Advanced Synthesis of N-(Methylthio-sulfur Carbonyl)-17-Steroidal Hydrazones for Oncology Applications
Advanced Synthesis of N-(Methylthio-sulfur Carbonyl)-17-Steroidal Hydrazones for Oncology Applications
The pharmaceutical landscape is constantly evolving, driven by the urgent need for more effective anticancer agents with reduced toxicity profiles. Patent CN101445541B introduces a groundbreaking class of N-(methylthio-sulfur carbonyl)-17-steroidal hydrazone compounds that represent a significant leap forward in medicinal chemistry. This technology leverages the robust biological framework of steroids, known for their protein assimilation functions, and conjugates them with thiosemicarbazone moieties, which are renowned for their diverse bioactivities including antitumor properties. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this patent offers a compelling pathway to develop next-generation oncology therapeutics. The innovation lies not just in the novel chemical structure but in the efficient and scalable synthetic methodology that allows for the rapid generation of diverse analogues for drug screening programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of steroidal anticancer drugs has relied heavily on modifying existing hormone structures with alkylating groups, such as nitrogen mustards, to create agents like Prednimustine and Estramustine phosphate. While these drugs have seen clinical success, they often suffer from significant limitations, including severe systemic toxicity, the development of drug resistance, and complex multi-step synthesis routes that drive up manufacturing costs. Furthermore, traditional aromatase inhibitors like Formestane and Exemestane, while effective for breast cancer, operate through specific enzymatic inhibition mechanisms that may not be universally applicable across different cancer types. The chemical modification of the steroid nucleus at specific positions, such as C-4 or C-6, often requires harsh reaction conditions and protecting group strategies that reduce overall atom economy. These conventional approaches frequently struggle to balance high potency with metabolic stability, leading to suboptimal pharmacokinetic profiles that hinder their long-term therapeutic utility in treating aggressive malignancies.
The Novel Approach
In stark contrast, the methodology described in CN101445541B utilizes a direct condensation strategy that functionalizes the C-17 position of the steroid skeleton with an N-(methylthio-sulfur carbonyl) hydrazone group. This approach is chemically elegant and operationally simple, bypassing the need for complex catalytic cycles or hazardous reagents often associated with traditional steroid functionalization. By introducing sulfur and nitrogen heteroatoms directly into the side chain, the resulting compounds gain unique electronic properties that enhance their interaction with biological targets, potentially overcoming resistance mechanisms seen with older drugs. The reaction proceeds under mild thermal conditions in common protic solvents, significantly reducing energy consumption and safety risks associated with high-pressure or cryogenic processes. This streamlined synthesis not only accelerates the timeline from bench to bedside but also aligns perfectly with the industry's demand for cost reduction in API manufacturing by minimizing waste and simplifying purification protocols.
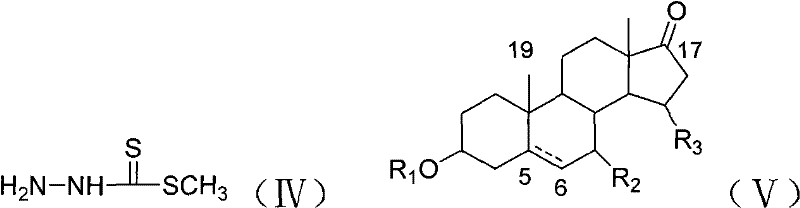
Mechanistic Insights into Steroidal Hydrazone Condensation
The core of this technological advancement lies in the nucleophilic addition-elimination mechanism between the carbonyl group of the sterone and the hydrazine moiety of the reagent. The reaction initiates with the nucleophilic attack of the terminal nitrogen of the methyl hydrazinodithioformate on the electrophilic carbon of the steroid ketone at the C-17 position. This forms a tetrahedral intermediate which subsequently undergoes dehydration to yield the stable C=N double bond characteristic of hydrazones. The presence of the thiocarbonyl group (C=S) adjacent to the hydrazone linkage is critical, as it introduces significant resonance stabilization and alters the electron density distribution across the molecule. This structural feature is believed to facilitate stronger hydrogen bonding interactions with enzyme active sites or DNA bases within cancer cells, thereby enhancing cytotoxicity. Understanding this mechanism is vital for process chemists aiming to optimize reaction kinetics and minimize the formation of geometric isomers that could complicate downstream purification efforts.
Furthermore, the preparation of the key reagent, methyl hydrazinodithioformate, is a crucial upstream step that dictates the purity of the final product. As illustrated in the patent, this reagent is synthesized via the reaction of hydrazine hydrate with carbon disulfide in the presence of a base, followed by S-methylation. This sequence ensures the generation of a highly reactive nucleophile capable of efficiently attacking the sterically hindered C-17 ketone of the steroid backbone. The control of temperature during this reagent synthesis is paramount to prevent the decomposition of the unstable hydrazine intermediate. By mastering this two-stage process—reagent preparation followed by condensation—manufacturers can achieve consistent batch-to-batch quality. The incorporation of heteroatoms like sulfur and nitrogen creates a pharmacophore that mimics natural substrates while resisting metabolic degradation, offering a distinct advantage over purely carbon-based modifications in terms of biological half-life and efficacy.
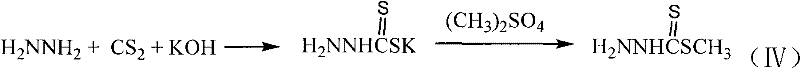
How to Synthesize N-(Methylthio-sulfur Carbonyl)-17-Steroidal Hydrazones Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these high-value intermediates with excellent reproducibility. The process begins with the careful preparation of the hydrazine reagent, ensuring strict temperature control to maintain stability, followed by its immediate reaction with the chosen sterone substrate. The condensation is typically carried out in anhydrous ethanol, a solvent choice that balances solubility of the organic reactants with the ability to facilitate the removal of water formed during the reaction. Monitoring the reaction progress via Thin Layer Chromatography (TLC) allows for precise determination of the endpoint, preventing over-reaction or decomposition of the sensitive hydrazone bond. Once the reaction is complete, the product often precipitates out of the solution upon cooling, simplifying the isolation process significantly compared to liquid-liquid extraction methods. For a detailed breakdown of the specific operational parameters, stoichiometry, and safety precautions required for this synthesis, please refer to the standardized guide below.
- Preparation of Methyl Hydrazinodithioformate: React hydrazine hydrate with carbon disulfide and potassium hydroxide, followed by methylation with dimethyl sulfate under controlled low temperatures.
- Condensation Reaction: Reflux the prepared methyl hydrazinodithioformate with the target sterone (e.g., epiandrosterone) in anhydrous ethanol at 70-80°C for 3-5 hours.
- Purification: Cool the reaction mixture to room temperature, filter the resulting precipitate, wash with cold ethanol, and recrystallize from absolute ethanol to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The reliance on commodity chemicals such as ethanol, hydrazine, and carbon disulfide means that raw material sourcing is not constrained by geopolitical bottlenecks or single-source supplier dependencies. This abundance of feedstock translates directly into enhanced supply chain reliability, ensuring that production schedules can be maintained even during periods of market volatility. Moreover, the simplicity of the work-up procedure, which primarily involves filtration and recrystallization, eliminates the need for expensive chromatographic purification steps that are often the bottleneck in scaling up fine chemical processes. This operational efficiency allows for a drastic simplification of the manufacturing workflow, reducing the footprint of the production facility and lowering the overall capital expenditure required for equipment.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of ambient pressure conditions significantly lower the operational costs associated with this synthesis. By avoiding precious metals, manufacturers also sidestep the rigorous and costly regulatory requirements for heavy metal residue testing in the final API, streamlining the quality control process. The high atom economy of the condensation reaction ensures that the majority of the starting material mass is incorporated into the final product, minimizing waste disposal costs and maximizing yield per batch. These factors collectively contribute to a leaner manufacturing model that supports competitive pricing strategies in the global pharmaceutical market.
- Enhanced Supply Chain Reliability: The use of standard glass-lined reactors and common solvents means that this process can be easily transferred between different manufacturing sites without the need for specialized infrastructure. This flexibility is crucial for mitigating risks associated with plant downtime or regional disruptions. Additionally, the stability of the intermediate reagents allows for potential stockpiling strategies, further buffering the supply chain against short-term fluctuations in raw material availability. The robust nature of the chemistry ensures that minor variations in process parameters do not lead to catastrophic batch failures, thereby guaranteeing a consistent flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of aqueous washes and spent solvent that can be readily treated or recycled using standard industrial methods. The absence of toxic byproducts aligns with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing facilities. Scaling this reaction from laboratory to commercial tonnage is straightforward, as the exothermic nature of the condensation is manageable with standard cooling systems. This scalability ensures that the technology can meet the growing demand for novel anticancer agents without compromising on safety or environmental stewardship, making it an attractive option for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel steroidal hydrazones. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on the feasibility and potential of this chemical platform. Understanding these nuances is essential for stakeholders evaluating the integration of this technology into their existing drug discovery pipelines or manufacturing portfolios. The answers reflect a commitment to transparency and technical accuracy, ensuring that partners have a clear understanding of the capabilities and limitations of the described synthesis.
Q: What is the primary therapeutic potential of N-(methylthio-sulfur carbonyl)-17-steroidal hydrazones?
A: These compounds exhibit significant anticancer activity, particularly against human gastric, oral epithelial, liver, and lung cancer cell lines, due to the synergistic effect of the steroid skeleton and the thiosemicarbazone pharmacophore.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the process utilizes standard condensation reactions in common solvents like ethanol with mild heating (70-80°C), making it highly scalable and cost-effective for commercial manufacturing without requiring exotic catalysts.
Q: What are the key starting materials required for this synthesis?
A: The synthesis relies on readily available steroidal ketones such as epiandrosterone or dehydroepiandrosterone and methyl hydrazinodithioformate, which can be prepared in situ from basic chemical reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(Methylthio-sulfur Carbonyl)-17-Steroidal Hydrazone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chemistry described in CN101445541B and are fully equipped to support its development from gram-scale research to commercial production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to the manufacturing floor. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-(methylthio-sulfur carbonyl)-17-steroidal hydrazone meets the highest international standards for pharmaceutical intermediates. Our commitment to quality is matched only by our dedication to speed and efficiency, allowing us to respond rapidly to the dynamic needs of the global oncology drug market.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthetic route. By leveraging our expertise in process optimization, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can accelerate the development of life-saving anticancer therapies and bring these promising new molecules to patients who need them most, ensuring a reliable supply of high-purity pharmaceutical intermediates for the future.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
