Advanced Manufacturing of 1,4-Pregnadiene-17α,21-dihydroxy-3,20-dione-17α,21-diacetate for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and efficient pathways for the production of critical corticosteroid intermediates, specifically 1,4-pregnadiene-17α,21-dihydroxy-3,20-dione-17α,21-diacetate, which serves as a pivotal precursor for high-value drugs such as prednisolone, dexamethasone, and budesonide. Patent CN1724554A introduces a groundbreaking semi-synthetic methodology that addresses the longstanding challenges of resource scarcity and complex processing associated with traditional steroid manufacturing. By leveraging 1,4-androstadiene-3,17-diketone (ADD) derived from microbial fermentation of phytosterols, this innovation bypasses the reliance on diminishing natural resources like Dioscorea yams. The disclosed process not only streamlines the reaction sequence but also optimizes yield and operational simplicity, making it a highly attractive candidate for reliable pharmaceutical intermediates supplier networks aiming to secure stable supply chains for anti-inflammatory therapeutics.
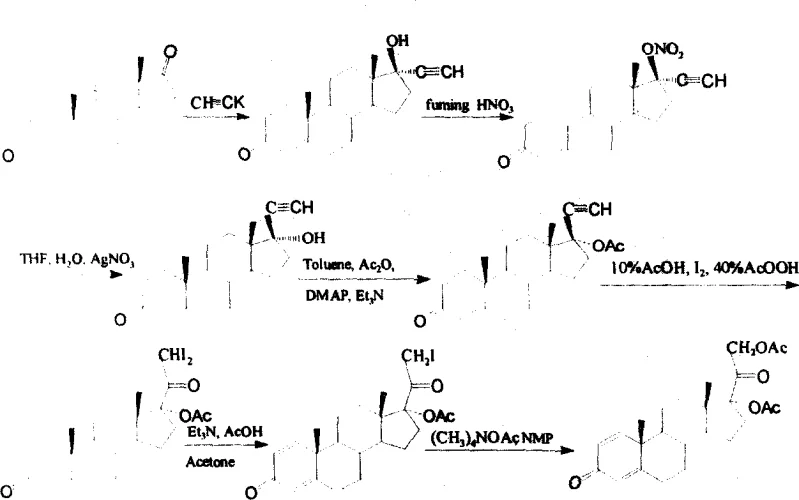
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex corticosteroid intermediates has been plagued by inefficient multi-step routes that rely heavily on scarce natural precursors or excessively long synthetic sequences. Prior art, such as the method described by Issei et al., necessitates a cumbersome seven-step reaction pathway starting from ADD to achieve the target 17,21-diacetate structure. These conventional approaches often involve specialized reagents that are difficult to source in bulk, harsh reaction conditions that compromise safety, and cumulative yield losses that render the final product economically unviable for mass production. Furthermore, the reliance on wild-harvested plant materials for steroid backbones has led to ecological strain and inconsistent raw material quality, creating significant bottlenecks for procurement managers tasked with maintaining cost-effective manufacturing operations in the face of fluctuating agricultural outputs.
The Novel Approach
In stark contrast, the methodology outlined in CN1724554A presents a sophisticated yet practical five-step synthesis that dramatically reduces operational complexity while enhancing overall throughput. By strategically utilizing fuming nitric acid for nitration and silver nitrate-catalyzed rearrangement, the process achieves high conversion rates at each stage, with reported yields reaching up to 96% in the nitration step and maintaining robust efficiency throughout the sequence. This novel approach eliminates the need for obscure reagents, instead favoring industrially standard solvents like THF, acetone, and N-methyl pyrrolidone (NMP), which facilitates easier scale-up and waste management. For supply chain heads, this translates to a more resilient production model where the reduction in unit operations directly correlates to reduced lead times and lower overhead costs, ensuring a consistent flow of high-purity intermediates to downstream API manufacturers.
Mechanistic Insights into Silver-Catalyzed Rearrangement and Functionalization
The core chemical innovation lies in the strategic functionalization of the D-ring of the steroid nucleus, specifically transforming the 17-position through a sequence of ethynylation, nitration, and subsequent rearrangement. The process initiates with the nucleophilic addition of a metal acetylide to the 17-ketone of ADD, forming a 17α-ethynyl-17β-hydroxy intermediate under controlled low-temperature conditions in THF. This is followed by a critical nitration step where fuming nitric acid in acetic anhydride converts the 17β-hydroxyl group into a nitrate ester, a transformation that activates the molecule for the subsequent carbon-carbon bond cleavage and rearrangement. The mechanistic elegance continues with the use of N-bromosuccinimide (NBS) and silver nitrate, which facilitates the formation of a bromo-alkyne species that undergoes a silver-mediated rearrangement in NMP. This rearrangement effectively extends the carbon skeleton from a C19 androstane to a C21 pregnane framework, installing the crucial C21 functionality required for corticosteroid activity.
Impurity control is meticulously managed through the selection of specific reaction conditions and purification techniques at each stage. For instance, the hydrolysis of the acetate and nitrate groups is carefully timed to prevent over-degradation of the sensitive enone system in the A-ring. The use of column chromatography with specific eluent systems, such as petroleum ether and ethyl acetate mixtures, ensures that side products like unreacted starting materials or over-brominated species are effectively removed. Additionally, the final acetylation step using potassium acetate in acetone is optimized to selectively substitute the C21-bromine without affecting the 17α-acetate, thereby preserving the stereochemical integrity of the molecule. This rigorous attention to mechanistic detail ensures that the final product meets the stringent purity specifications required for pharmaceutical applications, minimizing the burden on downstream purification processes.
How to Synthesize 1,4-Pregnadiene-17α,21-dihydroxy-3,20-dione-17α,21-diacetate Efficiently
The synthesis protocol described in the patent offers a clear roadmap for laboratory and pilot-scale production, emphasizing precise temperature control and stoichiometric ratios to maximize yield. The procedure begins with the preparation of the ethynyl intermediate at temperatures between -20°C and 10°C, followed by a careful nitration step that requires maintaining the reaction mixture below -20°C to prevent decomposition. Subsequent bromination and rearrangement steps are conducted at ambient to moderate temperatures (15-50°C), allowing for manageable heat dissipation and safe handling of reactive intermediates. For a comprehensive understanding of the specific molar ratios, solvent volumes, and workup procedures required to replicate these results, please refer to the detailed standardized synthesis guide provided below.
- React ADD with metal acetylide in THF at low temperature to form the 17α-ethynyl intermediate.
- Treat the hydroxy intermediate with fuming nitric acid in acetic anhydride to generate the nitrate ester.
- Perform bromination using NBS and silver nitrate, followed by rearrangement in NMP with acetic anhydride to form the C21-bromo ketone.
- Complete the synthesis by substituting the bromine with acetate using potassium acetate in acetone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this five-step synthesis route offers substantial strategic benefits for organizations focused on cost reduction in pharmaceutical intermediates manufacturing. The elimination of two entire reaction steps compared to prior art methods inherently reduces the consumption of solvents, reagents, and energy, leading to a significantly lowered cost of goods sold (COGS). Moreover, the high yields reported in the patent examples suggest a drastic improvement in material efficiency, meaning less raw material is wasted per kilogram of final product. For procurement managers, this efficiency gain provides a buffer against raw material price volatility and allows for more competitive pricing strategies in the global market for corticosteroid APIs.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive and specialized reagents often required in longer synthetic routes, replacing them with commodity chemicals like acetic anhydride and potassium acetate. By reducing the number of isolation and purification stages, the process minimizes labor costs and equipment usage time, resulting in substantial cost savings without compromising product quality. The high overall yield further amplifies these savings by maximizing the output from each batch of starting material, effectively lowering the unit cost of the intermediate.
- Enhanced Supply Chain Reliability: Utilizing ADD as a starting material leverages the well-established microbial fermentation supply chain for phytosterols, which is far more stable and scalable than relying on agricultural extraction from yams. The use of common organic solvents such as THF, acetone, and toluene ensures that supply disruptions due to specialty chemical shortages are minimized. This robustness in raw material sourcing translates to improved delivery reliability and the ability to scale production rapidly in response to market demand fluctuations.
- Scalability and Environmental Compliance: The reaction conditions described are amenable to large-scale batch processing, with temperatures and pressures that do not require exotic high-pressure reactors or cryogenic facilities beyond standard industrial capabilities. The reduction in step count inherently reduces the volume of chemical waste generated, simplifying effluent treatment and aiding in compliance with increasingly strict environmental regulations. This environmental efficiency not only reduces disposal costs but also aligns with the sustainability goals of modern pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and process advantages. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines. The answers provided reflect the specific chemical transformations and operational parameters defined in the intellectual property.
Q: What is the primary advantage of the synthesis method in CN1724554A?
A: The primary advantage is the reduction of the synthetic route from seven steps to five steps, significantly improving overall yield and reducing processing time compared to conventional methods.
Q: What are the critical reagents used in the rearrangement step?
A: The rearrangement step utilizes N-Methyl pyrrolidone (NMP) as a solvent, silver nitrate (AgNO3) as a catalyst, and acetic anhydride to facilitate the conversion to the 21-bromo intermediate.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the process is designed for industrial suitability, utilizing accessible raw materials like ADD and standard solvents such as THF, acetone, and toluene.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Pregnadiene-17α,21-dihydroxy-3,20-dione-17α,21-diacetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and manufacture of life-saving corticosteroid medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay at every stage of production.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this more efficient manufacturing process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency, quality, and mutual success.
