Revolutionizing Carbapenem Production: A Rhodium-Free Route for Meropenem Intermediates
The pharmaceutical landscape for beta-lactam antibiotics is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable manufacturing processes. Patent CN101891746B introduces a groundbreaking preparation method for Meropenem, a critical broad-spectrum carbapenem antibiotic. This technology addresses long-standing industry pain points by eliminating the reliance on noble metal rhodium catalysts, which have historically plagued production costs and supply chain stability. The invention outlines a streamlined four-step synthetic route starting from a specific thioether intermediate, leveraging organophosphorus chemistry to construct the crucial carbapenem nucleus. By shifting away from precious metal catalysis to more abundant reagents like triethyl phosphite and phenol derivatives, this method offers a robust alternative for large-scale manufacturing. The strategic design of this pathway not only enhances atom economy but also simplifies downstream processing, making it an attractive option for generic drug manufacturers seeking to optimize their production lines. 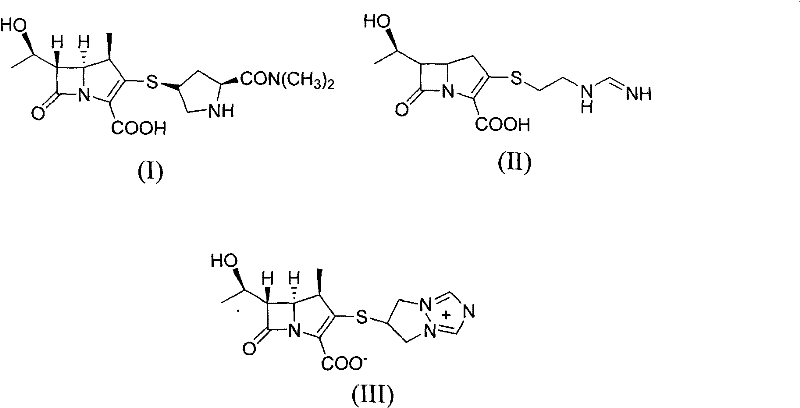
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of beta-methyl carbapenem antibiotics has been dominated by two primary methodologies, notably the Sumitomo Pharmaceuticals route and the Lederle Ltd. route. The Sumitomo approach, often referred to as Reaction Scheme A, utilizes a long synthetic sequence that requires multiple isolation steps for chiral intermediates. This complexity inherently violates the principles of atom economy, generating significant waste and requiring extensive solvent usage. Furthermore, a critical bottleneck in this traditional pathway is the dependence on a noble metal rhodium catalyst for the formation of the key bicyclic intermediate. As global demand for rhodium fluctuates and prices soar due to scarcity, the economic viability of this route diminishes, creating substantial financial risk for manufacturers. Similarly, the Lederle route, while slightly shorter, suffers from the acquisition difficulties of its specific starting materials and shares the same fundamental defects regarding catalyst cost and environmental impact. 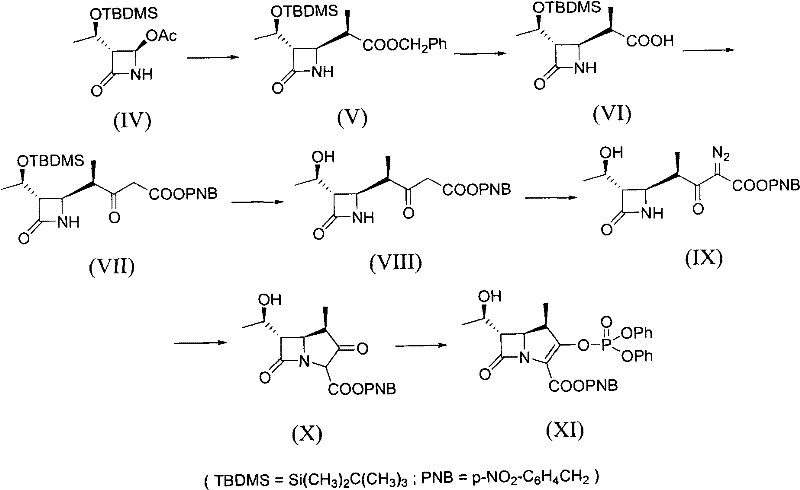
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent presents a concise and highly efficient strategy that bypasses the need for rhodium entirely. The core innovation lies in a four-step transformation that converts a readily available thioether precursor directly into the Meropenem skeleton. This route capitalizes on the reactivity of organophosphorus reagents to generate ylides, which subsequently undergo cyclization under the influence of phenolic catalysts. This mechanistic shift allows for the construction of the double bond within the five-membered ring without the steric and electronic constraints imposed by metal-catalyzed reactions. The result is a process that is not only chemically elegant but also operationally superior, requiring fewer unit operations and less energy. By reducing the step count and eliminating the most expensive catalyst from the equation, this method fundamentally alters the cost structure of Meropenem production, offering a clear path toward margin improvement for commercial entities. 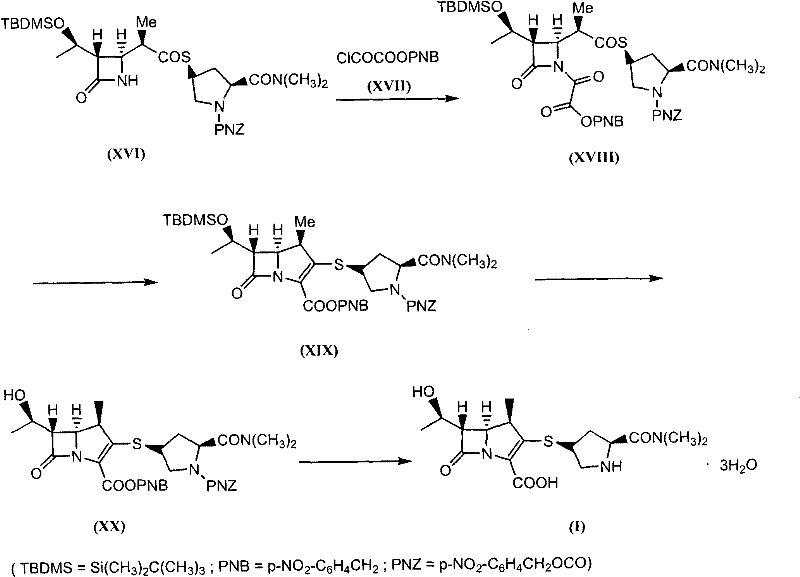
Mechanistic Insights into Organophosphorus-Mediated Cyclization
The heart of this technological advancement is the sophisticated use of organophosphorus chemistry to drive the formation of the carbapenem double bond. In the second step of the synthesis, the intermediate compound (XVIII), which contains an activated oxalyl group, reacts with a phosphorus reagent such as triethyl phosphite. This interaction generates a reactive phosphorus ylide species in situ. Unlike traditional Wittig reactions that might require harsh conditions, this specific variation proceeds under relatively mild thermal conditions in aromatic solvents like xylene. The presence of a phenol or hydroquinone catalyst is crucial here; it facilitates the intramolecular cyclization by stabilizing the transition state and promoting the elimination of the phosphorus oxide byproduct. This mechanism ensures high stereoselectivity, preserving the critical (5R, 6S) configuration required for biological activity. The ability to achieve this cyclization without metal coordination complexes demonstrates a deep understanding of physical organic chemistry, allowing for a cleaner reaction profile with fewer metal-based impurities.
Furthermore, the process incorporates robust impurity control mechanisms through strategic purification steps. While many intermediates in this route are used directly in subsequent steps (telescoped synthesis) to save time, the key cyclized intermediate (XIX) undergoes recrystallization from ethyl acetate. This step is vital for removing phosphorus-containing byproducts and unreacted starting materials, ensuring that the purity and content of the intermediate reach levels exceeding 97%. The final step involves a catalytic hydrogenation using palladium on carbon, which serves a dual purpose: it removes the p-nitrobenzyl protecting groups and reduces the azide or other reducible functionalities if present, finalizing the active pharmaceutical ingredient. The choice of palladium over rhodium for this reduction is economically significant, as palladium is generally more accessible and easier to recover, further enhancing the sustainability profile of the overall manufacturing process.
How to Synthesize Meropenem Efficiently
The synthesis of Meropenem via this patented route is designed for operational simplicity and high throughput, making it ideal for industrial scale-up. The process begins with the activation of the side-chain amine on the thioether precursor using chlorooxalic acid p-nitrobenzyl ester, followed by the critical organophosphorus cyclization described previously. Subsequent hydrolysis removes the silyl protecting group to reveal the hydroxyethyl side chain, and the final hydrogenation step delivers the target molecule. This sequence minimizes the handling of unstable intermediates and reduces the total number of isolation events, which is a common source of yield loss in pharmaceutical manufacturing. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to execute this synthesis safely and effectively, please refer to the standardized guide below.
- React compound (XVI) with chlorooxalic acid p-nitrobenzyl ester (XVII) in an organic solvent with an organic base to form compound (XVIII).
- Treat compound (XVIII) with an organophosphorus reagent to generate a ylide, followed by cyclization using a phenol or hydroquinone catalyst to yield compound (XIX).
- Hydrolyze compound (XIX) using a fluoride source or acid mixture in an organic solvent to remove the silyl protecting group, obtaining compound (XX).
- Perform catalytic hydrogenation on compound (XX) using palladium on carbon and hydrogen gas to reduce the nitro groups and finalize the Meropenem structure (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-free synthesis route represents a strategic opportunity to de-risk the supply of critical antibiotic intermediates. The traditional reliance on rhodium creates a vulnerability to geopolitical supply shocks and volatile commodity pricing, which can erode profit margins unpredictably. By transitioning to a process that utilizes commodity chemicals like triethyl phosphite and phenol, manufacturers can stabilize their raw material costs and secure long-term supply contracts with greater confidence. Additionally, the shortened synthetic route directly translates to reduced manufacturing cycle times, allowing for faster response to market demand fluctuations and improved inventory turnover rates. The simplified purification protocols also mean lower solvent consumption and reduced waste disposal costs, contributing to a leaner and more agile production model.
- Cost Reduction in Manufacturing: The elimination of the noble metal rhodium catalyst is the primary driver for cost optimization in this process. Rhodium is one of the most expensive precious metals, and its removal from the catalytic cycle significantly lowers the direct material cost per kilogram of product. Furthermore, the high yields achieved in the cyclization and hydrogenation steps minimize the loss of valuable chiral intermediates, ensuring that the maximum amount of input material is converted into saleable product. The ability to recycle solvents like toluene and xylene after simple recovery further compounds these savings, creating a highly efficient economic model for large-scale production.
- Enhanced Supply Chain Reliability: The raw materials required for this novel route, such as organophosphorus reagents and basic organic solvents, are widely available from multiple global suppliers, unlike specialized rhodium catalysts which may have limited sources. This diversification of the supply base reduces the risk of production stoppages due to single-source failures. Moreover, the robustness of the reaction conditions, which tolerate a range of temperatures and pressures without compromising quality, ensures consistent output even when facing minor variations in utility supplies or operational parameters, thereby guaranteeing reliable delivery schedules to downstream customers.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers distinct advantages for scaling up to commercial volumes. The reduction in step count and the avoidance of heavy metal catalysts simplify the waste stream treatment, making it easier to meet stringent environmental discharge standards. The process generates less hazardous waste compared to traditional routes, reducing the burden on effluent treatment plants. Additionally, the telescoping of certain steps reduces the need for intermediate storage and handling, which lowers the overall footprint of the manufacturing facility and aligns with modern green chemistry principles, facilitating smoother regulatory approvals in key markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-free Meropenem synthesis. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of yield, purity, and operational feasibility. Understanding these nuances is essential for technical teams evaluating the transfer of this process to their own production facilities.
Q: Why is the rhodium-free synthesis of Meropenem significant for cost reduction?
A: Traditional methods rely on expensive noble metal rhodium catalysts, which are subject to price volatility and supply shortages. This novel route eliminates the need for rhodium entirely, utilizing readily available organophosphorus reagents and phenol catalysts instead, drastically lowering raw material costs and dependency on scarce metals.
Q: How does this new method improve upon the Sumitomo and Lederle synthetic routes?
A: Conventional routes like Sumitomo's Scheme A involve lengthy multi-step processes with difficult chiral separations and poor atom economy. This invention shortens the synthesis to just four key steps from the key intermediate, simplifies purification through recrystallization rather than complex chromatography, and avoids the use of hazardous or expensive reagents found in older methodologies.
Q: What are the purity specifications achievable with this organophosphorus cyclization method?
A: The process is designed for high efficiency, with the key cyclization step yielding intermediates with purity and content reaching over 97% after simple recrystallization. The final hydrogenation step further ensures the production of high-purity Meropenem trihydrate suitable for pharmaceutical applications without requiring extensive downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meropenem Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively analyzed the potential of this rhodium-free route and possesses the technical capability to implement it seamlessly. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is smooth and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of carbapenem intermediate we produce adheres to the highest international standards for safety and efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective technology for their Meropenem supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, high-quality supply of Meropenem intermediates that drives value and stability for your organization.
