Advanced Two-Step Synthesis of Chiral Sulfoxide Styrene Monomers for Scalable Polymer Catalyst Production
Advanced Two-Step Synthesis of Chiral Sulfoxide Styrene Monomers for Scalable Polymer Catalyst Production
The landscape of asymmetric catalysis is undergoing a significant transformation, driven by the urgent industrial need for sustainable, recoverable, and highly efficient chiral ligands. Patent CN112679394A introduces a groundbreaking preparation method for a styrene monomer containing a chiral sulfoxide group, a critical building block for next-generation polymer-supported catalysts. This innovation addresses the longstanding limitations of traditional neutral coordination organic catalysts, which often require stoichiometric quantities and cannot be easily recovered, thereby imposing a substantial environmental burden and operational cost on fine chemical manufacturing. By leveraging a concise two-step synthetic route starting from bis-ketal glucose and methyl sulfinyl chloride, this technology enables the production of monomers that can be copolymerized into robust polymer backbones. These resulting polymers serve as reusable chiral sulfoxide ligands, capable of being loaded into microchannels for continuous flow chemistry, representing a paradigm shift towards greener and more economical pharmaceutical and agrochemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral sulfoxides for use in asymmetric catalysis has been plagued by significant inefficiencies and environmental drawbacks. Conventional schemes typically rely on the oxidation of sulfides using strong oxidants, a process that is notoriously difficult to control regarding stereoselectivity and often results in over-oxidation to sulfones or low yields of the desired sulfoxide. Furthermore, in many established asymmetric catalytic processes, the chiral sulfoxide catalyst is employed in chemical doses, ranging from 1 to 3 equivalents, rather than in catalytic amounts. This stoichiometric usage not only drives up the raw material costs drastically but also generates substantial chemical waste, as the catalyst cannot be easily separated from the reaction mixture for recovery and reuse. The inability to recycle these valuable chiral ligands creates a bottleneck in large-scale manufacturing, where the accumulation of waste and the high cost of chiral pool materials render many potential synthetic routes commercially unviable for cost-sensitive applications.
The Novel Approach
In stark contrast to these archaic methods, the novel approach detailed in the patent utilizes a highly controlled, low-temperature substitution strategy that bypasses the need for harsh oxidants entirely. The process begins with the reaction of bis-ketal glucose and methyl sulfinyl chloride in an organic solvent at a cryogenic temperature of -78 ℃, ensuring precise stereochemical control during the formation of the chiral center. This intermediate is then coupled with a (4-vinylphenyl) magnesium chloride Grignard reagent at mild temperatures between 0 ℃ and 5 ℃, efficiently installing the polymerizable styrene moiety. 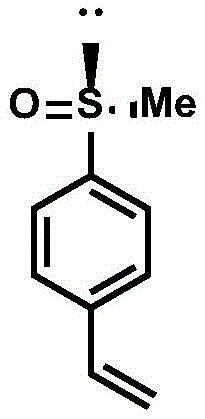 This streamlined two-step sequence not only simplifies the operational complexity but also achieves exceptional enantiomeric excess values, reaching up to 98% ee, which is critical for high-performance catalytic applications. By creating a monomer that can be polymerized, this method transforms the chiral sulfoxide from a disposable reagent into a durable, recoverable asset, fundamentally altering the economics of asymmetric synthesis.
This streamlined two-step sequence not only simplifies the operational complexity but also achieves exceptional enantiomeric excess values, reaching up to 98% ee, which is critical for high-performance catalytic applications. By creating a monomer that can be polymerized, this method transforms the chiral sulfoxide from a disposable reagent into a durable, recoverable asset, fundamentally altering the economics of asymmetric synthesis.
Mechanistic Insights into Grignard-Mediated Sulfoxide Functionalization
The core of this technological advancement lies in the meticulous orchestration of nucleophilic substitution and organometallic coupling under strictly anhydrous and cryogenic conditions. The initial step involves the activation of the sulfur center in methyl sulfinyl chloride, which acts as an electrophile towards the hydroxyl groups of the bis-ketal glucose scaffold. The use of solvents such as tetrahydrofuran mixed with pyridine, or toluene with diisopropylethylamine, plays a dual role: acting as the reaction medium and as a base to scavenge the hydrochloric acid byproduct, thereby driving the equilibrium towards the formation of the chiral sulfoxide-substituted glucose intermediate. The stereochemical integrity is preserved through the rigid cyclic structure of the glucose derivative, which directs the approach of the incoming sulfinyl group, resulting in either the R or S configuration depending on the specific solvent system and reaction conditions employed.
Following the isolation of the chiral intermediate, the second mechanistic phase involves a Grignard exchange reaction that is pivotal for introducing the vinyl functionality required for polymerization. The reaction of the chiral sulfoxide-substituted bis-ketal glucose with (4-vinylphenyl) magnesium chloride proceeds via a nucleophilic attack on the sulfur atom or a displacement mechanism that retains the chiral information while attaching the aromatic vinyl group. 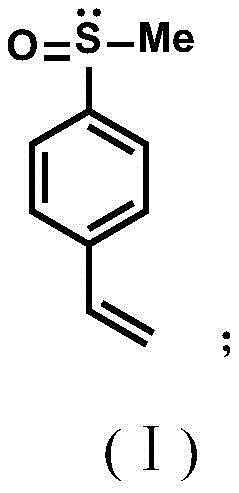 The resulting 1-(methylsulfinyl)-4-vinylbenzene monomer possesses a robust chiral environment that, once copolymerized, creates a heterogeneous catalytic surface. This structural design ensures that the active chiral sites are accessible to substrates in solution while remaining anchored to the solid support, facilitating easy filtration and recycling. The high ee value of up to 98% observed in the final product confirms that the harsh conditions of the Grignard reaction do not racemize the sensitive sulfoxide center, a testament to the stability and robustness of this specific synthetic pathway.
The resulting 1-(methylsulfinyl)-4-vinylbenzene monomer possesses a robust chiral environment that, once copolymerized, creates a heterogeneous catalytic surface. This structural design ensures that the active chiral sites are accessible to substrates in solution while remaining anchored to the solid support, facilitating easy filtration and recycling. The high ee value of up to 98% observed in the final product confirms that the harsh conditions of the Grignard reaction do not racemize the sensitive sulfoxide center, a testament to the stability and robustness of this specific synthetic pathway.
How to Synthesize 1-(methylsulfinyl)-4-vinylbenzene Efficiently
The synthesis of this high-value chiral monomer is designed for reproducibility and scalability, utilizing standard laboratory equipment that can be readily adapted for pilot and commercial plant operations. The process relies on widely available commodity chemicals, eliminating the supply chain risks associated with exotic or proprietary reagents. Operators must maintain strict temperature control, particularly during the initial sulfinylation step at -78 ℃, to maximize stereoselectivity. The subsequent workup involves standard aqueous quenching and extraction protocols, making the purification process straightforward and amenable to large-scale batch processing. For a comprehensive, step-by-step technical guide including specific molar ratios, solvent volumes, and safety precautions, please refer to the standardized synthesis protocol provided below.
- React bis-ketal glucose with methyl sulfinyl chloride in an organic solvent at -78 ℃ for 1-3 hours to form the chiral sulfoxide intermediate.
- Quench the first reaction, extract, and purify the bis-ketal glucose containing chiral sulfoxide substitution.
- React the purified intermediate with (4-vinylphenyl) magnesium chloride Grignard reagent in toluene at 0-5 ℃ to yield the final styrene monomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic advantages that extend far beyond simple technical feasibility. The shift from stoichiometric, single-use chiral catalysts to polymer-supported systems derived from this monomer represents a fundamental optimization of the cost structure in fine chemical manufacturing. By enabling the recovery and reuse of the chiral ligand, facilities can drastically reduce their consumption of expensive chiral starting materials, leading to significant long-term savings on raw material expenditures. Furthermore, the simplified workflow, which avoids the use of hazardous strong oxidants and complex purification steps often required for traditional sulfoxide synthesis, translates directly into reduced operational overheads and lower waste disposal costs, enhancing the overall economic viability of producing high-purity chiral intermediates.
- Cost Reduction in Manufacturing: The elimination of strong oxidants and the ability to recover the catalyst through polymerization support leads to a drastic simplification of the production process. This reduction in reagent complexity and waste generation inherently lowers the cost of goods sold (COGS) without compromising on the quality or purity of the final chiral products. The use of inexpensive, bulk-available starting materials like bis-ketal glucose further insulates the supply chain from price volatility associated with specialized chiral reagents.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly strengthened by the reliance on commoditized raw materials such as glucose derivatives and common Grignard reagents, which are readily available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks and ensures consistent production continuity even during periods of market fluctuation. Additionally, the robust nature of the synthetic route allows for flexible manufacturing schedules, enabling rapid response to changing demand patterns from downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-ton commercial production without the need for specialized high-pressure or high-temperature equipment. The avoidance of toxic heavy metal catalysts and strong oxidizing agents aligns perfectly with increasingly stringent global environmental regulations, reducing the regulatory burden and facilitating smoother permitting processes for new manufacturing lines. This green chemistry profile not only future-proofs the operation against tightening emissions standards but also enhances the brand reputation of the end-product in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral sulfoxide monomer technology. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing a clear understanding of the operational parameters and potential applications. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating polymer-supported catalysts into their existing synthetic workflows and for procurement teams assessing the long-term value proposition of this innovative approach.
Q: What is the enantiomeric excess (ee) achievable with this synthesis method?
A: According to patent CN112679394A, the described method can achieve an enantiomeric excess (ee) value of up to 98% for the resulting chiral sulfoxide monomers, ensuring high optical purity essential for asymmetric catalysis.
Q: Why is this styrene monomer significant for pharmaceutical manufacturing?
A: This monomer allows for the creation of polymer-supported chiral sulfoxide catalysts. Unlike traditional stoichiometric catalysts, these polymer-bound versions can be recovered and reused, significantly reducing waste and cost in drug synthesis.
Q: What are the key starting materials for this process?
A: The synthesis utilizes readily available and cost-effective raw materials, specifically bis-ketal glucose and methyl sulfinyl chloride, followed by coupling with a Grignard reagent, avoiding the need for expensive strong oxidants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Sulfoxide Styrene Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chiral sulfoxide styrene monomer technology in advancing the field of asymmetric catalysis. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, consistently delivering monomers with the high enantiomeric excess required for demanding pharmaceutical applications. We are committed to supporting your innovation with reliable supply and technical expertise that meets the highest international standards.
We invite you to collaborate with us to unlock the full commercial potential of this sustainable catalytic solution. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and process requirements. Please contact our technical procurement team today to request specific COA data, detailed route feasibility assessments, and to discuss how we can accelerate your project timelines with our reliable supply of high-purity chiral monomers.
