Revolutionizing Corticosteroid Intermediates: A Deep Dive into One-Pot 16-Beta-Methyl Steroid Synthesis
Revolutionizing Corticosteroid Intermediates: A Deep Dive into One-Pot 16-Beta-Methyl Steroid Synthesis
The pharmaceutical industry continuously seeks robust methodologies to streamline the production of critical corticosteroid intermediates, particularly those serving as precursors for high-value drugs like betamethasone. A significant breakthrough in this domain is detailed in patent CN109734762B, which discloses a highly efficient one-pot method for synthesizing 16-beta-methyl steroid compounds. This innovation addresses long-standing challenges in stereochemical control and process efficiency, offering a viable pathway for reliable pharmaceutical intermediate suppliers aiming to optimize their manufacturing portfolios. By eliminating the need for multiple isolation steps and expensive acetylation reagents, this technology represents a paradigm shift in how complex steroid scaffolds are constructed at scale.
For R&D directors and process chemists, the ability to access high-purity 16-beta-methyl steroids through a consolidated workflow is transformative. The patent outlines a sequence where an etherate precursor reacts with methyl bromide under the influence of a non-nucleophilic strong base, followed by in-situ silylation and pH adjustment. This approach not only simplifies the operational burden but also enhances the overall atom economy of the synthesis. As we delve deeper into the technical specifics, it becomes evident that this method offers substantial advantages in terms of impurity profile management and scalability, crucial factors for any organization focused on cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 16-beta-methyl steroid compounds has been plagued by inefficiencies inherent to multi-step batch processes. The traditional route, as highlighted in the background of the patent, typically involves a cumbersome three-step sequence. Initially, a 16-acetyl group is introduced using sodium alkoxide and diethyl oxalate to activate the 16-position. However, due to the steric influence of the 18-position beta-methyl group, the resulting 16-acetyl configuration predominantly exists in the alpha position. Subsequently, methylation is performed using methyl iodide under alkaline conditions, which forces the incoming methyl group into the beta configuration due to the blocking effect of the alpha-acetyl group. Finally, the 16-acetyl group must be removed under acidic catalysis to reveal the desired 16-beta-methyl product.
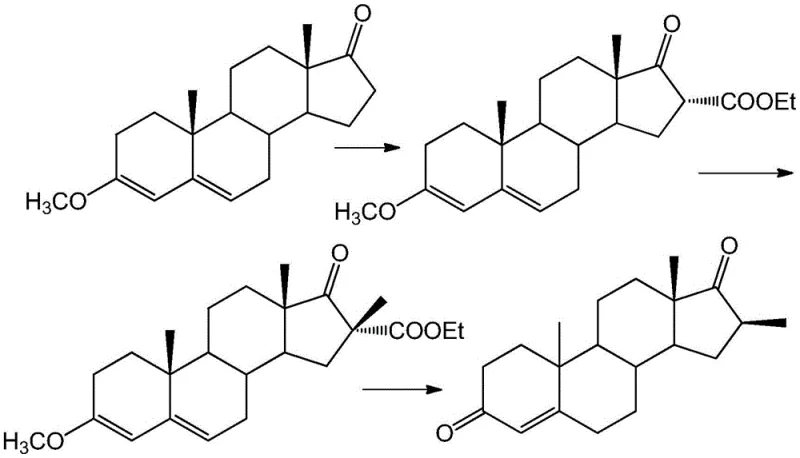
This conventional pathway presents several critical bottlenecks for commercial scale-up of complex pharmaceutical intermediates. The requirement for diethyl oxalate introduces significant raw material costs, and the use of methyl iodide poses handling and environmental concerns due to its toxicity. Furthermore, the necessity of performing three distinct chemical transformations with intermediate workups increases the risk of yield loss at each stage. The operational complexity is further compounded by the need for rigorous purification between steps to remove byproducts, leading to extended production cycles and higher solvent consumption. For procurement managers, these factors translate into volatile pricing and potential supply chain disruptions, making the traditional route less attractive for high-volume production.
The Novel Approach
In stark contrast to the legacy methods, the novel one-pot strategy described in patent CN109734762B offers a streamlined alternative that bypasses the need for acetylation entirely. The process begins with the reaction of 3-methoxy-3,5-diene-17-one (etherate) with methyl bromide in the presence of a non-nucleophilic strong base, directly yielding a 16-alpha-methyl substance. Instead of isolating this intermediate, the protocol proceeds immediately to the addition of trimethylchlorosilane (TMSCl), which forms a 17-position silyl ether within the same reaction vessel. The final transformation is achieved by adjusting the pH of the system, typically to around 5, which facilitates the stereoinversion to the thermodynamically stable 16-beta-methyl steroid compound.
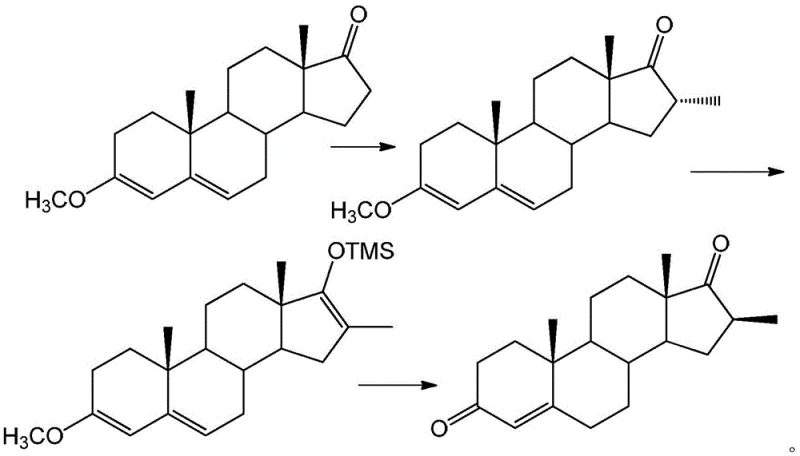
The elegance of this new approach lies in its convergence of multiple synthetic operations into a single pot, thereby drastically reducing the physical footprint and equipment time required for production. By utilizing methyl bromide instead of the more hazardous methyl iodide and avoiding the expensive diethyl oxalate reagent, the process inherently lowers the cost of goods sold (COGS). Moreover, the in-situ formation of the silyl ether acts as a temporary protecting group that directs the stereochemistry effectively without the need for a bulky acetyl group. This results in a cleaner reaction profile with fewer side products, simplifying downstream purification and enhancing the overall throughput for commercial scale-up of complex steroid derivatives.
Mechanistic Insights into Stereoselective Methylation and Silylation
Understanding the mechanistic underpinnings of this one-pot synthesis is essential for R&D teams aiming to replicate or adapt the process. The reaction initiates with the deprotonation of the 3-methoxy-3,5-diene-17-one substrate by a strong, non-nucleophilic base such as Lithium Diisopropylamide (LDA) or Lithium Hexamethyldisilazide (LiHMDS). These bases are selected specifically for their ability to abstract the proton at the 16-position without engaging in nucleophilic attack on the carbonyl or enol ether functionalities. The resulting enolate intermediate is then trapped by methyl bromide. Due to the steric environment of the steroid nucleus, the methylation occurs preferentially from the alpha face, yielding the 16-alpha-methyl isomer as the kinetic product. This step is conducted at cryogenic temperatures, typically between -70°C and -60°C, to maintain strict kinetic control and prevent equilibration to unwanted isomers.
Following the methylation, the addition of trimethylchlorosilane (TMSCl) serves a dual purpose: it protects the 17-ketone as a silyl enol ether and sets the stage for stereoinversion. The subsequent acidification of the reaction mixture to a pH of roughly 5 triggers the hydrolysis of the silyl group and the equilibration of the steroid ring system. Under these mildly acidic conditions, the 16-alpha-methyl configuration is thermodynamically less stable than the 16-beta-methyl configuration due to 1,3-diaxial interactions with the angular methyl group at C13. Consequently, the system equilibrates to favor the 16-beta isomer. This mechanism allows for high stereoselectivity without the need for chiral catalysts or resolution steps, ensuring that the final product meets stringent purity specifications required for pharmaceutical applications.
How to Synthesize 16-Beta-Methyl Steroid Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins by dissolving the etherate substrate in a suitable solvent such as tetrahydrofuran (THF) or 2-methyltetrahydrofuran, followed by cooling the mixture to cryogenic temperatures. Methyl bromide is introduced, and the base is added slowly to manage the exotherm. Once the methylation is complete, the temperature is raised slightly before the addition of TMSCl. The final acid quench and crystallization steps are critical for isolating the product in high purity. Detailed standardized operating procedures regarding stoichiometry, addition rates, and safety protocols for handling methyl bromide are essential for successful technology transfer.
- React 3-methoxy-3,5-diene-17-one with methyl bromide and a non-nucleophilic strong base like LDA at low temperatures (-70 to -60°C) to form the 16-alpha methyl intermediate.
- Without isolation, add trimethylchlorosilane (TMSCl) to the reaction mixture to generate the 17-position silyl ether species.
- Adjust the system pH to approximately 5 using acid (e.g., HCl) and maintain at low temperature to effect stereoinversion and crystallization of the final 16-beta product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot methodology offers compelling benefits for procurement managers and supply chain heads tasked with optimizing the sourcing of corticosteroid intermediates. The consolidation of three reaction steps into a single vessel significantly reduces the consumption of solvents, energy, and labor hours associated with intermediate isolations and drying processes. This reduction in processing intensity translates directly into lower manufacturing costs, allowing suppliers to offer more competitive pricing structures for high-purity pharmaceutical intermediates. Furthermore, the elimination of diethyl oxalate and methyl iodide from the bill of materials mitigates exposure to volatile commodity prices and regulatory restrictions associated with these hazardous chemicals.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the synthetic route. By removing the acetylation and deacetylation steps, manufacturers save on the direct costs of reagents like diethyl oxalate and the associated waste disposal fees. Additionally, the one-pot nature of the reaction minimizes product loss during transfer and filtration stages, leading to higher overall mass balance efficiency. The use of methyl bromide, while requiring careful handling, is generally more cost-effective than methyl iodide, further contributing to the economic viability of the process. These cumulative savings enable a more robust margin structure for producers of betamethasone derivatives.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reduced dependency on complex multi-step workflows that are prone to bottlenecks. With fewer unit operations, the lead time for producing a batch of 16-beta-methyl steroid is shortened, allowing for faster response to market demand fluctuations. The simplified process also reduces the risk of batch failures caused by errors in intermediate handling or purification, ensuring a more consistent supply of material for downstream API synthesis. This reliability is crucial for maintaining continuous production schedules in the highly regulated pharmaceutical sector, where delays can have cascading effects on drug availability.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than that of traditional methods, aligning with modern green chemistry principles. The reduction in solvent usage and the avoidance of heavy metal catalysts or toxic alkylating agents simplify waste treatment protocols. This ease of waste management facilitates smoother regulatory approvals and reduces the environmental compliance burden on manufacturing sites. Moreover, the process is inherently scalable; the exothermic nature of the reactions can be managed effectively in larger reactors through controlled addition rates, making it suitable for commercial scale-up from pilot plants to multi-ton annual production capacities without significant re-engineering.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the synthesis and quality attributes of the 16-beta-methyl steroid produced via this method. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for assessing process feasibility. Understanding these nuances is vital for integrating this intermediate into existing supply chains and ensuring compatibility with downstream corticosteroid synthesis pathways.
Q: What are the key advantages of this one-pot method over traditional routes?
A: The traditional route requires three distinct steps involving diethyl oxalate and separate deacetylation, whereas this patented one-pot method consolidates methylation and stereoinversion into a single vessel, drastically reducing operational complexity and solvent usage.
Q: Which bases are suitable for maintaining high stereoselectivity in this reaction?
A: The patent specifies the use of non-nucleophilic strong bases such as Lithium Diisopropylamide (LDA) or Lithium Hexamethyldisilazide (LiHMDS) to ensure precise kinetic control during the initial methylation step.
Q: What is the expected purity and yield for the final 16-beta-methyl steroid?
A: Experimental data within the patent demonstrates that this method consistently achieves high yields of approximately 95-96% with HPLC purity levels reaching 98% or higher, making it highly suitable for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16-Beta-Methyl Steroid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the one-pot 16-beta-methyl steroid synthesis can be seamlessly transitioned from the laboratory to industrial reality. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex steroid chemistry allows us to provide a secure and consistent supply of critical intermediates for the manufacture of life-saving corticosteroids.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthetic route can benefit your specific project requirements. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic advantages of switching to this streamlined process. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
