Revolutionizing Organic Carboxylate Production with Ionic Liquid Promoters
Revolutionizing Organic Carboxylate Production with Ionic Liquid Promoters
The chemical manufacturing landscape is constantly evolving, driven by the urgent need for greener, more efficient synthetic routes that minimize environmental impact while maximizing yield. A pivotal advancement in this domain is detailed in Chinese Patent CN101781200B, which discloses a novel method for producing organic carboxylates through an amine transesterification reaction. This technology specifically addresses the longstanding challenges associated with synthesizing alpha-hydroxy carboxylic acid esters, which are critical building blocks in the pharmaceutical and agrochemical industries. By utilizing a unique catalytic system comprising metal amides or alkali metal alcoholates paired with ionic liquid promoters, this process operates under significantly milder conditions than conventional techniques. For procurement leaders and R&D directors seeking a reliable organic carboxylate supplier, understanding the mechanistic advantages of this patent is essential for securing a sustainable and cost-effective supply chain for high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of organic carboxylic acid esters, particularly those derived from alpha-hydroxy acids, has relied heavily on the alcoholysis of alkyl nitriles. This traditional pathway typically employs sulfuric acid as a catalyst to drive the reaction forward. While chemically effective, this approach generates substantial quantities of ammonium sulfate as a stoichiometric by-product. In the past, this by-product could be sold as fertilizer, but market saturation has turned it into a costly waste disposal liability, severely impacting the overall economics of the process. Furthermore, alternative methods described in prior art often necessitate the use of expensive precious metal catalysts, toxic metal carbonyls, or extremely harsh reaction conditions involving high temperatures and pressures that demand specialized, costly reactor alloys. These factors collectively create significant bottlenecks in cost reduction in fine chemical manufacturing, making the final products less competitive in a price-sensitive global market.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN101781200B introduces a transformative strategy that bypasses the formation of ammonium sulfate entirely. By shifting the feedstock to organic carboxylic acid amides and employing a dual-component catalytic system, the process achieves high efficiency without the environmental baggage of sulfate waste. The core innovation lies in the synergistic combination of a base catalyst, such as sodium amide or sodium methoxide, with an ionic liquid promoter. This combination allows the transesterification to proceed at moderate temperatures ranging from 60°C to 160°C and pressures between 10 to 40 kg/cm². This operational flexibility means that manufacturers can utilize standard stainless steel equipment rather than exotic high-pressure vessels, drastically lowering capital expenditure. For supply chain heads, this translates to enhanced supply chain reliability, as the process is less susceptible to the logistical complexities associated with handling hazardous strong acids or sourcing rare precious metals.
Mechanistic Insights into Ionic Liquid-Promoted Amine Transesterification
The efficacy of this synthesis route is rooted in the unique physicochemical properties of the ionic liquid promoter, which acts not merely as a solvent but as a crucial activity enhancer. The cation of the ionic liquid typically features a nitrogen-containing heterocyclic structure, such as the imidazolium ring shown below, which interacts favorably with the reaction intermediates to stabilize the transition state.
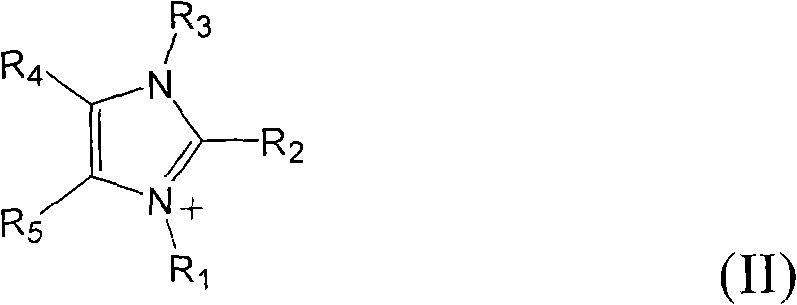
This structural motif, characterized by substituents R1 through R5 which can be varied to tune solubility and electronic properties, facilitates the dissolution of the metal amide catalyst and improves the nucleophilicity of the reacting species. The anion of the ionic liquid, which can range from halides like bromide to complex ions like hexafluorophosphate, further modulates the reaction environment to suppress side reactions. By carefully selecting the ionic liquid pair, the process achieves exceptional selectivity, often exceeding 90% for the desired ester product, even when dealing with sterically hindered substrates like alpha-hydroxyisobutyric acid amide. This level of control over the impurity profile is paramount for R&D directors who require high-purity organic carboxylate intermediates for downstream API synthesis, ensuring that purification steps are minimized and overall throughput is maximized.
The reaction mechanism proceeds through two primary pathways depending on the choice of reactants, both of which are elegantly simple yet highly effective. In the first pathway, the organic carboxylic acid amide reacts directly with an ester, exchanging the amine group for an alkoxy group. In the second pathway, the amide reacts with an alcohol in the presence of carbon monoxide, effectively carbonylating the alcohol in situ to form the ester while releasing formamide. The general reaction scheme for the direct transesterification with an ester is illustrated below, demonstrating the clean conversion of the amide starting material into the target ester.
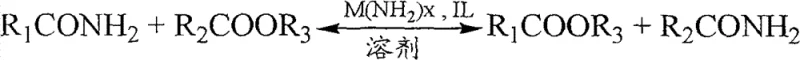
Alternatively, when utilizing the alcohol and carbon monoxide route, the process offers a versatile method to introduce the carbonyl functionality without needing pre-formed esters. This pathway is particularly advantageous for commercial scale-up of complex polymer additives or specialty chemicals where specific ester groups are required. The ability to toggle between these pathways provides manufacturers with immense flexibility in raw material sourcing. If a specific ester is expensive or unavailable, the alcohol and CO route serves as a robust backup, ensuring continuity of supply. This mechanistic versatility is a key differentiator that positions this technology as a superior choice for producing high-purity OLED material precursors or pharmaceutical intermediates where supply security is non-negotiable.
How to Synthesize Alpha-Hydroxy Carboxylic Acid Esters Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the ionic liquid promoter. The process begins with the careful selection of the metal amide catalyst, with sodium amide being a preferred choice due to its high reactivity and commercial availability. The ionic liquid promoter is then added in a molar ratio optimized to balance cost and performance, typically between 10% to 40% relative to the amide substrate. The reaction is conducted in a sealed pressure vessel capable of withstanding moderate pressures, heated to a target temperature that balances reaction rate with selectivity. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the organic carboxylic acid amide substrate with the chosen ester or alcohol solvent in a pressure reactor.
- Add the metal amide catalyst (such as sodium amide) and the ionic liquid promoter (such as [BMIM]PF6) to the mixture under inert atmosphere.
- Heat the reactor to a temperature between 60°C and 160°C and maintain carbon monoxide pressure if using the alcohol pathway, stirring for 0.2 to 5 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this ionic liquid-promoted technology offers compelling economic and operational advantages that go beyond simple yield improvements. The most significant benefit is the elimination of the ammonium sulfate waste stream, which removes a major cost center associated with waste treatment and regulatory compliance. This shift aligns perfectly with modern green chemistry initiatives, reducing the environmental footprint of the manufacturing site and mitigating the risk of future regulatory penalties related to sulfate discharge. Furthermore, the use of readily available base metal catalysts like sodium amide or sodium methoxide replaces the need for expensive and supply-constrained precious metals, leading to substantial cost savings in raw material procurement.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the drastic simplification of the downstream processing requirements. Because the reaction exhibits high selectivity, the crude product contains fewer impurities, reducing the load on distillation columns and crystallization units. Additionally, the mild reaction conditions (temperatures around 100°C) significantly lower energy consumption compared to traditional high-temperature processes. The avoidance of sulfuric acid also eliminates the corrosion costs associated with maintaining acid-resistant equipment, allowing for the use of standard stainless steel reactors which have a longer lifecycle and lower maintenance overhead.
- Enhanced Supply Chain Reliability: From a logistics perspective, the raw materials for this process are commodity chemicals with stable global supply chains. Sodium amide, methanol, and common ionic liquids are produced at scale by multiple vendors, reducing the risk of single-source dependency. This diversification ensures reducing lead time for high-purity organic carboxylates, as manufacturers are not held hostage by the production schedules of niche catalyst suppliers. The robustness of the reaction also means that batch-to-batch variability is minimized, providing customers with consistent quality and predictable delivery schedules essential for Just-In-Time manufacturing environments.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in laboratory and pilot settings with clear pathways to multi-ton production. The absence of toxic heavy metals and corrosive acids simplifies the permitting process for new production lines, accelerating time-to-market for new products. Moreover, the by-products generated, such as formamide or exchanged amides, are often valuable chemicals in their own right or are easier to treat than inorganic salts. This circular economy approach enhances the overall sustainability profile of the facility, appealing to end-users in the pharmaceutical and consumer goods sectors who prioritize environmentally responsible sourcing.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the operational parameters and scope of this patent. The following insights are derived directly from the experimental data and claims within CN101781200B, providing a factual basis for feasibility assessments.
Q: What are the primary advantages of using ionic liquids in this transesterification process?
A: Ionic liquids act as highly effective promoters that significantly enhance selectivity towards the desired ester product while allowing the reaction to proceed at much milder temperatures and pressures compared to traditional methods, thereby reducing energy costs and equipment stress.
Q: Does this method eliminate the formation of ammonium sulfate waste?
A: Yes, unlike the traditional alcoholysis of alkyl nitriles which uses sulfuric acid and generates large amounts of ammonium sulfate by-product, this amine transesterification method produces formamide or exchanged amides as by-products, avoiding the environmental burden of sulfate waste disposal.
Q: Can this process be scaled for industrial production of alpha-hydroxy esters?
A: Absolutely. The patent data demonstrates high conversion and selectivity rates using standard stainless steel reactors at moderate pressures (10-40 kg/cm²), indicating that the process is robust and suitable for commercial scale-up without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the ionic liquid-promoted amine transesterification technology in reshaping the production of critical chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a practical, industrial setting. Our facilities are equipped with state-of-the-art pressure reactors and rigorous QC labs capable of meeting stringent purity specifications required by top-tier pharmaceutical and agrochemical clients. We are committed to leveraging this advanced chemistry to deliver high-quality organic carboxylates that meet the most demanding performance criteria.
We invite potential partners to engage with our technical team to explore how this innovative synthesis route can be tailored to your specific product portfolio. Whether you require a Customized Cost-Saving Analysis for an existing product line or need to develop a new route for a complex intermediate, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value for your organization.
