Revolutionizing 4-Substituted Oxazolidinone Production: A Green, One-Step Commercial Scale-Up Strategy
Revolutionizing 4-Substituted Oxazolidinone Production: A Green, One-Step Commercial Scale-Up Strategy
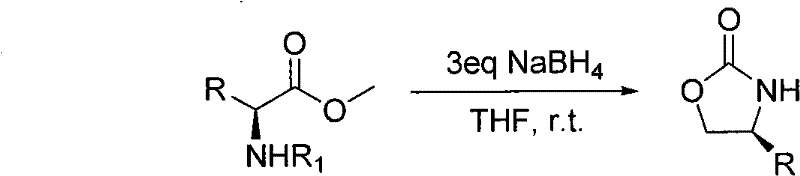
The landscape of fine chemical manufacturing is undergoing a critical transformation, driven by the urgent need for greener, more efficient synthetic routes that do not compromise on purity or scalability. Patent CN101948443B introduces a groundbreaking methodology for the synthesis of 4-substituted oxazolidinone derivatives, a class of compounds indispensable to the pharmaceutical and agrochemical industries. This innovation replaces hazardous, multi-step traditional protocols with a streamlined, one-step reductive cyclization using sodium borohydride. For R&D directors and supply chain leaders, this represents a paradigm shift towards sustainable manufacturing, offering a pathway to produce high-purity intermediates while drastically simplifying process engineering and waste management protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-oxazolidinone and its derivatives has relied heavily on the use of phosgene, phosgene derivatives, urea, cyanates, or selenium-catalyzed carbonylation methods. These conventional pathways are fraught with significant operational and environmental challenges that hinder efficient commercial production. The use of phosgene and cyanates necessitates extremely harsh reaction conditions, often involving high pressures or temperatures, which leads to severe equipment corrosion and poses substantial safety risks to personnel. Furthermore, methods employing selenium or palladium catalysts introduce the complexity of heavy metal removal, requiring additional purification steps that increase both production time and cost. The reliance on such toxic and expensive reagents creates a bottleneck for scalable manufacturing, as regulatory compliance regarding hazardous waste disposal becomes increasingly stringent and costly for modern chemical enterprises.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a mild, one-step reduction strategy that fundamentally alters the economic and safety profile of oxazolidinone production. By employing sodium borohydride as the sole reducing agent in a polar aprotic solvent like tetrahydrofuran, the process achieves high yields under ambient room temperature conditions without the need for any cocatalyst bases. This simplification eliminates the requirement for specialized high-pressure reactors or corrosion-resistant alloys, thereby lowering capital expenditure for production facilities. The absence of toxic gases like carbon monoxide or phosgene ensures a safer working environment, while the use of inexpensive, commodity-grade reagents significantly reduces the raw material cost basis. This method not only accelerates the reaction timeline but also facilitates easier downstream processing, making it an ideal candidate for continuous flow chemistry or large-batch manufacturing.
Mechanistic Insights into NaBH4-Mediated Reductive Cyclization
The core of this technological breakthrough lies in the elegant mechanism of reductive cyclization, where the ester functionality of the N-protected amino acid ester is selectively reduced to an intermediate alcohol, which subsequently undergoes intramolecular nucleophilic attack on the carbamate carbonyl. This cascade occurs seamlessly in a single pot, driven by the hydride source provided by sodium borohydride. The reaction conditions are meticulously optimized to ensure that the reduction potential is sufficient to convert the ester but controlled enough to prevent over-reduction or degradation of the sensitive oxazolidinone ring system. The use of three equivalents of NaBH4 ensures complete conversion of the starting material, driving the equilibrium towards the desired cyclic product. This mechanistic efficiency is crucial for maintaining high atom economy, as it minimizes the formation of side products that would otherwise complicate purification and lower overall process yield.
For R&D teams focused on chiral synthesis, the preservation of stereochemical integrity is paramount, and this protocol excels in maintaining optical purity throughout the transformation. The mild, non-acidic, and non-basic nature of the reaction medium prevents the epimerization of the chiral center at the 4-position of the oxazolidinone ring, a common pitfall in harsher cyclization methods. As demonstrated in the patent examples, chiral substrates yield single enantiomers with no detectable racemization, ensuring that the resulting 4-substituted oxazolidinones retain their utility as high-performance chiral auxiliaries or bioactive intermediates. This robustness against racemization is a critical quality attribute for pharmaceutical applications, where enantiomeric excess directly correlates with drug efficacy and safety profiles.
How to Synthesize 4-Substituted Oxazolidinones Efficiently
The implementation of this synthesis route is designed for operational simplicity, allowing chemists to transition from bench-scale discovery to pilot production with minimal friction. The standardized protocol involves dissolving the N-protected amino acid ester in THF, followed by the controlled addition of sodium borohydride, a procedure that can be easily automated or scaled in standard glass-lined reactors. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup parameters are essential for maximizing yield and consistency across batches. For a comprehensive, step-by-step technical guide on executing this synthesis, please refer to the standardized protocol section below.
- Dissolve the N-protected alpha-substituted amino acid ester substrate in tetrahydrofuran (THF) under ambient conditions.
- Slowly add 3 equivalents of sodium borohydride (NaBH4) to the reaction mixture while monitoring progress via TLC.
- Upon completion, quench with dilute HCl, neutralize with bicarbonate, extract with ethyl acetate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative advantages that directly impact the bottom line and operational resilience. By shifting away from scarce, regulated, or hazardous reagents like phosgene and palladium, manufacturers can secure a more stable and predictable supply chain for raw materials. The substitution of expensive transition metal catalysts with commodity chemicals like sodium borohydride decouples production costs from the volatile pricing of precious metals. Furthermore, the simplified workup procedure, which involves standard extraction and crystallization rather than complex chromatographic separations or metal scavenging, significantly reduces the consumption of solvents and consumables. This streamlining of the manufacturing process translates into shorter cycle times and higher throughput, enabling suppliers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and toxic reagents results in substantial cost savings across the entire production lifecycle. Without the need for specialized metal removal resins or extensive wastewater treatment for heavy metals, the operational expenditure is drastically lowered. The high atom economy of the one-step process ensures that raw material utilization is maximized, reducing the cost per kilogram of the final API intermediate. Additionally, the ability to recover and recycle the organic solvent further enhances the economic viability of the process, making it a highly competitive option for cost-sensitive bulk manufacturing.
- Enhanced Supply Chain Reliability: Sourcing sodium borohydride and THF is significantly less risky than procuring phosgene or selenium compounds, which are subject to strict regulatory controls and transportation restrictions. This accessibility ensures a continuous supply of key inputs, minimizing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions also means that the process is less susceptible to variations in utility quality or minor environmental fluctuations, leading to more consistent batch-to-batch quality and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The benign nature of the reagents and the absence of gaseous byproducts make this process inherently scalable from kilogram to multi-ton quantities without significant engineering hurdles. The reduction in hazardous waste generation aligns perfectly with modern environmental, social, and governance (ESG) goals, reducing the burden on waste treatment facilities and lowering compliance costs. This green chemistry approach not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible and sustainable partner in the global value chain.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a set of frequently asked questions based on the patent data and practical application scenarios. These insights are designed to clarify the operational benefits and technical feasibility for potential partners evaluating this route for their supply chain. Understanding these nuances is critical for making informed decisions about process adoption and long-term sourcing strategies.
Q: Does this synthesis method preserve the chirality of the starting material?
A: Yes, the patent explicitly states that for chiral substrates, neither the reaction process nor the post-treatment causes racemization, ensuring the production of a single enantiomer with high optical purity.
Q: What are the primary safety advantages over traditional phosgene methods?
A: This method eliminates the need for toxic phosgene, isocyanates, or heavy metal catalysts like selenium or palladium, significantly reducing equipment corrosion risks and hazardous waste generation.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Absolutely. The process operates at room temperature, uses inexpensive and readily available reagents like NaBH4, and allows for easy solvent recovery, making it highly conducive to large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Substituted Oxazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the one described in CN101948443B to enhance the competitiveness of our product portfolio. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4-substituted oxazolidinone meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cost-effective and environmentally friendly technology for their projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and reliability in your supply chain.
