Advanced Synthesis of Minodronate Dimer: Enhancing Stability and Commercial Viability for Osteoporosis Therapeutics
Advanced Synthesis of Minodronate Dimer: Enhancing Stability and Commercial Viability for Osteoporosis Therapeutics
The pharmaceutical landscape for osteoporosis treatment is continuously evolving, driven by the need for more stable and efficacious bisphosphonate derivatives. Patent CN111233933A introduces a groundbreaking synthetic methodology for Minodronate Dimer, addressing critical stability issues inherent in previous monomeric forms. This technical disclosure outlines a robust, multi-step process that leverages silylated phosphite intermediates and mild condensation conditions to achieve high yields and exceptional product purity. For R&D directors and procurement strategists, this innovation represents a significant opportunity to optimize the supply chain for next-generation bone resorption inhibitors. The method not only simplifies the operational complexity but also ensures the final active pharmaceutical ingredient meets stringent quality standards required for global regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Minodronic Acid has been plagued by significant technical hurdles that hinder large-scale commercial adoption. Traditional routes often rely on the preparation of the minodronic acid monomer or its hydrate forms, which suffer from inherent chemical instability and poor storage characteristics. These conventional processes frequently necessitate harsh reaction conditions, including extreme temperatures and aggressive reagents, which inevitably lead to a proliferation of side reactions and complex impurity profiles. Furthermore, the low product yields associated with these older methodologies result in substantial material waste and inflated production costs, making them economically unviable for mass manufacturing. The inability to consistently produce high-purity material without extensive and costly purification steps has long been a bottleneck for suppliers aiming to meet the growing global demand for potent osteoporosis medications.
The Novel Approach
In stark contrast to legacy methods, the novel approach detailed in the patent utilizes a strategic dimerization pathway that fundamentally enhances molecular stability. By synthesizing a specialized intermediate reactant C (methyl bis(trimethylsilyl) phosphite) and employing it in a controlled coupling reaction, the process avoids the degradation pathways common to monomeric bisphosphonates. The reaction conditions are remarkably mild, with key steps proceeding efficiently at temperatures ranging from 25°C to 35°C, thereby preserving the integrity of the sensitive imidazopyridine moiety. This methodological shift not only streamlines the synthetic route by reducing the number of isolation steps but also significantly boosts the overall yield, making it highly suitable for industrial scale-up. The resulting Minodronate Dimer demonstrates superior physicochemical properties, ensuring better patient compliance and therapeutic outcomes.
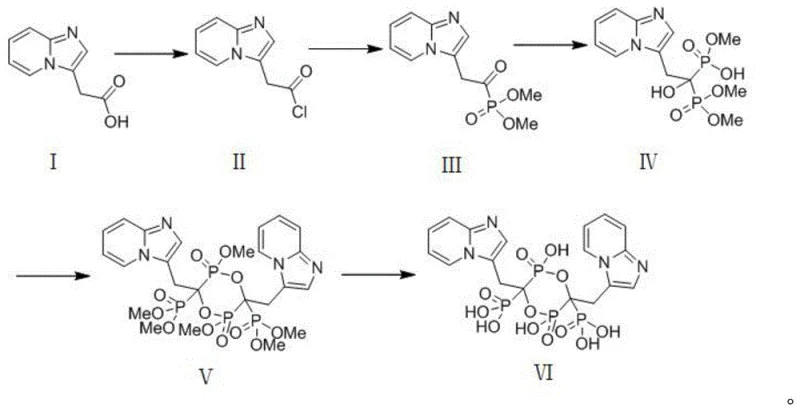
Mechanistic Insights into Silylated Phosphite Coupling and Condensation
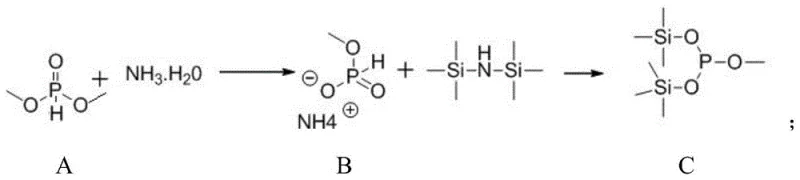
The core of this synthetic innovation lies in the meticulous generation and utilization of the silylated phosphite intermediate. The process begins with the conversion of dimethyl phosphite into ammonium methyl phosphate using ammonia water under ice-water bath conditions, followed by a crucial silylation step with hexamethyldisilazane under nitrogen protection. This generates the highly nucleophilic methyl bis(trimethylsilyl) phosphite (Intermediate C), which is essential for the subsequent Abramov-type reaction. The use of hexamethyldisilazane acts as both a silylating agent and a scavenger for byproducts, driving the equilibrium towards the desired phosphite species. This specific activation strategy allows for the efficient formation of the carbon-phosphorus bond under mild conditions, avoiding the violent exotherms often seen with traditional phosphite reagents.
Following the formation of the alpha-keto phosphonate backbone, the process employs a sophisticated peptide-coupling strategy to achieve dimerization. The use of EDCI (1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride) in conjunction with HOBt (1-hydroxybenzotriazole) and DMAP (4-dimethylaminopyridine) facilitates the formation of the ester linkage between two monomeric units with high fidelity. This catalytic system is particularly effective at suppressing racemization and minimizing the formation of N-acylurea byproducts, which are common pitfalls in carbodiimide-mediated couplings. The final hydrolysis step using an alkaline substance like lithium hydroxide at controlled temperatures (30-35°C) ensures the gentle removal of protecting groups without compromising the P-C-P backbone. This precise control over reaction kinetics and thermodynamics is what enables the production of Minodronate Dimer with an impurity profile that far exceeds industry standards.
How to Synthesize Minodronate Dimer Efficiently
Implementing this synthesis requires strict adherence to the specified stoichiometry and environmental controls to maximize efficiency. The process is divided into two primary stages: the preparation of the reactive silylated phosphite and the assembly of the dimeric framework. Operators must ensure anhydrous conditions during the silylation phase to prevent premature hydrolysis of the silicon reagents. Detailed standard operating procedures regarding temperature ramping and reagent addition rates are critical for reproducibility. For a comprehensive breakdown of the specific experimental parameters, reagent grades, and workup procedures, please refer to the standardized synthesis guide below.
- Prepare Intermediate C by reacting dimethyl phosphite with ammonia water followed by silylation with hexamethyldisilazane under nitrogen protection.
- Convert 2-(imidazo[1,2-a]pyridin-3-yl)acetic acid to its acyl chloride using thionyl chloride, then react with trimethyl phosphite to form the alpha-keto phosphonate intermediate.
- Couple the alpha-keto phosphonate with Intermediate C, followed by EDCI-mediated dimerization and final alkaline hydrolysis to yield the pure Minodronate Dimer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis offers transformative benefits for procurement managers and supply chain directors seeking to optimize their API sourcing strategies. The shift towards a dimeric structure inherently solves stability issues that often lead to product spoilage and inventory write-offs during long-term storage and transportation. By adopting a route that operates under mild thermal conditions, manufacturing facilities can significantly reduce energy consumption and lower the burden on cooling infrastructure, translating directly into operational expenditure savings. Moreover, the high yield and reduced side reactions mean that less raw material is required to produce the same amount of active ingredient, effectively lowering the cost of goods sold. This efficiency makes the supply chain more resilient against fluctuations in raw material pricing and availability.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the reduction in side products drastically simplify the downstream purification process. Traditional methods often require multiple recrystallizations or complex chromatographic separations to remove stubborn impurities, which are both time-consuming and expensive. By achieving high crude purity through controlled synthesis, the need for extensive purification is minimized, leading to substantial cost savings in solvent usage and labor. Furthermore, the use of readily available reagents like hexamethyldisilazane and standard coupling agents ensures that material costs remain competitive without relying on exotic or proprietary catalysts.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to downstream formulation partners. The stability of the Minodronate Dimer intermediate allows for flexible inventory management, reducing the risk of stockouts caused by product degradation. Additionally, the scalability of the process means that suppliers can rapidly ramp up production volumes to meet surges in market demand without requiring significant capital investment in new reactor types or safety systems. This reliability fosters stronger partnerships between chemical manufacturers and pharmaceutical developers.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions aligns perfectly with modern green chemistry principles, reducing the generation of hazardous waste streams. Lower operating temperatures and the avoidance of toxic heavy metal catalysts simplify wastewater treatment and废气 handling, ensuring compliance with increasingly stringent environmental regulations. The process is designed for seamless translation from laboratory bench scale to multi-ton commercial production, utilizing standard glass-lined or stainless steel reactors. This ease of scale-up reduces the time-to-market for new generic formulations and ensures a sustainable manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Minodronate Dimer. These insights are derived directly from the patent specifications and are intended to clarify the advantages of this specific synthetic pathway for industry stakeholders. Understanding these nuances is essential for making informed decisions about technology transfer and vendor qualification.
Q: Why is the Minodronate Dimer preferred over the monomer for pharmaceutical applications?
A: The Minodronate Dimer exhibits significantly superior stability compared to the monomer or hydrate forms, which are prone to degradation. This enhanced stability ensures a longer shelf life and consistent therapeutic efficacy in treating osteoporosis and hypercalcemia.
Q: What are the critical reaction conditions for the silylation step in this synthesis?
A: The silylation step requires strict anhydrous conditions under nitrogen protection. The reaction between ammonium methyl phosphate and hexamethyldisilazane typically involves reflux conditions to ensure complete conversion to the reactive methyl bis(trimethylsilyl) phosphite intermediate.
Q: How does this patented method improve impurity control compared to traditional routes?
A: By utilizing mild reaction temperatures (25-35°C) for the critical phosphonate coupling and condensation steps, the method minimizes thermal decomposition and side reactions. Additionally, the use of specific coupling agents like EDCI and HOBt allows for precise control over the dimerization process, resulting in a cleaner impurity profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Minodronate Dimer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stability and purity in the development of advanced osteoporosis therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot plant to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of bisphosphonate chemistry, including rigorous moisture control and specialized filtration systems. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Minodronate Dimer meets the highest international pharmacopeial standards, providing you with a secure and compliant supply foundation.
We invite you to collaborate with us to leverage this innovative synthesis for your next-generation drug candidates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development pipeline while optimizing your overall production costs.
