Advanced Manufacturing of Doripenem Intermediate I: A Technical Breakthrough for Global API Supply Chains
Advanced Manufacturing of Doripenem Intermediate I: A Technical Breakthrough for Global API Supply Chains
The pharmaceutical industry continuously demands higher purity standards and more sustainable manufacturing processes for critical antibiotic intermediates. Patent CN102093278B introduces a transformative preparation process for Doripenem Intermediate I, a key side-chain precursor for the potent carbapenem antibiotic Doripenem. This technology addresses longstanding challenges in carbapenem synthesis by replacing hazardous solvents and expensive reagents with eco-friendly alternatives, achieving a remarkable purity increase from 90.5% to 99.5%. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this process represents a significant leap forward in both quality assurance and cost efficiency. The structural integrity of the target molecule, featuring a pyrrolidinyl group substituted with methyl and aminosulfonamide moieties, is preserved through meticulous control of reaction conditions.
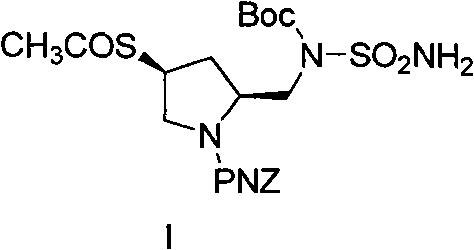
Doripenem is renowned for its broad-spectrum activity against Gram-positive and Gram-negative bacteria, including Pseudomonas aeruginosa, making its supply chain vital for global health security. The intermediate described herein, (2S,4S)-4-acetylthio-2-[[N-aminosulfonyl-N-(tert-butoxycarbonyl)amino]methyl]pyrrolidine-1-carboxylic acid p-nitrobenzyl ester, serves as the chiral side chain that confers specific biological activity. Traditional manufacturing routes have struggled with low overall yields ranging between 45% and 55%, primarily due to inefficient purification and the use of unstable intermediates. By implementing the optimized route detailed in this patent, manufacturers can secure a consistent supply of high-purity materials, thereby reducing lead time for high-purity pharmaceutical intermediates and ensuring robust production schedules for finished drug products.
The Limitations of Conventional Methods vs. The Novel Approach
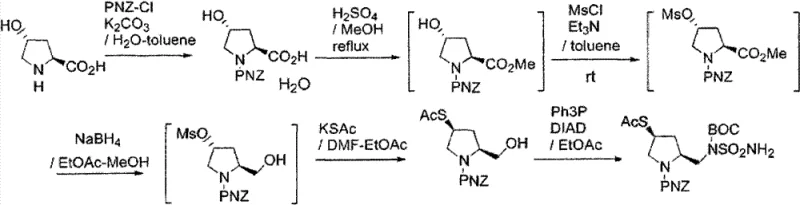
The Limitations of Conventional Methods
Historically, the industrial synthesis of this critical intermediate relied on two primary routes that shared significant operational defects. As illustrated in the prior art, these conventional methods often employed toxic solvents such as toluene, posing severe environmental and occupational health risks during scale-up. Furthermore, the reliance on expensive and hazardous reagents like triphenylphosphine and diisopropyl azodicarboxylate (DIAD) in Mitsunobu-type couplings not only inflated production costs but also generated difficult-to-remove phosphine oxide byproducts. The lack of effective isolation and purification between steps in these one-pot strategies resulted in the accumulation of impurities, capping the final product purity at approximately 90.5%. This level of purity is often insufficient for direct use in sensitive beta-lactam coupling reactions without extensive and yield-losing chromatographic purification.
The Novel Approach
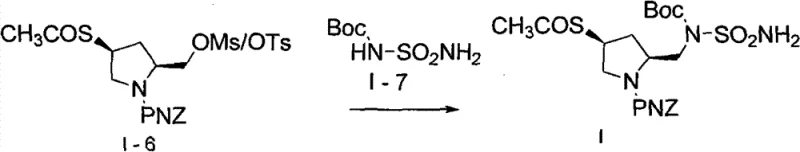
The innovative process disclosed in CN102093278B fundamentally reengineers the synthetic pathway to eliminate these bottlenecks. A pivotal improvement involves the substitution of toluene with ethyl acetate, a greener solvent that reduces toxicity and simplifies waste management while maintaining excellent solubility profiles for the intermediates. Additionally, the process replaces the costly Mitsunobu reagents in the final coupling step with a specialized nucleophilic substitution strategy using N-BOC-sulfamide. This modification not only drastically simplifies the workup procedure by avoiding phosphine oxide removal but also enhances the overall atom economy. By separating the reaction steps and optimizing conditions for each transformation, the new route achieves a total yield exceeding 75%, representing a substantial improvement in material throughput and cost reduction in API manufacturing.
Mechanistic Insights into Optimized Reduction and Coupling Chemistry
A deeper analysis of the reaction mechanism reveals why this process offers superior control over stereochemistry and functional group tolerance. In the reduction step converting the mesylated ester to the hydroxymethyl intermediate, the patent specifies the use of potassium borohydride (KBH4) instead of the more common sodium borohydride. While both are hydride donors, KBH4 offers distinct advantages in this specific solvent system (ethyl acetate/ethanol), providing a more controlled reduction rate that minimizes side reactions. The molar ratio is tightly controlled between 1.8 and 2.3 equivalents, ensuring complete conversion of the ester without excessive reagent waste. This precise stoichiometric balance is crucial for maintaining the integrity of the adjacent chiral centers on the pyrrolidine ring, preventing epimerization that could compromise the biological activity of the final antibiotic.
The most critical mechanistic innovation occurs in the final coupling step where the primary alcohol is displaced by the sulfonamide nitrogen. Typically, the acetyl thiol protecting group at the 4-position is highly susceptible to nucleophilic attack or base-catalyzed hydrolysis under coupling conditions. To counteract this, the process employs sterically hindered organic bases such as diisopropylethylamine (DIEA) or 2,2,6,6-tetramethylpiperidine (TEMP). These bulky bases facilitate the deprotonation of the sulfonamide without aggressively attacking the thioester moiety. Furthermore, the inclusion of activated molecular sieves in the reaction mixture plays a dual role: they sequester trace water that could hydrolyze the sensitive intermediates and help drive the equilibrium towards product formation. This sophisticated control of the reaction environment ensures that the thiol protecting group remains intact until the final deprotection stage in the API synthesis.
How to Synthesize Doripenem Intermediate I Efficiently
The synthesis of this complex chiral building block requires strict adherence to the optimized parameters regarding temperature, solvent ratios, and reagent addition rates to ensure reproducibility at scale. The process begins with the N-protection of 4-hydroxypyrrolidine-2-carboxylic acid using p-nitrobenzyloxycarbonyl chloride (PNZ-Cl) in a biphasic system, followed by esterification and mesylation. Each intermediate is isolated and purified to remove carryover impurities before proceeding to the next step, a deviation from the telescoped processes of the past that significantly boosts final purity. The detailed standardized synthesis steps, including specific temperatures (0-5°C for protection, 60°C for esterification) and workup procedures (acid/base washes, recrystallization), are outlined below to guide process chemists in implementation.
- Protect 4-hydroxypyrrolidine-2-carboxylic acid with PNZ-Cl in ethyl acetate/water to form Intermediate I-1.
- Esterify I-1 with methanol/sulfuric acid, mesylate the hydroxyl group, and reduce the ester to alcohol using potassium borohydride.
- Substitute the mesylate with potassium thioacetate, activate the primary alcohol, and couple with N-BOC-sulfamide using sterically hindered bases.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages beyond mere technical specifications. The shift away from hazardous materials like toluene and triphenylphosphine reduces the regulatory burden associated with handling and disposing of dangerous chemicals, thereby lowering compliance costs and insurance premiums. Moreover, the simplified purification workflow, which eliminates the need for column chromatography in favor of crystallization, significantly shortens the production cycle time. This efficiency gain allows for faster turnaround on orders and enhances the reliability of supply, which is critical for meeting the demands of the global antibiotic market. The ability to source this intermediate from a supplier utilizing such an optimized route ensures a more stable and cost-effective supply chain for downstream API manufacturers.
- Cost Reduction in Manufacturing: The replacement of sodium borohydride with potassium borohydride alone drives down raw material costs by nearly 30%, as explicitly noted in the patent data. When combined with the elimination of expensive coupling reagents like DIAD and triphenylphosphine, the overall cost of goods sold (COGS) is significantly reduced. Additionally, the higher overall yield (>75% vs 45-55%) means less starting material is required to produce the same amount of final product, further amplifying the economic benefits and allowing for more competitive pricing strategies in the marketplace.
- Enhanced Supply Chain Reliability: By utilizing readily available and less regulated solvents like ethyl acetate, the process mitigates risks associated with supply chain disruptions for controlled or hazardous chemicals. The robust nature of the intermediates, achieved through effective isolation and purification at each stage, ensures that the material can withstand transportation and storage without significant degradation. This stability is paramount for maintaining inventory levels and ensuring continuous production lines, reducing the risk of stockouts that could halt the manufacturing of life-saving antibiotics.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard unit operations such as filtration, distillation, and crystallization that are easily transferable from pilot plant to commercial scale. The minimization of toxic waste streams aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes and reducing the carbon footprint of the manufacturing facility. This commitment to green chemistry principles not only protects the environment but also enhances the corporate social responsibility profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Doripenem Intermediate I. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these nuances is essential for evaluating the feasibility of integrating this intermediate into your existing manufacturing workflows.
Q: How does the new process improve the purity of Doripenem Intermediate I compared to conventional methods?
A: The novel process replaces the traditional one-pot method with discrete, optimized steps and eliminates toxic reagents like triphenylphosphine. This strategic modification, combined with a specific recrystallization protocol using isopropanol/water, elevates product purity from 90.5% to over 99.5%, significantly reducing impurity profiles for downstream API synthesis.
Q: What are the cost-saving advantages of using potassium borohydride in this synthesis?
A: By substituting sodium borohydride with potassium borohydride in the reduction step, the process achieves a raw material cost reduction of nearly 30%. Furthermore, the specific molar ratio optimization (1.8-2.3 eq) minimizes waste treatment costs associated with acidic quenching, offering substantial economic benefits for large-scale manufacturing.
Q: Why is the final coupling step critical for the stability of the thiol protecting group?
A: The acetyl thiol protecting group is highly labile under standard coupling conditions. The patented method utilizes sterically hindered bases like DIEA or TEMP alongside activated molecular sieves. This unique combination facilitates the nucleophilic substitution while preventing the premature deprotection of the thiol group, ensuring high yield and structural integrity of the final intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doripenem Intermediate I Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of advanced antibiotics like Doripenem. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patented process are fully realized in practical manufacturing settings. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch meets the 99.5% purity benchmark required for GMP API synthesis. Our commitment to quality assurance guarantees that our clients receive materials that are consistent, safe, and ready for immediate use in their downstream processes.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the efficiency and profitability of your pharmaceutical supply chain.
