Advanced Synthesis of Symmetric Two-Photon Luminescent Compounds for High-Performance Imaging
Advanced Synthesis of Symmetric Two-Photon Luminescent Compounds for High-Performance Imaging
The rapid evolution of optoelectronic technologies demands materials with superior nonlinear optical properties, particularly for applications in three-dimensional information storage and high-resolution biological imaging. Patent CN101768112B introduces a groundbreaking class of two-photon induced luminescent compounds characterized by a symmetric terminal structure based on 1,2,2,4-tetramethyldihydroquinoline. This innovation addresses critical limitations in current luminescent materials by combining a robust stilbene skeleton with a rigidified nitrogen-containing heterocyclic system. For R&D directors and procurement specialists in the electronic chemical sector, this patent represents a significant leap forward in achieving high fluorescence quantum yields while maintaining structural stability. The disclosed synthesis method offers a reliable pathway for producing these advanced materials, positioning them as ideal candidates for next-generation fluorescent probes and optical storage devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to developing two-photon absorbing materials have often relied on asymmetric molecular architectures or sol-gel processing techniques that introduce significant performance bottlenecks. Asymmetric molecules typically exhibit lower two-photon absorption cross-sections because the lack of symmetry disrupts the efficient charge transfer required for strong nonlinear optical responses. Furthermore, conventional methods for creating solid-state luminescent materials frequently utilize sol-gel glass matrices, which are notoriously prone to brittleness and cracking during thermal treatment. These mechanical failures not only compromise the integrity of the optical device but also lead to substantial light scattering losses due to the inherent porosity of the gel glass. Consequently, the overall efficiency and durability of devices based on these legacy technologies are severely restricted, limiting their practical application in demanding environments such as in vivo imaging or long-term data storage.
The Novel Approach
The methodology outlined in CN101768112B overcomes these historical challenges through a dual strategy of molecular symmetry and advanced matrix engineering. By designing molecules with a symmetric stilbene backbone flanked by 1,2,2,4-tetramethyldihydroquinoline groups, the inventors have successfully maximized the conjugation length and electron-donating capability without inducing excessive rotational freedom that leads to non-radiative decay. This structural rigidity is further enhanced by locking the nitrogen atom within a ring system, which drastically reduces energy loss and boosts the fluorescence quantum yield to impressive levels, far exceeding previous benchmarks. Additionally, the patent proposes doping these compounds into an epoxy resin solid matrix rather than fragile glass. This novel composite approach yields materials with exceptional transparency, mechanical flexibility, and resistance to thermal stress, effectively eliminating the scattering losses associated with porous gel glasses and ensuring consistent optical performance over extended operational lifetimes.
Mechanistic Insights into Horner-Wadsworth-Emmons Condensation
The core chemical transformation enabling this technology is a base-catalyzed condensation reaction, specifically utilizing a Horner-Wadsworth-Emmons (HWE) type mechanism to construct the central stilbene double bond. In this process, a phosphonate ester reagent, such as 2,5-substituted 1,4-bis(diethoxyphosphoryl)methyl-benzene, acts as the nucleophile precursor after deprotonation by sodium methoxide in an anhydrous DMF solvent. The resulting carbanion attacks the carbonyl carbon of the 1,2,2,4-tetramethyl-1,2-dihydro-6-formylquinoline aldehyde, followed by elimination of the phosphate group to form the trans-alkene linkage. This reaction pathway is highly advantageous for industrial synthesis because it proceeds efficiently at room temperature, avoiding the harsh thermal conditions often required for Wittig reactions. The use of DMF as a polar aprotic solvent ensures excellent solubility for both the organic reactants and the ionic intermediates, facilitating a homogeneous reaction environment that promotes high conversion rates and minimizes side product formation.
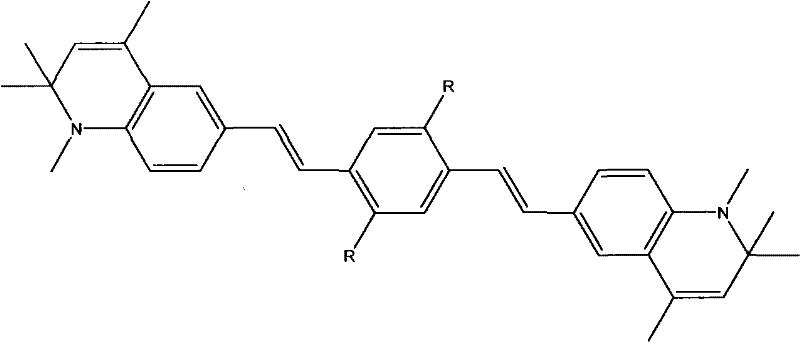
Beyond the synthesis mechanics, the photophysical mechanism of these compounds is equally critical for their end-use performance. The symmetric arrangement of strong electron-donating dihydroquinoline groups at both ends of the conjugated system creates a push-pull electronic distribution that significantly enhances the two-photon absorption cross-section. Crucially, the cyclization of the nitrogen atom prevents the free rotation of the amine group, a common defect in linear aminostilbenes that typically dissipates excited state energy as heat. By restricting this intramolecular rotation, the molecule channels a much higher proportion of absorbed energy into radiative fluorescence emission. This results in a fluorescence quantum yield of approximately 0.83 for the optimized derivatives, a substantial improvement over the 0.112 yield observed in comparable asymmetric structures. This mechanistic optimization ensures that the material delivers bright, stable signals essential for high-sensitivity detection applications.
How to Synthesize 1,2,2,4-Tetramethyldihydroquinoline Derivatives Efficiently
The synthesis protocol described in the patent provides a robust framework for laboratory and pilot-scale production of these high-value optoelectronic intermediates. The process is designed to be operationally simple, requiring standard chemical equipment and avoiding the need for inert atmosphere gloveboxes or cryogenic cooling, which simplifies the barrier to entry for manufacturing partners. The reaction relies on precise stoichiometric control, typically employing a molar ratio of aldehyde to phosphonate to base of roughly 2.2:1:6 to drive the equilibrium towards the desired symmetric product. Following the reaction period, the workup involves a straightforward precipitation into water, followed by purification via silica gel chromatography to remove unreacted starting materials and phosphonate byproducts. Detailed standardized synthetic steps see the guide below.
- Prepare the reaction mixture by dissolving the phosphate reagent in anhydrous DMF and adding sodium methoxide under stirring.
- Introduce the 1,2,2,4-tetramethyl-1,2-dihydro-6-formylquinoline aldehyde to the mixture and react at room temperature for 1.5 to 3 hours.
- Quench the reaction in water, filter the precipitate, wash with hot ethanol, and purify the final solid via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling economic and logistical benefits compared to traditional noble-metal catalyzed couplings. The elimination of expensive palladium or platinum catalysts not only reduces the direct material cost but also removes the complex and costly downstream purification steps required to meet strict heavy metal residue specifications for electronic grade chemicals. Furthermore, the reliance on commodity chemicals like sodium methoxide and DMF ensures a stable and diversified supply base, mitigating the risk of shortages that often plague specialized reagent markets. The mild reaction conditions also translate to lower energy consumption during production, contributing to a reduced carbon footprint and aligning with increasingly stringent environmental compliance standards required by global multinational corporations.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive phosphonate esters and avoiding precious metal catalysts entirely. This catalyst-free approach eliminates the need for specialized scavenging resins or activated carbon treatments to remove trace metals, thereby streamlining the purification workflow and reducing overall processing time. Additionally, the high selectivity of the room temperature condensation reaction minimizes the formation of difficult-to-separate isomers, leading to higher crude yields and reduced solvent usage during chromatography. These factors collectively drive down the cost per kilogram of the final active pharmaceutical ingredient or electronic material, enhancing margin potential for downstream manufacturers.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key raw materials, including substituted benzyl phosphonates and quinoline aldehydes, are derived from widely available petrochemical feedstocks. Unlike supply chains dependent on geographically concentrated rare earth elements or conflict minerals, the precursors for this synthesis can be procured from multiple global vendors, ensuring continuity of supply even during regional disruptions. The robustness of the chemical process itself, which tolerates minor variations in temperature and mixing without significant yield loss, further guarantees consistent batch-to-batch quality. This reliability allows supply chain planners to maintain leaner inventory levels while confidently meeting just-in-time delivery schedules for critical optoelectronic projects.
- Scalability and Environmental Compliance: The synthesis is inherently scalable due to its exothermic nature being manageable at room temperature, removing the need for complex heating or cooling infrastructure during scale-up from grams to tons. The waste stream is primarily composed of aqueous salts and organic solvents which can be readily treated or recycled using standard industrial wastewater management systems, avoiding the generation of hazardous heavy metal sludge. This environmental profile simplifies the permitting process for new manufacturing facilities and reduces the long-term liability associated with hazardous waste disposal. Consequently, companies can expand production capacity rapidly to meet growing market demand for two-photon materials without facing significant regulatory hurdles or environmental bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on performance metrics and process feasibility. Understanding these details is crucial for technical teams evaluating the integration of these materials into existing product lines or new development pipelines.
Q: What is the primary advantage of the symmetric structure in these luminescent compounds?
A: The symmetric stilbene skeleton significantly enhances the two-photon absorption cross-section compared to asymmetric analogs, leading to superior fluorescence efficiency essential for deep-tissue imaging.
Q: How does the epoxy resin matrix improve material performance?
A: Unlike traditional sol-gel glass which is brittle and porous, the epoxy resin matrix provides excellent mechanical flexibility, high transparency, and prevents scattering losses, ensuring stable optical performance.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes mild room temperature conditions and common reagents like sodium methoxide and DMF, avoiding expensive transition metal catalysts, which facilitates easy scale-up and cost reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,2,4-Tetramethyldihydroquinoline Derivative Supplier
As the demand for high-performance nonlinear optical materials continues to surge in the biomedical and data storage sectors, having a manufacturing partner with deep technical expertise is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed analysis of two-photon absorption cross-sections and fluorescence quantum yields. We understand that the transition from laboratory synthesis to commercial scale requires meticulous attention to detail, and our team is dedicated to maintaining the high optical quality defined in patent CN101768112B throughout the entire production lifecycle.
We invite you to collaborate with us to optimize your supply chain for these advanced luminescent compounds. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity grades. Please contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can accelerate your product development timelines while reducing overall material costs. Let us be your strategic partner in bringing next-generation two-photon imaging technologies to market.
