Revolutionizing 2-Hydroxyphenylboronic Acid Production via Protected Phenol Lithiation for Commercial Scale
The global demand for high-purity organoboron compounds, particularly 2-hydroxyphenylboronic acid, has surged due to their critical role as building blocks in Suzuki-Miyaura cross-coupling reactions for pharmaceutical and agrochemical synthesis. Patent CN103896974A introduces a transformative manufacturing methodology that addresses longstanding inefficiencies in producing this valuable intermediate. By shifting the synthetic starting point from expensive halogenated aromatics to commodity phenol, this technology offers a compelling value proposition for industrial scale-up. The core innovation lies in the strategic combination of hydroxyl group protection and precise low-temperature lithiation, which collectively enhance regioselectivity and overall yield. For procurement and supply chain leaders, this patent represents a viable pathway to decouple production costs from volatile halogenated raw material markets while ensuring a robust supply of essential API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
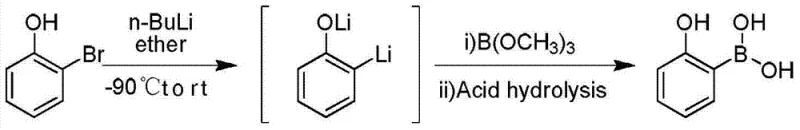
Historically, the industrial preparation of 2-hydroxyphenylboronic acid has relied heavily on routes starting from o-bromophenol or phenylboronic acid derivatives, which present significant economic and operational bottlenecks. Traditional methods often involve multi-step sequences including nitration, reduction, diazotization, and hydrolysis, resulting in lengthy operational paths that accumulate impurities and reduce overall throughput. Furthermore, the reliance on o-bromophenol as a primary feedstock imposes a substantial cost burden, as halogenated aromatics are inherently more expensive and subject to stricter environmental regulations regarding waste disposal. The use of two equivalents of n-butyllithium in older lithiation protocols not only inflates reagent costs but also increases safety risks associated with handling large quantities of pyrophoric materials. These conventional approaches frequently suffer from poor reaction selectivity, leading to complex impurity profiles that require extensive and costly purification processes to meet pharmaceutical grade standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN103896974A leverages inexpensive phenol as the foundational raw material, fundamentally altering the cost structure of the synthesis. By introducing a hydroxyl protecting group prior to lithiation, the process effectively masks the acidic proton, preventing wasteful consumption of the organolithium reagent and directing metalation exclusively to the ortho-position. This strategic modification allows for the use of stoichiometric amounts of n-butyllithium rather than the excess typically required in unprotected systems. The subsequent reaction with borate esters under controlled thermal conditions ensures high conversion rates while minimizing the formation of poly-lithiated byproducts. This streamlined approach not only shortens the synthetic route but also significantly reduces the generation of hazardous waste, aligning with modern green chemistry principles and facilitating easier regulatory approval for commercial manufacturing facilities.
Mechanistic Insights into Directed Ortho-Lithiation and Boronation
The success of this novel synthesis hinges on the precise manipulation of electronic and steric factors through hydroxyl protection. In the absence of a protecting group, the phenolic hydroxyl proton is significantly more acidic than the aromatic protons, leading to rapid deprotonation by n-butyllithium to form a phenoxide salt rather than the desired aryl lithium species. By converting the phenol into a protected intermediate, such as a silyl ether or tetrahydropyranyl ether, the acidity is neutralized, allowing the strong base to attack the aromatic ring. The coordination of the lithium cation with the oxygen atom of the protecting group can further facilitate directed ortho-metalation (DoM), enhancing the kinetic preference for substitution at the adjacent carbon. This mechanistic control is critical for achieving high regioselectivity, ensuring that the boron moiety is installed precisely at the 2-position relative to the hydroxyl group upon deprotection.
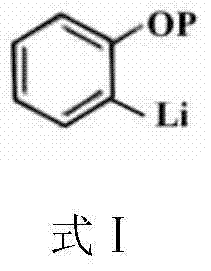
Temperature control serves as another pivotal variable in managing the reaction mechanism and impurity profile. The patent specifies maintaining the lithiation step at temperatures ranging from -70°C to -30°C, a range chosen to balance reaction kinetics with stability. At these cryogenic conditions, the kinetic energy of the molecules is reduced, which suppresses competing side reactions such as nucleophilic attack on the protecting group or metal-halogen exchange if trace halides are present. Furthermore, low temperatures stabilize the highly reactive aryl lithium intermediate, preventing decomposition or rearrangement before it can react with the borate ester. Upon warming to ambient temperatures for the final hydrolysis, the protecting group is cleaved under acidic conditions, regenerating the free phenolic hydroxyl group and yielding the target 2-hydroxyphenylboronic acid with minimal structural degradation or isomerization.
How to Synthesize 2-Hydroxyphenylboronic Acid Efficiently
The implementation of this synthesis route requires careful attention to solvent selection, reagent addition rates, and thermal management to ensure reproducibility and safety on a large scale. The process begins with the protection of phenol in a polar organic solvent, followed by the slow addition of n-butyllithium under an inert atmosphere to prevent moisture ingress. Detailed standard operating procedures regarding the specific molar ratios of protecting agents and the precise ramping of temperatures are essential for maximizing yield. For a comprehensive breakdown of the experimental parameters and workup procedures required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide provided below.
- Protect the phenolic hydroxyl group of phenol using a protecting agent like 3,4-dihydropyrane or TBDMS-Cl in an organic solvent with a catalyst.
- Perform directed ortho-lithiation by adding n-butyllithium to the protected phenol intermediate at temperatures between -70°C and -30°C.
- React the lithiated intermediate with a borate ester (e.g., triisopropyl borate) followed by acid hydrolysis to yield the final boronic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this phenol-based synthesis route offers tangible strategic benefits that extend beyond simple unit cost savings. By replacing specialized halogenated starting materials with commodity phenol, manufacturers can mitigate supply chain risks associated with the availability and price volatility of brominated intermediates. The simplified reaction sequence reduces the number of unit operations required, which directly translates to lower capital expenditure on equipment and reduced utility consumption per kilogram of product. Additionally, the improved selectivity of the protected lithiation method minimizes the formation of difficult-to-remove impurities, thereby reducing the load on downstream purification units and increasing overall plant throughput. These factors combine to create a more resilient and cost-effective supply chain capable of meeting the rigorous demands of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The substitution of o-bromophenol with phenol as the primary feedstock results in a drastic reduction in raw material expenses, as phenol is produced on a massive industrial scale with stable pricing. Furthermore, the stoichiometric efficiency gained through hydroxyl protection means less n-butyllithium is consumed per mole of product, lowering the cost of high-value reagents. The elimination of multiple synthetic steps found in traditional routes also reduces labor costs and solvent usage, contributing to a leaner manufacturing budget. Qualitative analysis suggests that these cumulative efficiencies can lead to substantial margin improvements for producers of fine chemical intermediates.
- Enhanced Supply Chain Reliability: Sourcing phenol is significantly less complex than procuring specialized halogenated aromatics, which are often subject to tighter environmental controls and limited supplier bases. This shift enhances supply security by diversifying the pool of potential raw material vendors and reducing dependency on single-source suppliers. The robustness of the reaction conditions, particularly the tolerance for standard industrial solvents like THF and toluene, ensures that production can be maintained even if specific niche reagents face temporary shortages. Consequently, lead times for high-purity pharmaceutical intermediates can be stabilized, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are readily transferable from laboratory to pilot and commercial scales without significant re-engineering. The reduction in hazardous waste generation, particularly the avoidance of heavy metal catalysts and excessive halogenated byproducts, simplifies waste treatment protocols and lowers compliance costs. This environmental advantage is increasingly critical as regulatory bodies worldwide impose stricter limits on industrial effluent, making this greener synthesis route a future-proof investment for sustainable chemical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing vendor capabilities. The answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing a reliable basis for decision-making.
Q: Why is hydroxyl protection necessary in this synthesis route?
A: Hydroxyl protection prevents the n-butyllithium from reacting with the acidic phenolic proton, ensuring selective ortho-lithiation and reducing reagent consumption.
Q: What are the cost advantages of using phenol over o-bromophenol?
A: Phenol is a commodity chemical with significantly lower market pricing compared to halogenated precursors like o-bromophenol, drastically reducing raw material costs.
Q: How does temperature control impact the purity of the final product?
A: Maintaining temperatures below -30°C during lithiation suppresses side reactions and poly-lithiation, leading to higher regioselectivity and improved product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxyphenylboronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for key pharmaceutical intermediates like 2-hydroxyphenylboronic acid. Our CDMO team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative techniques described in CN103896974A can be seamlessly integrated into your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for drug substance manufacturing. Our commitment to technical excellence allows us to deliver high-quality intermediates that support the development of life-saving medications worldwide.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this phenol-based methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency, quality, and mutual growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
