overall efficiency of the synthesis.
How to Synthesize 3,3'-Disulfonic Acid-4,4'-Dicarboxy Diphenyl Sulfone Efficiently
Implementing this synthesis requires careful attention to the isolation and purification stages to ensure the removal of inorganic salts and residual oxidants that could interfere with subsequent polymerization reactions. The process begins with the sulfonation of the dialkyl precursor, followed by a salting-out procedure using saturated sodium chloride to precipitate the sulfonated intermediate, which is then neutralized and purified via alcohol precipitation. The subsequent oxidation step generates the final dicarboxylic acid derivative, which is isolated by acidifying the cooled filtrate with hydrochloric acid to induce crystallization of the white needle-like product. Detailed standardized operating procedures for each unit operation, including specific stoichiometric ratios and temperature ramps, are essential for reproducibility.
- Sulfonation Stage: React 4,4'-dialkyl diphenyl sulfone with oleum (containing 3-12 mol free SO3) at 70-150°C for 6-10 hours to form the disulfonated intermediate.
- Isolation: Pour the reaction mixture into saturated sodium chloride solution to salt out the product, followed by neutralization and filtration to remove sulfuric acid.
- Oxidation and Purification: Oxidize the alkyl groups using potassium permanganate at 60-100°C, then filter hot, acidify the filtrate with HCl, and crystallize to obtain the final white needle-like crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for organizations looking to optimize their supply chain for specialty polymer intermediates. The reliance on commodity chemicals such as oleum, potassium permanganate, and simple alkyl diphenyl sulfones ensures that raw material availability is high and subject to minimal market volatility. Unlike processes that require rare earth catalysts or exotic reagents, this method utilizes widely accessible industrial inputs, thereby reducing the risk of supply disruptions and enabling more accurate long-term cost forecasting. Furthermore, the elimination of complex chromatographic purification steps in favor of crystallization and filtration significantly lowers the capital expenditure required for manufacturing equipment, making the technology highly attractive for scale-up.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the sulfonation and oxidation steps, which minimize waste generation and maximize the utilization of starting materials. By avoiding the use of expensive transition metal catalysts and eliminating the need for solvent-intensive purification techniques, the overall production cost is significantly reduced compared to traditional multi-step syntheses. The ability to recover and recycle the mother liquors from the crystallization steps further enhances the process economics, offering a compelling value proposition for large-scale manufacturing. Additionally, the high purity of the final product reduces the burden on downstream polymerization processes, potentially lowering the total cost of ownership for the final membrane material.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a highly reliable supply chain, as the process is less susceptible to minor fluctuations in temperature or reagent quality. The use of stable intermediates that can be isolated and stored allows for flexible production scheduling, decoupling the sulfonation and oxidation stages if necessary to manage inventory levels effectively. This modularity ensures that manufacturers can respond quickly to changes in demand without compromising product quality or delivery timelines. Moreover, the simplicity of the workup procedures reduces the dependency on highly specialized technical labor, further stabilizing the production workflow and mitigating operational risks.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is facilitated by the use of standard reactor configurations and common unit operations such as filtration and crystallization. The absence of hazardous gases or highly toxic reagents simplifies environmental compliance and waste management, aligning with modern green chemistry principles. The aqueous nature of the oxidation step and the ability to neutralize acidic waste streams make the process environmentally friendly, reducing the regulatory burden associated with chemical manufacturing. This ease of scale-up ensures that the technology can meet the growing global demand for sulfonated monomers without encountering the bottlenecks often associated with more complex synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these sulfonated monomers, based on the detailed specifications provided in the patent literature. Understanding these aspects is crucial for integrating this material into your specific polymer formulation or membrane casting process.
Q: What is the primary advantage of this sulfonated monomer for membrane applications?
A: The presence of both sulfonic acid and carboxylic acid groups allows for precise control over the degree of sulfonation when copolymerized, enhancing proton conductivity while maintaining mechanical stability in ion exchange membranes.
Q: How does the purification process ensure high purity for polymerization?
A: The process utilizes a combination of salting-out with saturated inorganic salts and recrystallization from alcohol/water mixtures, effectively removing inorganic byproducts and unreacted starting materials without requiring complex chromatography.
Q: Can the alkyl chain length be varied in this synthesis?
A: Yes, the patent specifies that R can be a straight or branched alkyl chain with 1 to 10 carbon atoms, although methyl is preferred for optimal reactivity and yield during the oxidation step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonated Polymer Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity monomers play in the development of advanced functional materials, and we are committed to supporting your innovation with superior manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pilot phase or full-scale manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3,3'-disulfonic acid-4,4'-dicarboxy diphenyl sulfone meets the exacting standards required for high-performance ion exchange membranes. Our dedication to quality assurance means that you can rely on us for consistent product performance, batch after batch.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development timelines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs and application requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our manufacturing expertise can enhance your project's success. Let us be your strategic partner in bringing next-generation polymer technologies to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
