Advanced Direct Sulfonation Technology for High-Purity 2-Phenylbenzimidazole-5-Sulfonic Acid Commercialization
Advanced Direct Sulfonation Technology for High-Purity 2-Phenylbenzimidazole-5-Sulfonic Acid Commercialization
The landscape of ultraviolet absorbent manufacturing is undergoing a significant transformation driven by the need for greener processes and higher purity standards, as exemplified by the breakthrough methodology detailed in patent CN110981814A. This pivotal intellectual property introduces a novel preparation method for 2-phenylbenzimidazole-5-sulfonic acid, a critical water-soluble UV filter extensively used in cosmetics and water-based coatings. Unlike legacy technologies that rely on harsh one-pot syntheses at extreme temperatures or multi-step sequences prone to oxidation, this invention leverages a controlled direct sulfonation strategy. By shifting the synthetic entry point to 2-phenylbenzimidazole and employing a composite sulfonating system within a halogenated hydrocarbon medium, the process achieves exceptional control over reaction thermodynamics. This technical leap not only ensures the production of a pure white final product meeting stringent cosmetic specifications but also fundamentally alters the economic and environmental footprint of cosmetic additive manufacturing by minimizing waste acid generation and eliminating the need for heavy metal decolorization agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
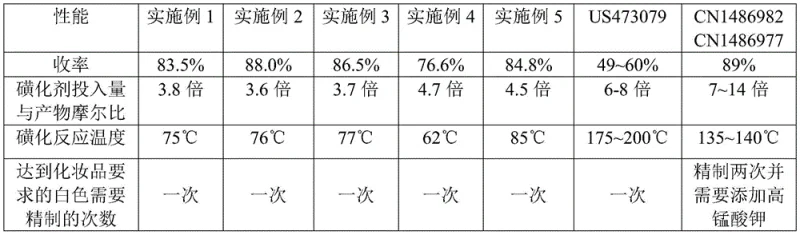
Historically, the industrial synthesis of 2-phenylbenzimidazole-5-sulfonic acid has been plagued by severe process inefficiencies and quality control challenges that hinder commercial scale-up of complex UV filters. For instance, the method disclosed in US5473079 relies on a one-pot condensation of o-phenylenediamine and benzoic acid in excessive sulfuric acid, necessitating heating to a staggering 178°C. Such extreme thermal conditions not only pose significant safety risks and energy costs but also result in dismal molar yields ranging merely from 49% to 60%. Furthermore, alternative routes like those in CN1486982 attempt to improve yields by using 3,4-diaminobenzenesulfonic acid; however, the precursor synthesis involves reacting o-phenylenediamine with fuming sulfuric acid at over 100°C, inevitably leading to oxidation side reactions. This oxidation causes the intermediate to turn dark gray or black, transmitting color defects to the final product and necessitating aggressive decolorization with potassium permanganate, which introduces toxic heavy metal residues into the wastewater and the final API, creating a major liability for cosmetic formulators.
The Novel Approach
In stark contrast, the innovative protocol defined in CN110981814A circumvents these historical bottlenecks by adopting a direct sulfonation pathway on the pre-formed benzimidazole ring. This strategic shift eliminates the oxidative degradation associated with aniline derivatives, ensuring that the reaction mixture remains stable and the product color remains inherently light. The core of this novelty lies in the use of a composite sulfonating agent—typically a precise blend of concentrated sulfuric acid and chlorosulfonic acid or fuming sulfuric acid—dispersed in a halogenated hydrocarbon solvent such as dichloroethane. This biphasic or semi-heterogeneous system allows for excellent heat dissipation, maintaining the reaction temperature within a mild window of 60°C to 100°C, thereby suppressing the formation of multi-sulfonated byproducts. Consequently, this approach delivers a robust molar yield between 76.6% and 88.0% while producing a crude solid that requires only a single refinement step to achieve pharmaceutical-grade whiteness, effectively solving the dual problems of low efficiency and heavy metal contamination.
Mechanistic Insights into Composite Sulfonation in Halogenated Media
The chemical elegance of this process stems from the synergistic interaction between the composite sulfonating agent and the organic solvent matrix, which creates a highly selective environment for electrophilic aromatic substitution. When concentrated sulfuric acid is combined with stronger sulfonating agents like chlorosulfonic acid, the resulting mixture generates a potent concentration of sulfur trioxide (SO3) species without the excessive viscosity and charring associated with using oleum alone. The presence of the halogenated hydrocarbon solvent, specifically dichloroethane or dichloropropane, plays a dual mechanistic role: firstly, it acts as a heat sink to moderate the exothermic nature of the sulfonation, preventing local hot spots that could degrade the sensitive benzimidazole nucleus; secondly, it facilitates a unique phase behavior where the sulfonated product, being highly polar and acidic, tends to separate or precipitate upon quenching, while unreacted starting materials and neutral byproducts remain soluble or are easily washed away. This differential solubility is crucial for high-purity 2-phenylbenzimidazole-5-sulfonic acid isolation, as it minimizes the entrapment of tarry impurities that typically necessitate extensive chromatographic or recrystallization purifications in other methods.
Furthermore, the mechanism of impurity control is intrinsically linked to the avoidance of oxidative conditions. In traditional routes involving diamines, the electron-rich amino groups are susceptible to oxidation by air or acidic oxidants, forming quinoid structures that act as chromophores. By starting with the cyclized 2-phenylbenzimidazole, the nitrogen atoms are tied up in the aromatic heterocycle, significantly reducing their nucleophilicity and susceptibility to oxidation. The subsequent sulfonation occurs selectively at the 5-position due to the electronic directing effects of the existing substituents, and the mild temperature range (preferably 70-90°C) ensures that the kinetic energy of the system is insufficient to drive secondary sulfonation at the 4 or 6 positions. This high regioselectivity means that the crude product is already of high purity (>99% HPLC), simplifying the downstream processing to a mere pH adjustment and activated carbon treatment, rather than complex chemical transformations to remove isomers.
How to Synthesize 2-Phenylbenzimidazole-5-Sulfonic Acid Efficiently
Implementing this advanced synthesis route requires precise adherence to the stoichiometric ratios and thermal profiles outlined in the patent to maximize the benefits of solvent recovery and yield optimization. The process is designed to be operationally simple yet chemically rigorous, making it ideal for reliable UV absorbent supplier operations aiming for consistent batch-to-batch quality. The following overview summarizes the critical operational phases, emphasizing the importance of the composite acid preparation and the phase separation technique which are key to the process economics. For a visual representation of the material flow and unit operations involved in this streamlined manufacturing sequence, please refer to the process diagram below which illustrates the transition from raw materials to the refined white solid.
- Sulfonation Reaction: Dissolve 2-phenylbenzimidazole in a halogenated hydrocarbon solvent (e.g., dichloroethane) and react with a composite sulfonating agent (concentrated sulfuric acid mixed with chlorosulfonic acid) at 60-100°C.
- Phase Separation and Quenching: Allow the reaction mixture to stand for layering. Recover the upper organic solvent layer for reuse. Quench the lower acidic layer with water to precipitate the crude sulfonic acid solid.
- Purification: Dissolve the crude solid in water, adjust pH to 8-10 with sodium hydroxide, decolorize with activated carbon, filter, and re-acidify to pH 2.5-3.5 to precipitate the pure white refined product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of the technology described in CN110981814A offers profound advantages that extend far beyond simple yield improvements, directly addressing the core mandates of cost reduction in cosmetic additive manufacturing and supply chain resilience. The most immediate impact is observed in the raw material and utility consumption profiles; by replacing the massive excesses of sulfuric acid required in legacy high-temperature processes with a catalytic-like amount of composite agent in a recoverable solvent, the volume of hazardous waste acid generated is drastically reduced. This reduction translates directly into lower disposal costs and reduced regulatory burden, as the facility no longer needs to manage the complex neutralization of tons of spent acid per ton of product. Additionally, the elimination of potassium permanganate and other heavy metal oxidants removes a significant cost center associated with wastewater treatment and heavy metal testing, ensuring that the final product is compliant with the strictest global cosmetic regulations without expensive post-processing.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the efficient recycling of the halogenated hydrocarbon solvent. Since the solvent forms a distinct upper layer after the reaction, it can be decanted and reused directly for the subsequent batch with minimal loss, or simply distilled to remove trace impurities. This closed-loop solvent management significantly lowers the variable cost of goods sold (COGS) compared to aqueous or single-use solvent systems. Furthermore, the ability to obtain a pure white product after only one refinement step reduces the consumption of decolorizing agents, filtration media, and energy-intensive drying cycles, leading to substantial operational expenditure savings without compromising on the quality metrics required by top-tier personal care brands.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses that rely on unstable intermediates. This direct sulfonation route shortens the critical path of production by consolidating the synthesis into fewer unit operations, thereby reducing lead time for high-purity cosmetic intermediates. The robustness of the reaction conditions (60-100°C) means that the process is less susceptible to thermal runaways or equipment failures that can halt production lines. Moreover, the starting material, 2-phenylbenzimidazole, is a stable and commercially available commodity, reducing the risk of supply bottlenecks associated with specialized sulfonated diamines. This stability ensures that manufacturers can maintain consistent inventory levels and meet sudden spikes in demand from the sunscreen and skincare markets with greater agility.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies heat transfer and mixing challenges, but the moderate temperature range and the use of a liquid organic medium in this method make it inherently scalable from pilot plants to multi-ton reactors. The process generates significantly less waste acid and avoids the introduction of persistent heavy metals into the effluent stream, aligning perfectly with modern ESG (Environmental, Social, and Governance) goals. This environmental compatibility simplifies the permitting process for capacity expansion and reduces the risk of regulatory shutdowns due to non-compliance. For global supply chain heads, this means securing a long-term source of UV absorbers that is not only cost-effective but also sustainable, future-proofing the supply base against tightening environmental legislation in key manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel sulfonation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms established industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of switching to this more efficient manufacturing route.
Q: How does the new direct sulfonation method improve product color compared to traditional routes?
A: Traditional methods starting with o-phenylenediamine often result in oxidation and dark-colored intermediates requiring harsh decolorization with potassium permanganate. The new method (CN110981814A) starts with 2-phenylbenzimidazole, fundamentally avoiding aniline oxidation, allowing a pure white product to be obtained after just one refinement without heavy metal oxidants.
Q: What are the yield advantages of using a composite sulfonating agent?
A: By utilizing a composite agent of concentrated sulfuric acid and chlorosulfonic acid (or fuming sulfuric acid) in a halogenated solvent, the reaction temperature is controlled between 60-100°C. This prevents excessive multi-sulfonation and degradation, achieving molar yields of 76.6% to 88.0%, significantly higher than the 49-60% yields of older high-temperature processes.
Q: Is the organic solvent used in this process recoverable for industrial scale-up?
A: Yes, the process utilizes halogenated hydrocarbons like dichloroethane which form a distinct upper layer after the reaction. This solvent layer can be directly separated and reused for the next batch without distillation, or distilled to remove enriched impurities, drastically reducing solvent consumption and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenylbenzimidazole-5-Sulfonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing protocols like the one described in CN110981814A requires a partner with deep technical expertise and robust infrastructure. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this direct sulfonation process are fully realized in large-scale operations. Our facilities are equipped with corrosion-resistant reactors capable of handling composite sulfonating agents safely, and our stringent purity specifications are enforced through rigorous QC labs that utilize HPLC and ICP-MS to guarantee the absence of heavy metals and isomeric impurities. We are committed to delivering a product that not only meets but exceeds the performance expectations of the global personal care industry.
We invite procurement leaders and R&D directors to engage with us to explore how this technology can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential reductions in waste disposal and raw material costs specific to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, allowing you to make informed decisions that drive both profitability and product excellence in your UV protection formulations.
