Optimizing Ertapenem Sodium Salt Production: A Technical Breakthrough in Carbapenem Synthesis
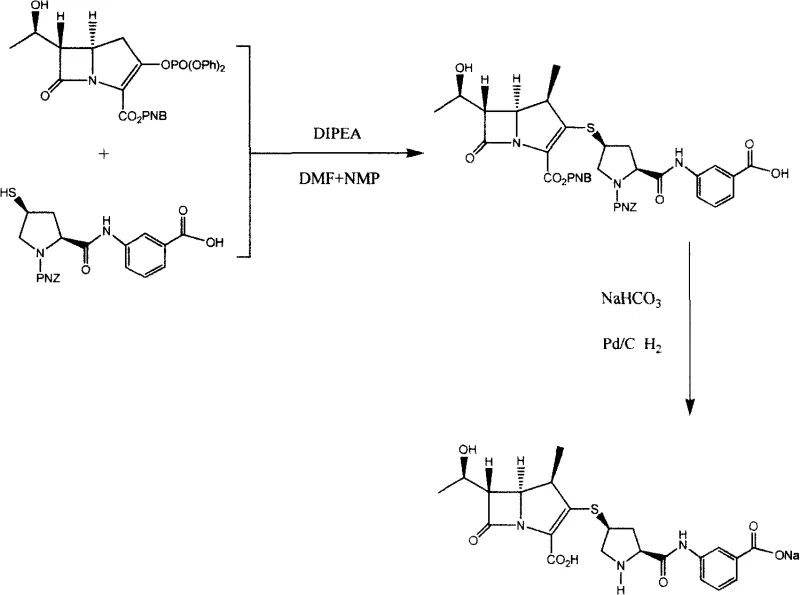
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing critical antibiotics, particularly carbapenems like Ertapenem. Patent CN100457760C introduces a refined preparation method for Ertapenem Sodium Salt that addresses longstanding challenges in yield stability and operational complexity. This technical disclosure outlines a strategic two-step reaction sequence that leverages a one-pot deprotection strategy, fundamentally altering the economic and logistical landscape for manufacturers. By utilizing readily available intermediates such as [4R, 5S, 6S, 8R]-3-[(diphenoxyphosphono)oxy]-6-(1-hydroxyethyl)-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid (4-nitrophenyl)methyl ester, the process achieves a streamlined workflow. The innovation lies not just in the chemical transformation but in the holistic approach to process engineering, where the elimination of intermediate isolation steps directly correlates to reduced waste generation and lower capital expenditure on processing equipment. For stakeholders evaluating reliable ertapenem sodium salt supplier options, understanding the underlying chemistry of this patent is essential for assessing long-term supply security.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those referenced in US patent applications, typically suffer from fragmented synthetic designs that introduce unnecessary inefficiencies. Traditional routes often require the synthesis of side chains where the secondary amine protecting group must be removed in a distinct, isolated step prior to coupling. This fragmentation leads to extended reaction timelines and increases the exposure of sensitive beta-lactam intermediates to potentially degrading conditions. Furthermore, conventional processes frequently rely on expensive solvents and alkalizing agents that drive up the variable cost of goods sold (COGS). The necessity for multi-step crystallization and purification between each transformation not only consumes significant volumes of organic solvents but also results in cumulative yield losses. From a supply chain perspective, these complex workflows create bottlenecks, making it difficult to respond rapidly to market demand fluctuations for high-purity carbapenem intermediates.
The Novel Approach
The methodology described in CN100457760C represents a paradigm shift by integrating the deprotection of both the carboxyl group and the secondary amine into a single catalytic event. By designing the starting materials with compatible protecting groups (specifically the p-nitrobenzyloxycarbonyl group), the process allows for simultaneous removal under hydrogenation conditions. This consolidation means the reaction system from the initial condensation step can be directly subjected to hydrogenolysis without any intermediate workup or isolation. This "telescoped" approach drastically simplifies the operational protocol, reducing the number of unit operations required. The use of sodium bicarbonate as a mild base during the hydrogenation phase further enhances the stability of the final product, minimizing the formation of degradation impurities that often plague carbapenem synthesis. This design rationalizes the entire production flow, offering a clear pathway for cost reduction in antibiotic manufacturing while maintaining rigorous quality standards.
Mechanistic Insights into Catalytic Hydrogenation and Deprotection
The core of this synthesis relies on a precise nucleophilic substitution followed by a sophisticated catalytic hydrogenolysis. In the first stage, the thiol group of the pyrrolidine side chain acts as a nucleophile, attacking the activated ester of the bicyclic core. The presence of DIPEA (diisopropylethylamine) serves to scavenge the acidic byproducts and maintain the nucleophilicity of the thiol, ensuring high conversion rates even at the low temperatures of -20°C to -60°C required to preserve stereochemical integrity. The subsequent step involves the introduction of palladium on carbon (Pd/C) under a hydrogen atmosphere. Mechanistically, the palladium catalyst facilitates the cleavage of the benzylic C-O and C-N bonds associated with the p-nitrobenzyl (PNZ) and p-nitrobenzyloxycarbonyl protecting groups. This hydrogenolysis is highly chemoselective, leaving the sensitive beta-lactam ring and the double bond within the bicyclic system intact. The concurrent presence of sodium bicarbonate neutralizes the acidic species generated during deprotection, preventing acid-catalyzed ring opening of the beta-lactam, which is a common failure mode in carbapenem chemistry.
Impurity control is intrinsically built into this mechanism through the choice of reagents and purification strategy. The use of specific solvent mixtures like NMP and DMF in the condensation step ensures complete solubility of reactants, preventing localized concentration gradients that could lead to oligomerization or side reactions. Following hydrogenation, the protocol employs a rigorous purification sequence involving activated carbon treatment to remove trace metal catalysts and colored impurities. The final isolation via evaporative crystallization from acetone and propanol mixtures exploits the differential solubility of the target sodium salt versus potential organic impurities. This crystallization step is critical for achieving the high-purity ertapenem sodium salt specifications required for pharmaceutical applications, effectively filtering out unreacted starting materials and minor byproducts without the need for chromatographic separation, which is impractical at scale.
How to Synthesize Ertapenem Sodium Salt Efficiently
Executing this synthesis requires strict adherence to the thermal and atmospheric conditions outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction mixture under an inert nitrogen atmosphere to prevent oxidation of the thiol component. Temperature control is paramount; the initial mixing must occur at 0°C, followed by a rapid cooldown to the reaction window of -20°C to -60°C before the addition of the base. Detailed standard operating procedures for scaling this reaction, including specific addition rates and quenching protocols, are essential for technology transfer.
- Condense the phosphonate ester intermediate with the protected pyrrolidine thiol side chain using DIPEA in a DMF/NMP solvent system at low temperatures (-20°C to -60°C).
- Without isolating the intermediate, perform catalytic hydrogenation using Pd/C and sodium bicarbonate under 5-20 atm hydrogen pressure to simultaneously remove protecting groups.
- Purify the crude product through activated carbon treatment, solvent extraction, and evaporative crystallization using acetone and propanol mixtures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the technical advantages of this patent translate directly into tangible business benefits. The primary value driver is the substantial simplification of the manufacturing process, which inherently lowers the barrier to entry for production and reduces the operational overhead associated with complex multi-step syntheses. By eliminating the isolation of unstable intermediates, the process reduces the risk of material loss due to degradation, thereby improving the overall mass balance of the production run. This efficiency gain allows for a more competitive pricing structure without compromising on the quality of the final API intermediate.
- Cost Reduction in Manufacturing: The consolidation of deprotection steps into a single hydrogenation reaction eliminates the need for separate reagent purchases, solvent exchanges, and drying cycles associated with intermediate isolation. The use of sodium bicarbonate as a base is significantly more economical than specialized organic bases often used in fine chemical synthesis. Furthermore, the reduction in unit operations decreases energy consumption for heating, cooling, and agitation, leading to a lower carbon footprint and reduced utility costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: The raw materials specified in this route, such as the phosphonate ester and the protected pyrrolidine thiol, are derived from established supply chains commonly used for other carbapenems like Meropenem. This commonality ensures that sourcing risks are minimized, as these precursors are commercially available from multiple vendors. The robustness of the one-pot method also means that production schedules are less susceptible to delays caused by intermediate quality failures, ensuring a more consistent and predictable delivery timeline for downstream formulation partners.
- Scalability and Environmental Compliance: The reliance on crystallization rather than chromatography for purification makes this process inherently scalable from pilot plant to commercial tonnage production. Crystallization is a standard unit operation in the pharmaceutical industry that is easily validated and controlled. Additionally, the process generates less hazardous waste compared to traditional methods that require extensive solvent swaps and silica gel treatments. The ability to recycle solvents like DMF and NMP further aligns with modern environmental, social, and governance (ESG) goals, facilitating regulatory approval in stringent markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating technology transfer and for procurement teams negotiating supply agreements.
Q: How does this patent method reduce production costs compared to conventional routes?
A: The method eliminates the need for isolating unstable intermediates and combines two deprotection steps into a single hydrogenation reaction. This significantly reduces solvent consumption, operational time, and the usage of expensive reagents required for multi-step purification.
Q: What are the critical reaction conditions for ensuring high purity?
A: Maintaining strict temperature control between -20°C and -60°C during the condensation phase is crucial to prevent side reactions. Additionally, the use of sodium bicarbonate as a mild alkalizing agent during hydrogenation helps preserve the sensitive beta-lactam ring structure.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It utilizes a one-pot technique that simplifies equipment requirements and employs evaporative crystallization, a robust purification method that is easily transferable from pilot plant to commercial tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ertapenem Sodium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of CN100457760C are fully realized in practice. We maintain stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to monitor every batch for impurities and residual solvents. Our commitment to quality assurance means that every gram of Ertapenem Sodium Salt we produce meets the exacting standards required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic impact of switching to this more efficient manufacturing method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of our high-quality intermediates into your final drug product manufacturing.
