Advanced Photocatalytic Synthesis of o-Trifluoromethyl Phenylamine for Commercial Scale-up
Advanced Photocatalytic Synthesis of o-Trifluoromethyl Phenylamine for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to introduce fluorine atoms into molecular scaffolds, particularly the trifluoromethyl group, due to its profound impact on metabolic stability and bioavailability. Patent CN103553857A presents a groundbreaking methodology for the preparation of o-trifluoromethyl phenylamine and its derivatives, utilizing a visible-light-driven photocatalytic system that operates under exceptionally mild conditions. This technology represents a significant leap forward from traditional thermal methods, offering a streamlined approach that eliminates the need for cumbersome protecting group strategies on the amino functionality. By leveraging the unique reactivity of trivalent iodine reagents in conjunction with an iridium-based photocatalyst, this process achieves direct ortho-selective trifluoromethylation with high precision. For R&D directors and procurement specialists, understanding the nuances of this patent is critical for evaluating new supply chains for high-purity pharmaceutical intermediates. The ability to functionalize aniline derivatives directly without prior modification reduces both material costs and processing time, aligning perfectly with modern green chemistry principles and cost reduction in fine chemical manufacturing initiatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group onto an aromatic ring, especially in the ortho position relative to an amino group, has been fraught with synthetic challenges that hinder efficient commercial production. Traditional methods often necessitate harsh reaction conditions, including elevated temperatures and the use of strong bases or aggressive fluorinating agents that can compromise sensitive functional groups elsewhere in the molecule. A major bottleneck in conventional synthesis is the requirement for protecting the amino group to prevent side reactions or oxidation, which adds multiple steps to the overall sequence, including protection and subsequent deprotection. These additional steps not only increase the consumption of raw materials and solvents but also generate substantial amounts of chemical waste, thereby escalating the environmental footprint and disposal costs associated with the manufacturing process. Furthermore, achieving regioselectivity in traditional electrophilic aromatic substitution can be difficult, often leading to mixtures of isomers that require energy-intensive purification techniques to separate. The cumulative effect of these limitations is a higher cost of goods sold and longer lead times for high-purity pharmaceutical intermediates, creating supply chain vulnerabilities for downstream drug manufacturers who rely on consistent quality and availability.
The Novel Approach
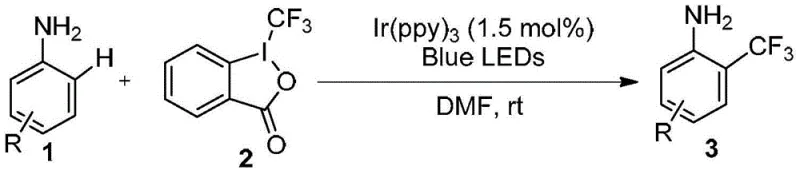
In stark contrast to these legacy methods, the technology disclosed in patent CN103553857A utilizes a photoredox catalytic cycle that enables direct C-H functionalization under ambient conditions, fundamentally reshaping the economic and operational landscape of trifluoromethylation. By employing tris(2-phenylpyridine) iridium [Ir(ppy)3] as a photocatalyst and a specific trivalent iodine reagent as the trifluoromethyl source, the reaction proceeds efficiently at room temperature upon irradiation with blue LEDs. This mild thermal profile eliminates the safety risks associated with high-pressure or high-temperature reactors, facilitating safer commercial scale-up of complex polymer additives and pharmaceutical building alike. Crucially, the method tolerates the free amino group, meaning that expensive and time-consuming protection-deprotection sequences are entirely obviated, resulting in a drastically simplified synthetic route. The use of visible light as the energy source is not only energy-efficient but also allows for precise control over the reaction initiation and termination, enhancing reproducibility across different batch sizes. This innovative approach directly addresses the pain points of modern chemical manufacturing by offering a pathway that is both atom-economical and operationally simple, making it an attractive option for reliable agrochemical intermediate supplier networks seeking to optimize their portfolios.
Mechanistic Insights into Ir(ppy)3-Catalyzed Photoredox Trifluoromethylation
The core of this transformative synthesis lies in the sophisticated interplay between the excited state of the iridium photocatalyst and the hypervalent iodine reagent, which generates the reactive trifluoromethyl radical species necessary for the transformation. Upon absorption of blue light photons, the ground state Ir(III) catalyst is promoted to a long-lived excited state capable of engaging in single-electron transfer (SET) processes with the trivalent iodine compound. This electron transfer event triggers the homolytic cleavage of the carbon-iodine bond within the reagent, releasing a highly reactive trifluoromethyl radical while regenerating the catalyst through a subsequent oxidative or reductive quenching cycle. The generated radical then adds selectively to the aromatic ring of the aniline derivative, preferentially at the ortho position due to electronic and steric factors directed by the amino group and the catalyst system. Following radical addition, the resulting cyclohexadienyl radical intermediate undergoes oxidation and deprotonation to restore aromaticity, yielding the desired o-trifluoromethyl aniline product. Understanding this mechanism is vital for R&D teams as it highlights the specificity of the reaction, ensuring that the impurity profile remains clean and manageable compared to non-catalytic thermal methods.
Beyond the primary catalytic cycle, the control of impurities is a critical aspect that determines the commercial viability of any pharmaceutical intermediate synthesis. The mild nature of the photoredox conditions minimizes the formation of thermal degradation byproducts that are common in high-temperature fluorination reactions. Additionally, the high regioselectivity observed in this system reduces the generation of meta- or para-substituted isomers, which are often difficult to remove and can compromise the purity specifications required for API synthesis. The tolerance of the system towards various functional groups, such as esters, halides, and ethers, as demonstrated in the patent examples, further underscores its robustness in handling complex molecular architectures. This level of control over the reaction outcome ensures that the final product meets stringent quality standards with minimal downstream purification effort. For supply chain heads, this translates to more predictable yields and reduced risk of batch failures, thereby enhancing the overall reliability of the supply chain for critical drug substances.
How to Synthesize o-Trifluoromethyl Phenylamine Efficiently
Implementing this patented methodology in a laboratory or pilot plant setting requires careful attention to the specific reaction parameters outlined in the intellectual property to ensure optimal performance and safety. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically using nitrogen or argon to prevent quenching of the excited catalyst by oxygen. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating the high yields reported in the patent examples.
- Prepare the reaction mixture by dissolving aniline derivatives and trivalent iodine reagent in DMF under inert gas protection.
- Add 1.5 mol% of Ir(ppy)3 catalyst and irradiate the solution with blue LEDs at room temperature until conversion is complete.
- Quench the reaction with saturated sodium carbonate, extract with ethyl acetate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this photocatalytic technology offers substantial cost savings and supply chain resilience that go beyond mere chemical efficiency. By eliminating the need for amino protection, the process removes entire unit operations from the manufacturing workflow, which directly correlates to reduced capital expenditure on equipment and lower operational expenses related to labor and utilities. The ability to run reactions at room temperature significantly lowers energy consumption compared to thermal processes that require heating and cooling cycles, contributing to a smaller carbon footprint and alignment with corporate sustainability goals. Furthermore, the use of commercially available starting materials and catalysts ensures that the supply chain is not dependent on exotic or single-source reagents that could pose availability risks. This robustness makes the technology highly suitable for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps results in a significantly reduced number of synthetic operations, which inherently lowers the consumption of solvents, reagents, and processing time. Without the need for harsh thermal conditions, energy costs are drastically simplified, and the wear and tear on reactor equipment are minimized, extending asset life. The high atom economy of the direct C-H functionalization approach means that less raw material is wasted in the form of byproducts, leading to substantial cost savings in material procurement. Additionally, the simplified workup procedure reduces the load on waste treatment facilities, further decreasing the overall operational expenditure associated with environmental compliance and disposal.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available reagents such as DMF and Ir(ppy)3 ensures that the manufacturing process is not vulnerable to supply disruptions of niche chemicals. The mild reaction conditions enhance operational safety, reducing the likelihood of accidents or shutdowns that could interrupt the continuity of supply for critical intermediates. The scalability of the photochemical process, supported by modern flow chemistry technologies, allows for flexible production volumes that can be adjusted to meet fluctuating demand without compromising quality. This flexibility provides procurement managers with greater confidence in securing long-term contracts and maintaining inventory levels for high-purity OLED material or pharmaceutical precursors.
- Scalability and Environmental Compliance: The transition from batch to continuous flow photochemistry is well-supported for this reaction type, facilitating seamless commercial scale-up from kilogram to multi-ton scales. The reduction in hazardous waste generation due to fewer synthetic steps and milder reagents simplifies the regulatory burden associated with environmental permits and emissions reporting. Energy efficiency is maximized through the use of LED light sources, which have long lifespans and low power requirements compared to traditional mercury lamps or heating mantles. This alignment with green chemistry principles not only improves the public image of the manufacturing entity but also future-proofs the supply chain against increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylation technology, based on the specific details and advantageous effects described in the patent documentation. These insights are designed to clarify the feasibility and benefits for stakeholders involved in process development and sourcing decisions.
Q: Does this method require amino group protection?
A: No, one of the primary advantages of this patented process is that the amino group does not require protection, significantly simplifying the synthetic route and reducing waste.
Q: What are the reaction conditions for this trifluoromethylation?
A: The reaction proceeds under mild conditions at room temperature using blue LED irradiation in DMF solvent, avoiding the need for high thermal energy input.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of mild conditions and commercially available catalysts suggests high potential for commercial scale-up of complex pharmaceutical intermediates with improved safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable o-Trifluoromethyl Phenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photocatalytic methodologies like the one described in CN103553857A for the production of next-generation pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial realities. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of o-trifluoromethyl phenylamine derivatives meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of photochemical scale-up, providing our partners with a secure and high-quality supply of critical building blocks.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs, whether you are optimizing an existing route or developing a new synthetic pathway from scratch. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and product quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
