Optimized Industrial Synthesis of 4,4'-Diaminobenzenesulphonamide for High-Performance Dye Manufacturing
Optimized Industrial Synthesis of 4,4'-Diaminobenzenesulphonamide for High-Performance Dye Manufacturing
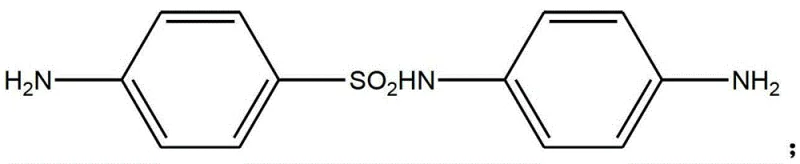
The global demand for high-performance azo dyes, particularly acid blacks like Acid Black 210 and Acid Black 234, necessitates a robust and reliable supply chain for their critical precursors. Patent CN103145593A introduces a refined preparation method for 4,4'-diaminobenzenesulphonamide (CAS 16803-97-7), a pivotal intermediate in the fine chemical sector. This technology addresses historical challenges in synthesis stability and yield by integrating a dual-reagent sulfonation strategy involving chlorosulfonic acid and thionyl chloride. For R&D directors and procurement specialists seeking a reliable dye intermediate supplier, this process offers a compelling value proposition through its ability to deliver consistent quality while minimizing production variability. The structural integrity of the target molecule is paramount for downstream dye performance, and this patented route ensures that the final product exhibits superior stability and color properties essential for textile applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for sulfonamide derivatives often suffer from uncontrolled side reactions during the initial sulfonation phase. When acetanilide is subjected to standard chlorosulfonation conditions without precise modulation, there is a significant risk of hyperoxidation, leading to the formation of complex impurity profiles that are difficult to separate. Furthermore, conventional hydrolysis steps frequently fail to protect the sensitive amino groups from oxidative degradation, resulting in dark-colored products that require extensive and costly purification processes. These inefficiencies not only drive up the cost reduction in dye intermediate manufacturing but also create bottlenecks in supply continuity due to batch-to-batch inconsistency. The reliance on single-reagent sulfonation systems often yields intermediates with lower purity, forcing manufacturers to accept lower overall yields or invest heavily in downstream refining infrastructure to meet stringent market specifications.
The Novel Approach
The methodology disclosed in CN103145593A revolutionizes this landscape by introducing thionyl chloride as a co-reagent during the sulfonation of acetanilide. This strategic modification effectively mitigates the risk of over-oxidation, ensuring the formation of p-acetaminobenzenesulfonyl chloride with exceptional purity. By carefully controlling the molar ratios of acetanilide to chlorosulfonic acid (1:1 to 1:6) and thionyl chloride (1:0.5 to 1:2), the process maximizes conversion efficiency while minimizing waste generation. Additionally, the incorporation of specific reductive agents during the hydrolysis stage acts as a safeguard against amino oxidation, preserving the light color and chemical integrity of the final 4,4'-diaminobenzenesulphonamide. This holistic approach to process design not only enhances the commercial scale-up of complex dye intermediates but also aligns with modern environmental standards by enabling partial wastewater recovery and reducing the load on effluent treatment systems.
Mechanistic Insights into Chlorosulfonation and Condensation Dynamics
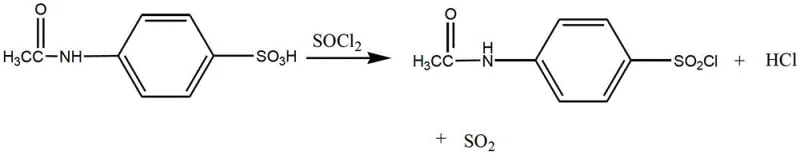
The core of this synthesis lies in the precise execution of the chlorosulfonation reaction, where acetanilide reacts with chlorosulfonic acid and thionyl chloride. As illustrated in the reaction scheme, the presence of thionyl chloride modifies the electrophilic environment, facilitating the selective formation of the sulfonyl chloride group at the para-position without degrading the acetamido protecting group. This step is critical because any degradation at this stage propagates through the entire synthesis, compromising the final yield. The reaction temperature is meticulously maintained between 30°C and 65°C to balance reaction kinetics with thermal stability, preventing the decomposition of the sensitive sulfonyl chloride intermediate. Following this, the intermediate undergoes an ice separation process where precipitation temperatures are controlled between -10°C and 20°C, ensuring the isolation of a high-purity filter cake ready for the subsequent condensation step.
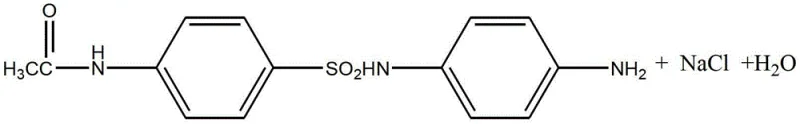
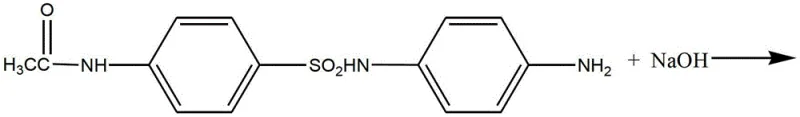
Following the isolation of the sulfonyl chloride, the process moves to the condensation phase with p-phenylenediamine. This nucleophilic substitution occurs under alkaline conditions, where sodium hydroxide serves as an acid binding agent to neutralize the HCl byproduct. The patent specifies a pH range of 4 to 9 and a temperature window of 10°C to 30°C to optimize the reaction rate while preventing the polymerization or degradation of the diamine reactant. The resulting intermediate, Formula (II), retains the acetyl protecting group, which is subsequently removed via hydrolysis. The final hydrolysis step is particularly ingenious; by adding reductive agents like sodium bisulfite or rongalite before neutralization, the process actively scavenges oxidative species that would otherwise convert the free amino groups into nitro derivatives. This mechanistic intervention is the key to achieving the reported 90-93% overall yield and the characteristic light pink to grey-white appearance of the commercial product.
How to Synthesize 4,4'-Diaminobenzenesulphonamide Efficiently
The synthesis of this critical dye intermediate requires strict adherence to thermal and stoichiometric parameters to ensure reproducibility on an industrial scale. The process begins with the careful addition of acetanilide to chlorosulfonic acid, followed by the introduction of thionyl chloride, necessitating robust temperature control systems to manage the exothermic nature of the reaction. Subsequent steps involve precise pH adjustments during condensation and the strategic timing of reductive agent addition during hydrolysis to prevent oxidation. For technical teams looking to implement this route, the detailed standardized synthesis steps outlined below provide a comprehensive guide to achieving the high purity and yield metrics described in the patent literature.
- Perform chlorosulfonation of acetanilide using chlorosulfonic acid and thionyl chloride at controlled temperatures (30-65°C) to form p-acetaminobenzenesulfonyl chloride.
- Execute condensation reaction between the sulfonyl chloride intermediate and p-phenylenediamine under alkaline conditions (pH 4-9) at 10-30°C.
- Conduct hydrolysis of the condensation product at 70-120°C with added reductive agents to prevent oxidation, followed by neutralization and filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible operational efficiencies and risk mitigation. The primary advantage lies in the substantial cost savings achieved through yield optimization; by pushing overall reaction yields to the 90-93% range, manufacturers can significantly reduce the raw material consumption per kilogram of finished product. This efficiency is further compounded by the elimination of expensive purification steps typically required to remove oxidative impurities, thereby streamlining the production timeline and reducing energy consumption. Moreover, the ability to recover and reuse portions of the wastewater generated during the sulfonation dilution step contributes to a more sustainable and cost-effective operation, aligning with increasingly stringent environmental regulations without compromising output.
- Cost Reduction in Manufacturing: The integration of thionyl chloride in the sulfonation step drastically improves the purity of the intermediate, which eliminates the need for costly downstream purification processes often associated with traditional methods. By preventing the formation of hard-to-remove by-products early in the synthesis, the process reduces the consumption of solvents and adsorbents, leading to a leaner and more economical production cycle that directly impacts the bottom line.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream dye manufacturers. The use of readily available industrial raw materials such as acetanilide and chlorosulfonic acid, combined with a process that tolerates slight variations in conditions without catastrophic failure, minimizes the risk of production delays and ensures a steady flow of high-purity dye intermediates to the market.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor configurations and temperature control protocols that are easily transferable from pilot to commercial scale. Furthermore, the inclusion of reductive agents to prevent oxidation not only improves product quality but also reduces the chemical oxygen demand (COD) of the effluent, simplifying wastewater treatment and ensuring compliance with environmental discharge standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4,4'-diaminobenzenesulphonamide. These insights are derived directly from the patented methodology to provide clarity on process capabilities and product specifications. Understanding these details is essential for partners evaluating the feasibility of integrating this intermediate into their own supply chains or product formulations.
Q: What is the primary advantage of using thionyl chloride in this synthesis route?
A: The addition of thionyl chloride during the sulfonation step effectively prevents the over-oxidation of acetanilide, resulting in a higher purity intermediate (Formula I) and significantly reducing the content of unwanted by-products compared to traditional chlorosulfonation methods.
Q: How does the process control impurity formation during hydrolysis?
A: The process incorporates specific reductive agents such as sodium bisulfite or rongalite prior to neutralization. This critical step suppresses the oxidation of amino groups into nitro compounds, ensuring the final product maintains a light color and high chemical stability.
Q: What represents the typical yield and purity for this manufacturing method?
A: Industrial implementation of this patented method consistently achieves an overall reaction yield between 90% and 93%, with product purity ranging from 93% to 99%, making it highly suitable for demanding dye intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Diaminobenzenesulphonamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of the global dye and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN103145593A can be executed with precision and reliability. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every shipment of 4,4'-diaminobenzenesulphonamide supports your manufacturing goals with consistency and excellence.
We invite you to engage with our technical procurement team to discuss how our advanced synthesis capabilities can support your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can enhance your operational efficiency. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the supply of high-performance chemical intermediates.
