Advanced Refining Technology for Tebipenem Pivoxil Side Chain: Enhancing Purity and Commercial Scalability
Advanced Refining Technology for Tebipenem Pivoxil Side Chain: Enhancing Purity and Commercial Scalability
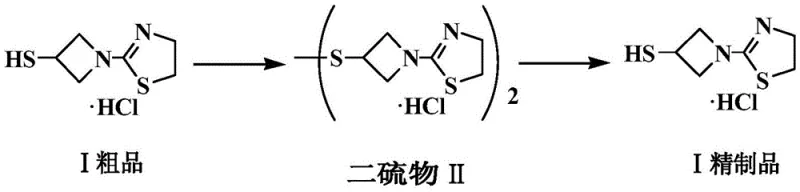
The pharmaceutical industry constantly seeks robust methodologies to enhance the purity and stability of critical antibiotic intermediates, particularly for next-generation carbapenems. Patent CN107445950B introduces a groundbreaking refining method for the tebipenem pivoxil side chain, specifically addressing the persistent challenges associated with 3-mercapto-1-(1,3-thiazoline-2-yl)-azetidine hydrochloride. This technical disclosure outlines a sophisticated oxidation-reduction strategy that transforms the unstable crude thiol into a stable disulfide intermediate, facilitating superior impurity removal before regenerating the final high-purity product. For global procurement leaders and R&D directors, this process represents a significant leap forward in manufacturing reliability, offering a pathway to overcome the hygroscopic nature and chemical instability that have historically plagued the direct physical refinement of this essential carbapenem building block.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of tebipenem ester side chains has relied heavily on direct physical refining techniques, primarily involving multiple crystallizations using complex mixed solvent systems. These conventional approaches suffer from inherent thermodynamic and kinetic limitations, often resulting in suboptimal yields and inconsistent product quality due to the extreme hygroscopicity of the crude material. The crude tebipenem ester side chain is notoriously unstable, readily absorbing moisture from the environment which triggers degradation pathways and complicates the crystallization process. Furthermore, traditional solvent-based purification struggles to effectively separate structurally similar impurities, leading to products that may meet basic specifications but fail to achieve the stringent purity profiles required for modern oral carbapenem formulations. The reliance on large volumes of mixed solvents not only increases operational costs but also generates significant waste streams, creating environmental compliance burdens for large-scale manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a chemical transformation strategy that fundamentally alters the physical properties of the intermediate to enable efficient purification. By oxidizing the crude thiol into a disulfide derivative, the process converts a highly polar, moisture-sensitive compound into a more stable, less polar species that is amenable to precise separation techniques. This chemical switch allows for the effective removal of impurities that are otherwise inseparable from the thiol form, leveraging differences in solubility and precipitation behavior between the disulfide and contaminant species. Following purification, the disulfide is quantitatively reduced back to the desired thiol hydrochloride salt, delivering a refined product with exceptional chemical stability and purity. This oxidative-refining-reductive cycle effectively bypasses the limitations of direct crystallization, providing a robust platform for industrial scale-up.
Mechanistic Insights into Oxidation-Reduction Purification
The core mechanistic advantage of this refining process lies in the reversible redox chemistry of the azetidine-thiazoline scaffold, which serves as a temporary protecting group strategy for the reactive mercapto functionality. In the initial oxidation step, typically catalyzed by iodine species or iodates in an aqueous medium, two molecules of the crude thiol couple to form the corresponding disulfide bond. This dimerization significantly reduces the molecule's polarity and hydrogen-bonding capacity, thereby altering its solvation shell and crystallization kinetics. The disulfide intermediate exhibits markedly different solubility characteristics compared to the starting thiol and its associated impurities, allowing for selective precipitation upon pH adjustment to alkaline conditions. This phase separation is critical, as it physically isolates the desired intermediate from water-soluble byproducts and organic-soluble impurities that remain in the mother liquor or are removed during the extraction phase.
Following the isolation of the purified disulfide, the reduction step employs phosphine-based reducing agents, such as triphenylphosphine or tributylphosphine, to cleave the disulfide bond and regenerate the free thiol. The mechanism involves the nucleophilic attack of the phosphine on the sulfur atom, forming a phosphine oxide byproduct while releasing the thiolate anion, which is subsequently protonated by hydrochloric acid to form the stable hydrochloride salt. This reduction is highly chemoselective, leaving the sensitive beta-lactam-like azetidine ring and the thiazoline moiety intact, which is crucial for maintaining the biological activity of the final antibiotic. The use of specific solvent systems during reduction and subsequent crystallization, such as ethanol combined with anti-solvents like isopropyl ether, ensures that the regenerated thiol precipitates in a highly crystalline form, effectively excluding any remaining trace impurities and locking in the high purity profile achieved during the disulfide stage.
How to Synthesize Tebipenem Pivoxil Side Chain Efficiently
The synthesis protocol outlined in the patent provides a clear, four-stage operational framework designed for reproducibility and high throughput in a GMP environment. The process begins with the dissolution of the crude starting material in water, followed by controlled oxidation to generate the disulfide precipitate, which is then isolated via filtration. The second phase involves the dissolution of this purified intermediate in an alcohol solvent, where it undergoes reduction in the presence of acid and a phosphine reagent. The final stage focuses on the crystallization of the reduced product through a careful temperature gradient and anti-solvent addition strategy to maximize recovery and crystal quality. Detailed standardized operating procedures regarding stoichiometry, temperature control, and solvent ratios are essential for replicating the high yields reported in the examples.
- Dissolve crude tebipenem pivoxil side chain in water and oxidize using an agent like potassium iodate with a catalyst to form disulfide.
- Extract with organic solvent, adjust aqueous phase to alkaline pH to precipitate the disulfide, then filter and dry.
- Dissolve the purified disulfide in a solvent like ethanol, add hydrochloric acid and a reducing agent like triphenylphosphine.
- Concentrate the mixture, dissolve the concentrate in a first solvent, cool, and add a second anti-solvent to crystallize the refined product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refining methodology offers substantial strategic benefits that extend beyond mere technical performance metrics. The shift from complex mixed-solvent crystallization to a defined chemical purification route significantly simplifies the raw material inventory, reducing the need for managing diverse and potentially hazardous solvent blends. By stabilizing the intermediate as a disulfide, the process mitigates the risks associated with the degradation of stock during storage and transit, ensuring a more reliable supply of quality-assured intermediates for downstream API synthesis. This enhanced stability translates directly into reduced waste generation and lower disposal costs, aligning with increasingly stringent global environmental regulations and sustainability goals within the fine chemical sector.
- Cost Reduction in Manufacturing: The elimination of extensive mixed-solvent systems and the improvement in overall process yield contribute to a significant reduction in the cost of goods sold. By avoiding the losses inherent in low-yield direct crystallization methods, manufacturers can maximize the output from each batch of crude starting material, effectively lowering the unit cost of the refined side chain. Furthermore, the use of commercially available and inexpensive reagents such as potassium iodate and triphenylphosphine ensures that the input costs remain predictable and manageable, avoiding the volatility associated with specialized or proprietary purification media.
- Enhanced Supply Chain Reliability: The robustness of the oxidation-reduction cycle ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted production schedules for the final antibiotic drug product. The ability to produce a non-hygroscopic, stable intermediate reduces the logistical complexities of storage and handling, minimizing the risk of supply disruptions caused by material degradation. This reliability allows supply chain planners to optimize inventory levels with greater confidence, knowing that the refined side chain will maintain its specifications over extended periods without requiring special atmospheric controls.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations such as aqueous oxidation, filtration, and solvent crystallization that are easily transferred from pilot plant to commercial manufacturing scales. The reduction in solvent usage and the ability to recycle certain aqueous streams contribute to a smaller environmental footprint, facilitating easier permitting and compliance with green chemistry initiatives. This scalability ensures that the supply of this critical carbapenem intermediate can be rapidly expanded to meet surging market demand without compromising on quality or regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this refining technology, derived from the specific advantages and procedural details disclosed in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines. The answers provided reflect the empirical data and mechanistic explanations found within the intellectual property, offering a transparent view of the method's capabilities and constraints.
Q: Why is direct crystallization insufficient for purifying tebipenem pivoxil side chain?
A: Direct physical refining often results in low yields and poor quality because the crude product is extremely hygroscopic and chemically unstable, making it difficult to remove specific impurities through simple solvent mixing.
Q: What represents the key innovation in this refining method compared to prior art?
A: The key innovation is the conversion of the unstable thiol crude product into a stable disulfide intermediate, which allows for effective impurity removal before reducing it back to the high-purity thiol form.
Q: Which reducing agents are preferred for the final step of this synthesis?
A: Triphenylphosphine and tributylphosphine are the preferred reducing agents, with triphenylphosphine being particularly effective when used in a molar ratio slightly greater than that of the disulfide intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tebipenem Pivoxil Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics, and we are uniquely positioned to support the commercialization of advanced refining technologies like the one described in CN107445950B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of tebipenem pivoxil side chain meets the exacting standards required by top-tier pharmaceutical companies worldwide.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements and timeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production expenses while enhancing product quality. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete performance metrics and our proven track record in the carbapenem intermediate sector.
