Advanced Synthesis of Sulfur-Bridged Diperylene Tetraimides for Organic Electronics
Advanced Synthesis of Sulfur-Bridged Diperylene Tetraimides for Organic Electronics
The rapid evolution of organic electronics demands materials with superior charge transport properties and environmental stability. Patent CN101423522A introduces a groundbreaking class of diperylene-3,4,6,7:12,13,15,16-octacarboxylic tetraimide compounds featuring sulfur or selenium atom bridges at the side positions. These novel structures represent a significant leap forward for reliable electronic chemical supplier networks seeking next-generation organic semiconductors. By integrating heteroatoms directly into the fused-ring aromatic system, this technology addresses critical limitations in traditional perylene diimide derivatives, offering enhanced electron affinity and improved molecular packing. For R&D teams focused on organic photovoltaics and field-effect transistors, this patent provides a robust synthetic pathway to access these high-value intermediates with defined structural precision.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of perylene-based functional materials often struggles with achieving extended conjugation along the short molecular axis without compromising solubility or processability. Conventional methods typically rely on simple imidization of perylene tetracarboxylic dianhydrides, which limits the structural diversity available for tuning electronic bandgaps. Furthermore, introducing heteroatoms like sulfur or selenium into rigid fused-ring systems usually requires harsh conditions or multi-step functionalization sequences that result in poor overall yields and difficult purification processes. The lack of efficient protocols for creating fully conjugated, heteroatom-doped diperylene cores has historically bottlenecked the development of high-performance n-type organic semiconductors, forcing manufacturers to rely on less efficient carbon-only analogues.
The Novel Approach
The patented methodology revolutionizes this landscape by enabling the direct construction of sulfur or selenium-bridged diperylene cores through transition metal-catalyzed cyclization. This approach utilizes readily available tetrahalogenated perylene precursors and reacts them with inorganic sulfides or selenides in the presence of a copper catalyst and amino acid ligands. This one-pot strategy effectively fuses two perylene units while simultaneously incorporating the heteroatom bridge, drastically simplifying the synthetic route. The versatility of this method allows for the modulation of the final product's properties by simply switching between sulfur and selenium sources or altering the N-substituents. This flexibility is crucial for cost reduction in electronic chemical manufacturing, as it permits the fine-tuning of material characteristics without necessitating entirely new synthetic pipelines.
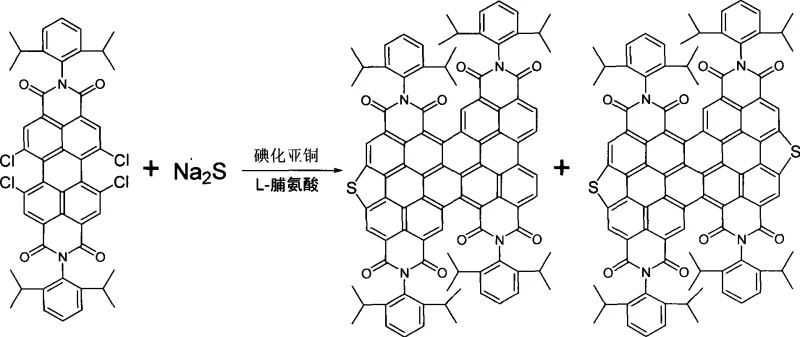
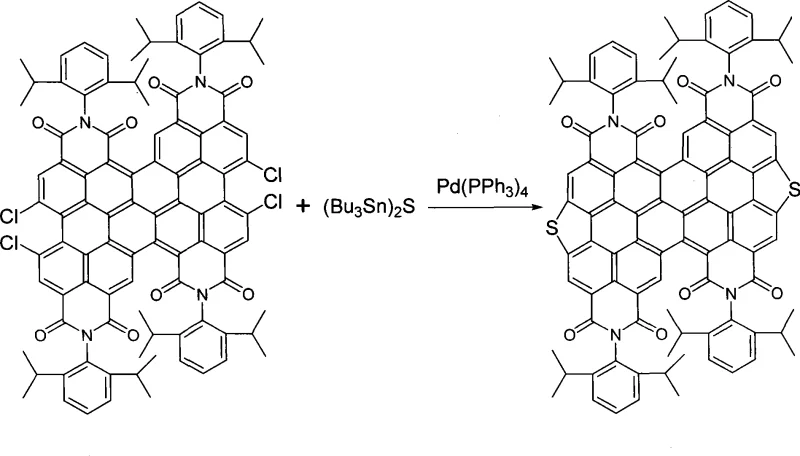
Mechanistic Insights into Cu-Catalyzed and Pd-Catalyzed Cyclization
The core innovation lies in the dual catalytic systems described, each offering distinct advantages for specific production scales. The primary route employs a copper-catalyzed mechanism, likely proceeding through an Ullmann-type coupling where the copper species, stabilized by L-proline ligands, facilitates the nucleophilic attack of the sulfide anion on the halogenated aromatic ring. This process promotes the formation of C-S bonds and subsequent intramolecular cyclization to close the central ring. The use of amino acid ligands is particularly noteworthy as it enhances the solubility of the copper catalyst in polar aprotic solvents like DMSO or DMF, ensuring homogeneous reaction conditions that are vital for consistent impurity profiles. This mechanistic understanding allows chemists to optimize reaction parameters such as temperature (30°C to 150°C) and stoichiometry to maximize the formation of the desired mono- or di-bridged species.
Alternatively, the patent discloses a high-yield pathway utilizing palladium catalysis with organotin reagents, which operates via a Stille-type cross-coupling mechanism. In this route, pre-formed diperylene precursors containing halogen handles react with sulfur-containing organotin compounds in the presence of Pd(PPh3)4. This method is exceptionally powerful for commercial scale-up of complex organic semiconductors because it achieves purified yields ranging from 60% to 95%, significantly outperforming the copper-catalyzed variant. The palladium cycle efficiently mediates the transmetallation and reductive elimination steps required to forge the C-S bonds with high fidelity. Understanding these mechanistic nuances empowers procurement teams to select the appropriate grade of material based on their specific purity requirements and budget constraints, balancing the cost of precious metal catalysts against the value of higher throughput.
How to Synthesize Diperylene Tetraimides Efficiently
Executing this synthesis requires careful control of reaction atmospheres and precise stoichiometric balancing of the halogenated precursors with the chalcogen sources. The process generally involves mixing the tetrahalogenated perylene diimide with sodium sulfide or selenide, a copper or palladium catalyst, and an inorganic base in a suitable organic solvent. The reaction mixture must be heated under inert gas protection to facilitate the cyclization while preventing oxidation of the sensitive intermediates. Detailed standardized synthetic steps for both the copper-mediated and palladium-mediated pathways are outlined below to ensure reproducibility and safety in your laboratory operations.
- Mix tetrahalogenated perylene diimide precursors with sodium sulfide or selenide, a copper or palladium catalyst, an amino acid ligand, and an inorganic base.
- Add an organic solvent such as DMSO, DMF, or toluene to the mixture under an inert gas atmosphere.
- Heat the solution to between 30°C and 150°C for 6 to 40 hours, then purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this technology offers substantial benefits by streamlining the production of high-value organic electronic materials. The reliance on commercially available starting materials, such as tetrachloroperylene diimides and common inorganic salts, mitigates the risk of raw material shortages that often plague specialty chemical manufacturing. Furthermore, the ability to utilize standard solvents like toluene, DMSO, and ethyl acetate simplifies waste management and solvent recovery processes, aligning with modern environmental compliance standards. For supply chain heads, this translates to a more resilient sourcing strategy where the dependency on exotic or hard-to-source reagents is minimized, ensuring continuous availability of critical intermediates for downstream device fabrication.
- Cost Reduction in Manufacturing: The implementation of the palladium-catalyzed route described in the patent enables a drastic improvement in overall process efficiency by achieving significantly higher yields compared to traditional methods. By eliminating the need for multiple intermediate isolation steps and reducing the formation of byproducts, the total cost of goods sold is substantially lowered. The use of earth-abundant copper catalysts in the alternative route further provides a cost-effective option for applications where ultra-high yields are less critical than raw material expense, allowing for flexible budget allocation across different product tiers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvent systems, ensures that production can be maintained consistently even with minor fluctuations in utility supplies. The synthetic pathway avoids the use of highly unstable or pyrophoric reagents that often require specialized storage and handling infrastructure, thereby reducing logistical complexities. This stability enhances the reliability of the supply chain, as manufacturers can maintain steady production schedules without the frequent interruptions associated with more sensitive chemical processes.
- Scalability and Environmental Compliance: The procedures outlined are inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot production without fundamental changes to the reaction engineering. The workup procedures involve standard aqueous washes and column chromatography, which are well-established unit operations in the fine chemical industry. This ease of scale-up facilitates the rapid transition from R&D to commercial production, while the use of less toxic sulfur sources compared to alternative thiolation reagents supports better environmental, health, and safety (EHS) outcomes.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these novel diperylene derivatives. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy for your technical assessment. Understanding these details is essential for evaluating the feasibility of integrating these materials into your existing product development workflows.
Q: What are the primary applications of sulfur-bridged diperylene tetraimides?
A: These compounds are primarily used as functional materials in organic electronics, including solar cell materials, liquid crystal displays, and organic field-effect transistors (OFETs), due to their excellent electron acceptor properties and broad-spectrum absorption.
Q: How does the choice of catalyst affect the yield in this synthesis?
A: The patent describes two main pathways: a copper-catalyzed route yielding 10%-40% and a palladium-catalyzed route using organotin reagents that significantly increases yield to between 60% and 95%.
Q: Why are sulfur or selenium atoms introduced into the perylene structure?
A: Introducing sulfur or selenium atoms enhances the molecule's stability, solubility, and rigidity through intermolecular interactions like S-S or Se-Se bonding, improving overall material performance in electronic devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diperylene Tetraimides Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercially viable chemical solutions. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of diperylene tetraimides meets the exacting standards required for high-performance organic electronic applications. We understand the critical nature of material purity in determining device efficiency and longevity.
We invite you to collaborate with our technical procurement team to explore how these sulfur-bridged architectures can enhance your product portfolio. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Let us help you secure a competitive advantage through superior material science and supply chain excellence.
