Advanced Rhodium-Catalyzed Synthesis of Tetrahydroisoquinoline Derivatives for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust synthetic routes for complex heterocyclic scaffolds that serve as critical backbones for new drug candidates. Patent CN111072562B introduces a groundbreaking methodology for the construction of novel tetrahydroisoquinoline derivatives, a class of compounds renowned for their diverse pharmacological profiles including antihypertensive and antitumor activities. This intellectual property details a highly efficient three-component coupling reaction that merges substituted anilines, C,N-cyclic imines, and diazo compounds into a unified molecular architecture. The significance of this technology lies not only in the structural novelty of the resulting products but also in the operational simplicity of the process, which operates under remarkably mild conditions. For R&D teams focused on oncology, particularly gastric cancer therapeutics, these derivatives exhibit potent inhibition against AGS cell lines, positioning them as valuable intermediates for next-generation anticancer agents. The strategic implementation of this synthesis route allows for the rapid generation of diverse chemical libraries, accelerating the hit-to-lead optimization phase in drug discovery pipelines.
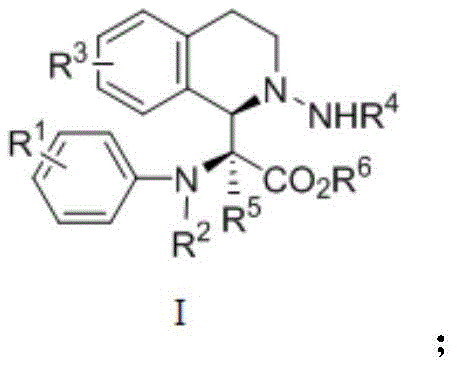
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized tetrahydroisoquinoline skeletons has been plagued by significant synthetic hurdles that impede large-scale production and rapid diversification. Traditional pathways often rely on multi-step sequences involving harsh reaction conditions, such as extreme temperatures or strongly acidic environments, which can degrade sensitive functional groups and limit substrate scope. Furthermore, many existing protocols utilize unstable or difficult-to-obtain starting materials that drive up costs and introduce supply chain vulnerabilities. The low atom economy associated with classical cyclization methods frequently results in substantial waste generation, creating environmental burdens and increasing disposal costs for manufacturing facilities. Additionally, controlling stereochemistry in these conventional routes often requires expensive chiral auxiliaries or resolution steps, further complicating the process and reducing overall throughput. These cumulative inefficiencies make traditional methods ill-suited for the agile demands of modern pharmaceutical process development.
The Novel Approach
In stark contrast, the methodology disclosed in the patent data presents a paradigm shift towards efficiency and sustainability through a direct, catalytic multicomponent reaction. This novel approach leverages a transition metal-catalyzed cascade that constructs the complex tetrahydroisoquinoline core in a single operational step from readily available precursors. By utilizing a dirhodium catalyst system, the reaction proceeds at room temperature, eliminating the energy costs and safety risks associated with heating. The process demonstrates exceptional functional group tolerance, allowing for the incorporation of various halogens and alkyl groups without the need for protective group strategies. This streamlining of the synthetic route drastically reduces the number of unit operations, thereby minimizing solvent consumption and processing time. The result is a high-yielding, selective transformation that aligns perfectly with the principles of green chemistry while delivering high-value intermediates suitable for commercial exploitation.
Mechanistic Insights into Rh2(OAc)4-Catalyzed Multicomponent Coupling
The core of this transformative synthesis lies in the unique reactivity of the rhodium catalyst in mediating carbene transfer and subsequent cyclization events. The mechanism initiates with the activation of the diazo compound by the dirhodium tetraacetate catalyst, leading to the extrusion of nitrogen gas and the formation of a highly reactive rhodium-carbene intermediate. This electrophilic species then undergoes a selective insertion or addition reaction with the nucleophilic C,N-cyclic imine component. The resulting zwitterionic or metallated intermediate is poised for an intramolecular cyclization with the aniline nitrogen, effectively stitching together the three distinct fragments into the final fused ring system. This cascade sequence is meticulously controlled by the catalyst ligand environment, ensuring high regioselectivity and diastereoselectivity throughout the bond-forming events. Understanding this mechanistic pathway is crucial for process chemists aiming to fine-tune reaction parameters for specific substrate classes.
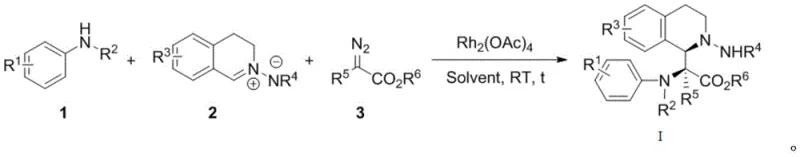
Impurity control in this catalytic system is inherently managed by the high specificity of the rhodium center and the mild reaction conditions. Unlike thermal reactions that often promote non-selective radical pathways leading to complex byproduct mixtures, this metal-catalyzed route follows a defined coordination sphere trajectory. The use of methyl tert-butyl ether as the preferred solvent further enhances purity profiles by providing a stable medium that does not participate in side reactions. The patent data indicates that diastereomeric ratios (dr) are consistently favorable, often exceeding 85:15, which simplifies downstream purification requirements. For manufacturing teams, this means that crude product streams are cleaner, reducing the load on chromatographic columns and crystallization steps. The ability to predict and manage impurity formation through catalyst choice and stoichiometry ensures that the final active pharmaceutical ingredient meets stringent regulatory standards for purity and safety.
How to Synthesize Tetrahydroisoquinoline Derivatives Efficiently
Executing this synthesis on a practical scale requires careful attention to reagent addition rates and mixing efficiency to manage the exothermic decomposition of the diazo species. The standard protocol involves dissolving the aniline and cyclic imine substrates along with the catalyst in the solvent prior to the introduction of the diazo compound. This sequential addition strategy is critical for maintaining low concentrations of the reactive carbene intermediate, thereby suppressing dimerization or other competing decomposition pathways. Detailed standardized operating procedures for this transformation, including specific molar ratios and workup techniques, are essential for reproducibility across different batch sizes. The following guide outlines the fundamental steps derived from the optimized examples provided in the technical disclosure.
- Dissolve the substituted aniline compound, C,N-cyclic imine, and Rh2(OAc)4 catalyst in methyl tert-butyl ether solvent.
- Slowly add the solution of diazo compound dissolved in methyl tert-butyl ether to the reaction mixture over one hour at 25°C.
- Continue stirring for two hours, remove solvent by rotary evaporation, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The reliance on commodity chemicals such as substituted anilines and simple diazo esters means that raw material sourcing is straightforward and cost-effective, avoiding the bottlenecks associated with exotic reagents. The elimination of cryogenic conditions or high-pressure equipment significantly lowers the capital expenditure required for manufacturing infrastructure, making this process accessible to a wider range of production facilities. Furthermore, the reduced number of synthetic steps translates directly into shorter lead times for batch completion, enhancing the agility of the supply chain to respond to market demands. These factors collectively contribute to a more resilient and economically viable production model for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy and the use of inexpensive, commercially available starting materials. By avoiding the need for precious metal ligands beyond standard rhodium acetate and eliminating multiple protection-deprotection sequences, the overall cost of goods sold is significantly optimized. The high yields reported across various substrate examples ensure that raw material waste is minimized, further driving down the effective cost per kilogram of the final product. Additionally, the simplified purification process reduces solvent usage and labor hours, contributing to substantial operational savings.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for long-term project success, and this methodology excels by utilizing robust and widely available feedstocks. The tolerance for diverse substituents on the aniline ring allows for flexibility in supplier selection, mitigating the risk of single-source dependency. The mild reaction conditions also reduce the likelihood of equipment failure or safety incidents that could disrupt production schedules. This reliability ensures a consistent flow of intermediates to downstream formulation teams, supporting uninterrupted drug development timelines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the ambient temperature operation of this reaction facilitates a smoother transition from laboratory to pilot and commercial scales. The use of methyl tert-butyl ether, a solvent with favorable handling properties, supports safe large-volume operations. Moreover, the high selectivity of the reaction minimizes the generation of hazardous waste streams, aligning with increasingly strict environmental regulations and corporate sustainability goals. This eco-friendly profile enhances the marketability of the final product to environmentally conscious partners.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope and operational parameters. These insights are derived directly from the experimental data and optimization studies detailed in the patent documentation. Addressing these questions early in the project lifecycle helps in setting realistic expectations for yield, purity, and timeline.
Q: What is the preferred catalyst system for this tetrahydroisoquinoline synthesis?
A: While several transition metals were screened, Dirhodium tetraacetate (Rh2(OAc)4) demonstrated superior performance, offering the highest yields and diastereoselectivity under mild conditions.
Q: Can this process accommodate diverse aniline substrates?
A: Yes, the method exhibits broad substrate scope, tolerating various substituents on the aniline ring including halogens (F, Cl, Br, I), alkyl groups, and alkoxy groups at ortho, meta, and para positions.
Q: What are the biological activities of the synthesized compounds?
A: The resulting tetrahydroisoquinoline derivatives show significant inhibitory activity against human gastric cancer AGS cells, with IC50 values generally below 5 μM, indicating strong potential as anticancer leads.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroisoquinoline Derivative Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to bring this advanced synthetic methodology to life on an industrial scale. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tetrahydroisoquinoline derivative meets the highest quality standards required for pharmaceutical applications. Our commitment to excellence ensures that you receive a product that is not only chemically pure but also fully compliant with global regulatory requirements.
We invite you to collaborate with us to leverage this cutting-edge technology for your anticancer drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project milestones and optimize your budget.
