Advanced Green Synthesis of Royal Jelly Acid: Scalable Production for Global Markets
Introduction to Next-Generation Royal Jelly Acid Manufacturing
The global demand for high-purity Royal Jelly Acid, chemically known as (E)-10-hydroxy-2-decenoic acid or 10-HDA, continues to surge across the pharmaceutical and nutraceutical sectors due to its potent biological activities. Patent CN102267893B introduces a transformative preparation method that addresses critical bottlenecks in traditional synthesis, specifically focusing on selectivity, catalyst recovery, and environmental safety. This technical breakthrough utilizes 1,8-octanediol as a robust starting material, employing a strategic monoacylation followed by a novel heterogeneous oxidation protocol. By integrating silica-supported Tempo catalysts and ionic liquid-mediated aqueous reactions, this methodology achieves exceptional yields while eliminating the reliance on stoichiometric heavy metal oxidants. For industry leaders seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic depth of this patent is crucial for evaluating long-term supply chain stability and cost efficiency.
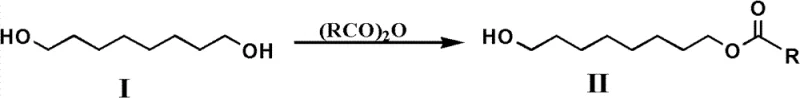
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 10-HDA has been plagued by significant economic and environmental hurdles that hinder scalable manufacturing. Prior art methods often relied on expensive and non-recyclable oxidants such as silver carbonate (Ag2CO3), which, while effective, imposed prohibitive raw material costs unsuitable for bulk production. Alternative routes utilizing pyridinium chlorochromate (PCC) introduced severe toxicity concerns due to hexavalent chromium contamination, necessitating complex and costly waste treatment protocols to meet modern environmental regulations. Furthermore, many existing synthetic pathways involve excessive step counts, leading to cumulative yield losses and increased operational complexity. The use of unstable Grignard reagents in some legacy processes further compounded safety risks, requiring stringent anhydrous conditions and specialized handling equipment that drive up capital expenditure.
The Novel Approach
The methodology disclosed in CN102267893B represents a paradigm shift by streamlining the synthetic route into four highly efficient steps that prioritize atom economy and catalyst recyclability. The core innovation lies in the protection-oxidation strategy where 1,8-octanediol is first selectively mono-acylated, preventing over-oxidation and ensuring high regioselectivity for the subsequent aldehyde formation. Crucially, the oxidation step employs a heterogeneous SiO2-Tempo catalyst system, which allows the expensive nitroxyl radical to be recovered via simple filtration and reused multiple times without significant loss of activity. Additionally, the carbon chain extension is achieved through a Wittig-Horner reaction conducted in an aqueous medium facilitated by ionic liquids, thereby eliminating the need for hazardous strong bases like sodium hydride and volatile organic solvents. This holistic approach not only enhances the overall process yield but also aligns perfectly with green chemistry principles required for sustainable cost reduction in pharmaceutical intermediates manufacturing.

Mechanistic Insights into SiO2-Tempo Catalyzed Oxidation and Ionic Liquid Mediated Coupling
The heart of this synthetic advancement is the sophisticated application of heterogeneous catalysis in the oxidation of the protected alcohol intermediate. The SiO2-Tempo catalyst functions by immobilizing the 2,2,6,6-tetramethylpiperidine-1-oxyl radical onto a silica support, creating a robust surface for the oxidation cycle. In the presence of a co-catalyst like sodium bromide and a terminal oxidant such as sodium hypochlorite, the nitroxyl radical cycles between its oxoammonium and hydroxylamine forms to selectively convert the primary alcohol to an aldehyde. This surface-mediated mechanism prevents the leaching of active species into the solution, ensuring that the catalyst remains intact for recovery. The high selectivity observed in this step is attributed to the steric environment provided by the silica support, which minimizes side reactions such as over-oxidation to carboxylic acids or chlorination of the alkyl chain.
Furthermore, the subsequent Wittig-Horner olefination demonstrates remarkable innovation by utilizing task-specific ionic liquids as phase-transfer catalysts in water. Traditionally, this reaction requires strictly anhydrous conditions and strong bases, but the ionic liquids (such as N-methylpyridinium bromide or imidazolium salts) stabilize the phosphonate carbanion and facilitate its interaction with the aldehyde in the aqueous phase. This unique solvation environment accelerates the reaction kinetics at room temperature, significantly reducing energy consumption compared to thermal reflux methods. The ionic liquid also acts as a surfactant, improving the interfacial contact between the organic substrate and the inorganic base, potassium carbonate. This dual functionality ensures high conversion rates and simplifies downstream processing, as the product can be extracted directly without complex quenching procedures typically associated with reactive organometallic reagents.
How to Synthesize Royal Jelly Acid Efficiently
The practical implementation of this synthesis route involves a carefully controlled sequence of reactions designed to maximize throughput while maintaining rigorous quality standards. The process begins with the precise stoichiometric control of the acylation step to ensure mono-substitution, followed by the critical oxidation where temperature and pH must be tightly regulated to preserve catalyst integrity. The aqueous coupling step offers significant operational simplicity, allowing for the direct use of crude intermediates without purification, which is a major advantage for industrial scale-up. For R&D teams looking to adopt this technology, the detailed standardized synthesis steps are outlined below to ensure reproducibility and safety.
- Perform selective monoacylation of 1,8-octanediol with acid anhydride using pyridine as a base to form 8-alkanoyloxyoctanol.
- Oxidize the mono-ester intermediate to 8-alkanoyloxyoctanal using recyclable SiO2-supported Tempo catalyst and hypochlorite.
- Conduct a Wittig-Horner reaction in water using ionic liquid catalysts to extend the carbon chain, followed by hydrolysis to yield Royal Jelly Acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers substantial benefits that directly impact the bottom line and supply chain resilience. By replacing precious metal oxidants and toxic chromium reagents with recyclable heterogeneous catalysts and benign hypochlorite, the process dramatically lowers the cost of goods sold (COGS) associated with raw material consumption. The ability to recover and reuse the SiO2-Tempo catalyst eliminates the recurring expense of purchasing fresh nitroxyl radicals, providing a sustainable economic model that is less susceptible to market volatility in precious metal prices. Moreover, the shift to aqueous reaction media for the coupling step reduces the dependency on large volumes of expensive anhydrous organic solvents, thereby cutting both material costs and hazardous waste disposal fees.
- Cost Reduction in Manufacturing: The elimination of stoichiometric heavy metal oxidants and the implementation of recyclable catalyst systems fundamentally alter the cost structure of 10-HDA production. By avoiding the use of silver salts and chromium reagents, manufacturers can bypass the high costs associated with these specialty chemicals and the rigorous remediation required for heavy metal waste. The use of common industrial reagents like acetic anhydride and sodium hypochlorite further stabilizes input costs, ensuring predictable budgeting for long-term contracts. Additionally, the high selectivity of the reactions minimizes the formation of difficult-to-remove impurities, reducing the load on purification units and increasing the overall yield of saleable product.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 1,8-octanediol and basic inorganic salts ensures a robust supply chain that is not vulnerable to the shortages often seen with exotic reagents. The simplified operational conditions, including room temperature reactions and ambient pressure, reduce the risk of unplanned downtime caused by equipment failure or safety incidents. This reliability allows suppliers to maintain consistent inventory levels and meet tight delivery schedules, which is critical for downstream pharmaceutical clients who operate on just-in-time manufacturing models. The stability of the ionic liquid catalysts also means that supply disruptions due to catalyst degradation are virtually non-existent.
- Scalability and Environmental Compliance: The green chemistry attributes of this process facilitate easier regulatory approval and permit acquisition for new production facilities. By generating significantly less hazardous waste and avoiding the discharge of heavy metals, the process aligns with increasingly stringent global environmental regulations, reducing the risk of fines or shutdowns. The inherent safety of using water as a solvent and avoiding pyrophoric reagents like sodium hydride lowers insurance premiums and improves the overall safety profile of the plant. This scalability ensures that production can be ramped up from pilot scale to multi-ton commercial quantities without encountering the engineering bottlenecks typical of more complex synthetic routes.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and quality implications for potential partners and technical stakeholders. Understanding these details is essential for making informed decisions about adopting this advanced manufacturing route.
Q: What are the key advantages of the SiO2-Tempo catalyst in this synthesis?
A: The SiO2-Tempo catalyst allows for heterogeneous oxidation, enabling easy filtration and recycling of the expensive nitroxyl radical catalyst, which significantly reduces material costs compared to homogeneous systems.
Q: How does this method improve environmental compliance?
A: By utilizing water as a solvent for the Wittig-Horner step and avoiding toxic heavy metal oxidants like PCC or silver carbonate, the process drastically reduces hazardous waste generation and simplifies effluent treatment.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the route uses stable starting materials like 1,8-octanediol, operates under mild conditions (0-5°C for acylation, room temperature for coupling), and avoids dangerous reagents like sodium hydride, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Royal Jelly Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to deliver superior value to our global clientele. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative strategies outlined in CN102267893B can be seamlessly translated into industrial reality. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Royal Jelly Acid meets the highest international standards for pharmaceutical and cosmetic applications. Our infrastructure is designed to handle complex catalytic systems and green chemistry protocols efficiently, positioning us as a leader in sustainable intermediate manufacturing.
We invite you to collaborate with us to leverage these technological advancements for your specific product requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and product quality. Let us partner together to drive innovation and value in the fine chemical industry.
