Advanced One-Pot Synthesis of Aryl Selenides for Scalable Pharmaceutical Intermediate Production
Introduction to Advanced Aryl Selenide Manufacturing
The landscape of organic synthesis is continually evolving to meet the rigorous demands of the pharmaceutical and fine chemical industries, particularly regarding the efficient production of selenium-containing compounds which possess significant biological and pharmacological activities. Patent CN113200899A introduces a groundbreaking methodology for the synthesis of aryl selenide compounds, utilizing a highly efficient one-pot Friedel-Crafts alkyl selenization reaction that addresses many of the historical challenges associated with organoselenium chemistry. This innovative approach leverages the catalytic power of ferric chloride to facilitate the three-component coupling of styrenes, electron-rich aromatic hydrocarbons, and diselenides under remarkably mild conditions. By enabling the direct construction of complex aryl selenide scaffolds with excellent regioselectivity and high yields, this technology represents a substantial leap forward for manufacturers seeking reliable aryl selenide supplier capabilities. The significance of this development extends beyond mere academic interest, offering tangible solutions for the scalable production of intermediates used in antioxidants, enzyme inhibitors, and potential anti-tumor agents. As the global demand for high-purity OLED material and pharmaceutical intermediates grows, mastering such efficient synthetic routes becomes a critical competitive advantage for forward-thinking chemical enterprises.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl selenide compounds has been plagued by significant operational hurdles that hindered their widespread adoption in large-scale industrial applications. Traditional methodologies often relied on harsh reaction conditions, including the use of strong bases, toxic reagents, or expensive transition metal catalysts that required stringent removal protocols to meet pharmaceutical purity standards. Many existing routes involved multi-step sequences that not only increased the overall processing time but also resulted in cumulative yield losses and generated substantial amounts of hazardous waste. Furthermore, the sensitivity of selenium reagents to oxidation and the difficulty in controlling regioselectivity during electrophilic substitution frequently led to complex product mixtures that were difficult and costly to purify. These inefficiencies created bottlenecks in the supply chain, making cost reduction in electronic chemical manufacturing and pharmaceutical production increasingly difficult to achieve. The reliance on precious metal catalysts in some older methods also introduced supply chain vulnerabilities and elevated raw material costs, rendering the final products less economically viable for commodity-scale applications. Consequently, there has been a persistent industry need for a more robust, economical, and environmentally benign synthetic strategy.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN113200899A offers a streamlined one-pot protocol that dramatically simplifies the manufacturing process while enhancing overall efficiency. This novel approach utilizes readily available ferric chloride as a catalyst to drive the three-component reaction between styrenes, electron-rich aromatics, and diselenides in a single vessel, eliminating the need for intermediate isolation steps. The reaction proceeds smoothly in acetonitrile at a moderate temperature of 80°C, avoiding the extreme thermal conditions that often degrade sensitive functional groups or lead to safety incidents. As demonstrated in the specific example where styrene reacts with 1,3,5-trimethoxybenzene and diphenyl diselenide, this method can achieve impressive yields of up to 88% under optimized conditions. 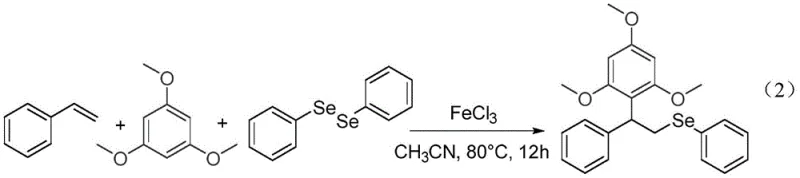 The versatility of this system is further highlighted by its tolerance to a wide range of substituents on the styrene and aromatic rings, allowing for the rapid generation of diverse chemical libraries. By consolidating multiple bond-forming events into a single operation, this process significantly reduces solvent consumption, labor hours, and waste generation, aligning perfectly with modern green chemistry principles. This paradigm shift enables manufacturers to produce high-purity aryl selenides with greater consistency and lower operational overhead.
The versatility of this system is further highlighted by its tolerance to a wide range of substituents on the styrene and aromatic rings, allowing for the rapid generation of diverse chemical libraries. By consolidating multiple bond-forming events into a single operation, this process significantly reduces solvent consumption, labor hours, and waste generation, aligning perfectly with modern green chemistry principles. This paradigm shift enables manufacturers to produce high-purity aryl selenides with greater consistency and lower operational overhead.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the ferric chloride catalyst, which acts as a potent Lewis acid to activate the reaction components. In the proposed mechanism, the FeCl3 coordinates with the diselenide species, enhancing its electrophilicity and promoting the formation of a reactive selenenyl cation or a similar activated complex. This activated selenium species then undergoes a selective addition across the double bond of the styrene derivative, generating a stabilized benzylic carbocation intermediate that is crucial for the subsequent step. The presence of the electron-rich aromatic hydrocarbon allows for a rapid Friedel-Crafts type alkylation, where the aromatic ring attacks the benzylic carbocation to form the final carbon-carbon bond.  This cascade sequence is highly efficient because the catalyst effectively lowers the activation energy for both the selenylation and the alkylation steps, ensuring that the reaction proceeds to completion without the accumulation of unstable intermediates. The choice of acetonitrile as the solvent is also mechanistically significant, as it stabilizes the ionic intermediates and facilitates the solubility of the inorganic catalyst, thereby maintaining a homogeneous reaction environment. Understanding this mechanistic nuance is vital for R&D teams aiming to further optimize the process or adapt it to novel substrates, as it provides a clear roadmap for manipulating reaction parameters to maximize throughput.
This cascade sequence is highly efficient because the catalyst effectively lowers the activation energy for both the selenylation and the alkylation steps, ensuring that the reaction proceeds to completion without the accumulation of unstable intermediates. The choice of acetonitrile as the solvent is also mechanistically significant, as it stabilizes the ionic intermediates and facilitates the solubility of the inorganic catalyst, thereby maintaining a homogeneous reaction environment. Understanding this mechanistic nuance is vital for R&D teams aiming to further optimize the process or adapt it to novel substrates, as it provides a clear roadmap for manipulating reaction parameters to maximize throughput.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions that are common in radical-based or harsh acidic conditions. The mild Lewis acidic nature of FeCl3 prevents the excessive polymerization of styrene derivatives, a frequent issue in traditional cationic polymerization processes that can severely impact yield and product quality. Additionally, the high regioselectivity observed in the Friedel-Crafts step ensures that the aromatic ring attaches at the desired position, reducing the formation of regioisomers that are notoriously difficult to separate via chromatography. The one-pot nature of the reaction also limits the exposure of reactive intermediates to atmospheric oxygen or moisture, which could otherwise lead to oxidation byproducts or hydrolysis. For quality assurance teams, this means a cleaner crude reaction profile that simplifies downstream purification and ensures that the final active pharmaceutical ingredients meet stringent regulatory specifications. The ability to predict and control the impurity profile through mechanistic understanding is a key factor in accelerating the regulatory approval timeline for new drug candidates utilizing these intermediates.
How to Synthesize Aryl Selenide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction parameters outlined in the patent data to ensure reproducibility and safety. The process begins with the precise weighing and dissolution of the styrene derivative, the electron-rich aromatic component, and the diselenide reagent in dry acetonitrile to create a homogeneous solution prior to catalyst addition. It is critical to maintain an inert atmosphere during the setup to prevent premature oxidation of the selenium species, although the reaction itself shows reasonable tolerance to ambient conditions once initiated. The addition of the ferric chloride catalyst should be done gradually to manage any exothermic effects, followed by heating the mixture to the specified 80°C for a duration of approximately 12 hours, with progress monitored via thin-layer chromatography. Detailed standard operating procedures for quenching, extraction, and purification are essential to isolate the product in its highest possible purity, leveraging the solubility differences between the organic product and inorganic salts.
- Dissolve styrene or its derivatives, electron-rich aromatic hydrocarbons, and diselenide in an organic solvent such as acetonitrile and mix uniformly.
- Add ferric chloride (FeCl3) catalyst to the mixture and react at a temperature below the solvent boiling point, typically around 80°C for 12 hours.
- Quench the reaction with saturated sodium chloride, extract with ethyl acetate, dry, concentrate, and purify via silica gel flash chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this FeCl3-catalyzed methodology presents a compelling value proposition driven by significant operational efficiencies and cost optimization opportunities. The shift away from expensive precious metal catalysts to abundant iron salts fundamentally alters the cost structure of the synthesis, removing the need for complex metal scavenging steps that often add considerable expense and time to the manufacturing cycle. This simplification of the workflow translates directly into reduced utility consumption and lower waste disposal costs, contributing to a more sustainable and economically attractive production model. Furthermore, the use of commodity chemicals like styrene and simple aromatic hydrocarbons ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. The robustness of the reaction conditions also implies a lower risk of batch failures, enhancing overall supply reliability and allowing for more accurate forecasting of delivery timelines. These factors collectively empower organizations to achieve substantial cost savings while maintaining the high quality standards required for downstream applications in the life sciences sector.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with ferric chloride eliminates the high capital expenditure associated with purchasing and recovering expensive metals like palladium or platinum. This change significantly lowers the raw material cost per kilogram of the final product, allowing for more competitive pricing strategies in the global market. Additionally, the one-pot nature of the reaction reduces the number of unit operations, which in turn decreases labor costs and energy consumption related to heating, cooling, and solvent recovery. The high yields reported in the patent examples mean that less starting material is wasted, further improving the atom economy and reducing the cost of goods sold. By streamlining the purification process due to cleaner reaction profiles, manufacturers can also save on chromatography media and solvent volumes, driving down the overall variable costs of production.
- Enhanced Supply Chain Reliability: Utilizing widely available feedstocks such as styrene derivatives and simple diselenides ensures that production schedules are not disrupted by the scarcity of exotic reagents. The stability of the ferric chloride catalyst allows for easier storage and handling compared to air-sensitive organometallic complexes, reducing the logistical complexity of inventory management. The mild reaction conditions reduce the wear and tear on reactor equipment, leading to less downtime for maintenance and higher overall equipment effectiveness. This reliability is crucial for meeting the just-in-time delivery requirements of major pharmaceutical clients who depend on consistent intermediate supplies for their own production lines. Moreover, the scalability of the process from gram to kilogram scales without significant re-optimization provides confidence in the ability to ramp up production quickly in response to market demand.
- Scalability and Environmental Compliance: The process aligns well with environmental regulations by minimizing the generation of hazardous heavy metal waste, which simplifies compliance reporting and reduces disposal fees. The use of acetonitrile, a solvent with established recovery protocols, facilitates closed-loop recycling systems that further reduce the environmental footprint of the manufacturing facility. The high atom efficiency of the three-component coupling means that a larger proportion of the input mass ends up in the final product, adhering to the principles of green chemistry and sustainability. Scaling this reaction to industrial volumes is straightforward due to the lack of highly exothermic or dangerous steps, making it suitable for large-scale batch reactors commonly found in fine chemical plants. This ease of scale-up ensures that the commercial supply of these valuable aryl selenides can be expanded rapidly to support growing market needs without compromising on safety or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this method for their own production pipelines. Understanding these details is essential for making informed decisions about process adoption and integration into existing manufacturing workflows. Comprehensive answers provide the necessary technical depth to satisfy the due diligence requirements of R&D and quality assurance teams.
Q: What are the optimal reaction conditions for this aryl selenide synthesis?
A: According to patent CN113200899A, the optimal conditions involve using acetonitrile as the solvent, ferric chloride as the catalyst, and maintaining a reaction temperature of 80°C for approximately 12 hours to achieve high yields.
Q: What is the substrate scope for this Friedel-Crafts alkyl selenization?
A: The method demonstrates a broad substrate scope, accommodating various styrene derivatives (including those with electron-withdrawing or donating groups), diverse electron-rich aromatic hydrocarbons like trimethoxybenzenes and indoles, and multiple diselenide variants.
Q: Why is ferric chloride preferred over other catalysts for this transformation?
A: Ferric chloride is preferred due to its stability, abundance, low toxicity, and economic advantages compared to other Lewis acids or iodine catalysts, while simultaneously delivering superior reaction yields and ecological benefits.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Selenide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this FeCl3-catalyzed synthesis route and are fully equipped to leverage it for the benefit of our global clientele. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aryl selenide intermediate meets the exacting standards required for pharmaceutical and electronic applications. Our commitment to technical excellence allows us to navigate the complexities of organoselenium chemistry with precision, delivering products that consistently perform in downstream reactions. By partnering with us, clients gain access to a supply chain that is not only robust and reliable but also deeply knowledgeable about the nuances of advanced organic synthesis.
We invite you to engage with our technical procurement team to discuss how this innovative manufacturing process can be tailored to your specific project requirements. Request a Customized Cost-Saving Analysis today to understand the economic benefits of switching to this efficient protocol for your supply chain. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate the viability of this approach for your target molecules. Let us collaborate to drive down costs and accelerate your time to market with high-quality aryl selenide intermediates produced through state-of-the-art catalytic technology.
