Advanced Manufacturing of Trabectedin Intermediate NT02a via Safe Sulfonyl Protection Strategy
Introduction to Advanced Trabectedin Intermediate Synthesis
The pharmaceutical landscape for oncology treatments continues to evolve with the demand for complex marine-derived natural products like Trabectedin (Yondelis). As a potent agent for treating advanced soft tissue sarcoma and ovarian cancer, the consistent supply of its key intermediates is paramount for global health security. Patent CN112745327A introduces a groundbreaking preparation method for the critical intermediate compound NT02a, addressing long-standing challenges in stability and yield. This technical breakthrough shifts the paradigm from hazardous diazotization protocols to a robust sulfonyl-protection strategy, ensuring a safer and more efficient supply chain for this high-value active pharmaceutical ingredient (API) precursor. The structural complexity of NT02a requires precise stereochemical control, which this new methodology delivers with exceptional fidelity.
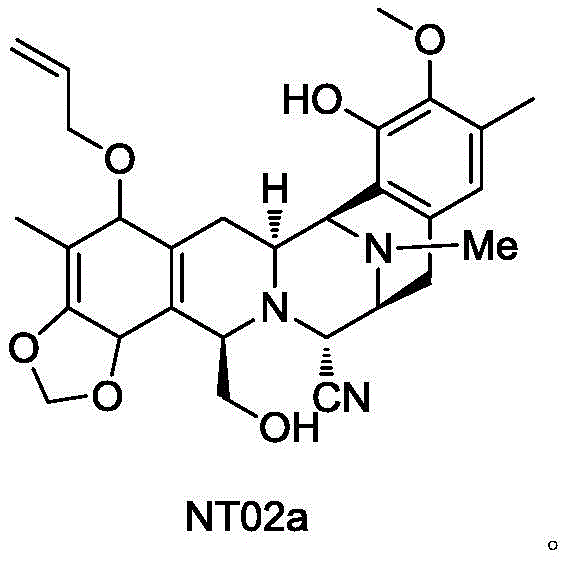
By leveraging this innovative synthetic route, manufacturers can overcome the bottlenecks that have historically constrained the availability of Trabectedin. The method described in the patent not only enhances the purity profile of the final product, achieving HPLC purity levels of 99.3%, but also fundamentally alters the risk profile of the manufacturing process. For stakeholders in the fine chemical and pharmaceutical sectors, understanding the nuances of this transformation is essential for evaluating potential partnerships and securing reliable sources of high-purity pharmaceutical intermediates. The transition to this safer chemistry represents a significant maturation in the industrial production of marine drug derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of compound NT02a from its precursor NT01a relied heavily on diazotization reactions, typically employing aqueous sodium nitrite and acetic acid. This traditional pathway, as disclosed in earlier literature such as WO2003066638, presents severe safety liabilities due to the formation of diazonium salt intermediates. These species are notoriously unstable and possess a high potential for explosive decomposition, creating unacceptable risks for large-scale industrial operations. Furthermore, the conventional process suffers from mediocre efficiency, with reported yields hovering around 46% to 50%, necessitating extensive purification via flash column chromatography which increases solvent waste and processing time. The operational complexity of extracting, drying, and concentrating crude products from such hazardous reactions adds layers of cost and regulatory burden that are increasingly difficult to justify in modern GMP environments.
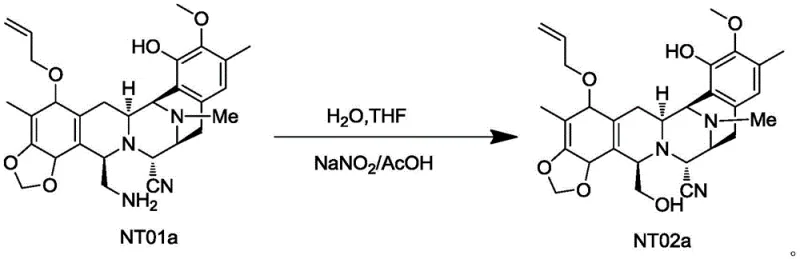
The Novel Approach
In stark contrast to the perilous legacy methods, the novel approach outlined in CN112745327A employs a sophisticated three-step sequence that completely circumvents the formation of explosive diazonium species. The process initiates with the strategic protection of the amine functionality in NT01a using a sulfonyl compound, such as methanesulfonyl chloride (MsCl), under mild basic conditions. This protection step is crucial as it stabilizes the molecule for subsequent oxidative transformations that would otherwise be incompatible with a free amine. The subsequent conversion involves a highly efficient one-pot oxidation and hydrolysis sequence, followed by a gentle reduction step. This modular design allows for precise control over reaction parameters, minimizing side reactions and impurity generation while maximizing throughput. The result is a streamlined workflow that transforms a dangerous, low-yielding batch process into a safe, high-efficiency manufacturing protocol suitable for commercial scale-up.
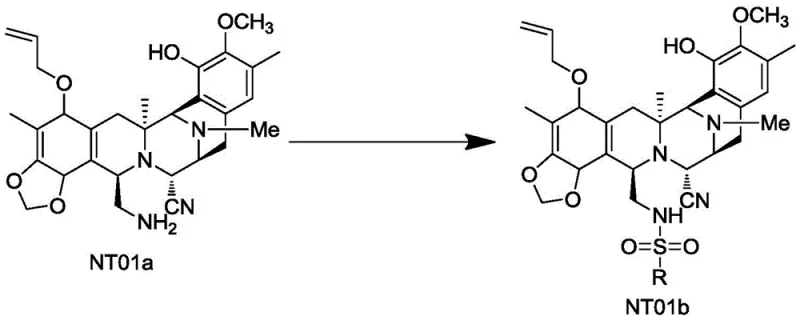
Mechanistic Insights into Sulfonyl-Protection and Oxidative Transformation
The core innovation of this synthesis lies in the mechanistic elegance of the sulfonyl protection strategy coupled with the specific oxidative conditions employed. In the first stage, the reaction of NT01a with MsCl in the presence of a base like triethylamine converts the reactive primary amine into a sulfonamide (NT01b). This modification effectively masks the nucleophilicity of the nitrogen atom, preventing unwanted side reactions during the harsh oxidative conditions required for the next step. The choice of base and the molar ratio are critical; maintaining a slight excess of base ensures complete conversion without promoting degradation of the sensitive molecular scaffold. This protection-deprotection logic is a hallmark of advanced organic synthesis, allowing chemists to perform transformations on one part of a complex molecule while leaving other functional groups intact.
Following protection, the conversion of NT01b to the aldehyde intermediate NT01c utilizes N-tert-butyl thiocyanato chloride as a specialized oxidant in the presence of DBU (1,8-Diazabicyclo[5.4.0]undec-7-ene). This reaction is conducted under a nitrogen atmosphere at cryogenic temperatures (-78°C) to ensure selectivity and prevent over-oxidation or thermal degradation. The mechanism likely involves the activation of the sulfonamide followed by oxidative cleavage or transformation to the aldehyde functionality. Crucially, this oxidation is telescoped directly into a hydrolysis step within the same vessel. By adding dilute hydrochloric acid after the oxidation is complete, the sulfonyl protecting group is removed, and the molecule rearranges to form NT01c. This one-pot design minimizes handling losses and significantly reduces the volume of organic solvents required, aligning with green chemistry principles while maintaining high stereochemical integrity throughout the transformation.
How to Synthesize NT02a Efficiently
The practical execution of this synthesis requires careful attention to temperature control and reagent stoichiometry to replicate the high yields reported in the patent data. The process begins with the dissolution of NT01a in a suitable solvent like dichloromethane, followed by cooling and the controlled addition of the sulfonylating agent. Once the protected intermediate NT01b is secured, it is subjected to the oxidative conditions using the thiocyanato chloride reagent under inert gas. The final reduction step utilizes sodium borohydride to convert the aldehyde NT01c into the target alcohol NT02a. Each stage is designed to be robust, with the patent examples demonstrating that crude products from early steps can often be used directly in subsequent reactions without rigorous purification, further enhancing the overall process economy. Detailed standardized operating procedures for scaling this route from laboratory to production are essential for maintaining quality.
- React compound NT01a with a sulfonyl compound (e.g., MsCl) under basic conditions to form the protected intermediate NT01b.
- Perform a one-pot oxidation and hydrolysis on NT01b using N-tert-butyl thiocyanato chloride and DBU to generate compound NT01c.
- Reduce compound NT01c using sodium borohydride (NaBH4) to obtain the final target intermediate NT02a with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and logistical advantages that extend beyond simple yield metrics. The elimination of diazotization chemistry removes a major safety bottleneck, thereby reducing the insurance premiums and specialized infrastructure costs associated with handling explosive intermediates. This shift allows for manufacturing in a broader range of facilities, increasing the potential supply base and reducing geopolitical supply risks. Furthermore, the significant improvement in total yield means that less raw material is required to produce the same amount of final API, directly translating to substantial cost savings in raw material procurement. The ability to perform the oxidation and hydrolysis in a single pot reduces the number of unit operations, shortening the production cycle time and increasing the throughput capacity of existing manufacturing assets without the need for capital-intensive expansion.
- Cost Reduction in Manufacturing: The transition to a non-explosive pathway eliminates the need for expensive blast-proof reactors and specialized safety containment systems, leading to significant capital and operational expenditure reductions. Additionally, the high yields observed in each step (over 96% for the first two steps) minimize the waste of expensive starting materials and reduce the cost of goods sold (COGS). The reduction in solvent usage due to the one-pot methodology further decreases waste disposal costs and environmental compliance burdens, creating a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: By removing the reliance on hazardous diazonium chemistry, the supply chain becomes more resilient to regulatory shutdowns and safety incidents that frequently disrupt production of high-risk intermediates. The robustness of the sulfonyl protection method ensures consistent batch-to-batch quality, reducing the likelihood of failed batches and supply shortages. This reliability is critical for pharmaceutical companies managing tight inventory levels for orphan drugs like Trabectedin, where continuity of supply is a matter of patient access and regulatory compliance.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of multi-gram examples in the patent without loss of efficiency. The use of standard reagents like MsCl and NaBH4 ensures easy sourcing, while the reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations. This eco-friendly profile facilitates smoother regulatory approvals and supports the sustainability goals of modern pharmaceutical enterprises, making it an attractive option for long-term strategic sourcing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. Understanding these details helps stakeholders assess the feasibility of integrating this technology into their existing supply chains. The answers are derived directly from the experimental data and technical disclosures within patent CN112745327A, providing a factual basis for decision-making. These insights clarify the operational benefits and safety improvements inherent in the new route compared to legacy methods.
Q: Why is the new synthesis method for NT02a safer than previous methods?
A: The novel method avoids the use of diazotization reactions involving sodium nitrite and acetic acid, which generate unstable and potentially explosive diazonium salt intermediates. By utilizing a sulfonyl protection strategy followed by controlled oxidation, the process eliminates significant safety hazards associated with industrial scale-up.
Q: What represents the key yield improvement in this patent compared to prior art?
A: While conventional methods reported yields around 46% to 50% with complex purification steps, the new three-step route achieves individual step yields of approximately 97.8%, 96.5%, and 88%. This cumulative efficiency results in a substantially higher total yield and simplified downstream processing.
Q: How does the one-pot oxidation-hydrolysis step benefit manufacturing?
A: The conversion of NT01b to NT01c is performed in a single reactor vessel without isolating the oxidation intermediate. This telescoping of steps reduces solvent consumption, minimizes material loss during transfer, and shortens the overall production cycle time, enhancing operational efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NT02a Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of complex intermediates like NT02a for the global oncology market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our state-of-the-art facilities are equipped to handle the specific requirements of this sulfonyl-protection chemistry, including cryogenic reactions and inert atmosphere processing, guaranteeing stringent purity specifications and rigorous QC labs oversight for every batch produced. We are committed to delivering the reliability and technical excellence required to support the development and commercialization of life-saving marine-derived therapeutics.
We invite you to engage with our technical team to explore how this advanced synthesis route can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact our technical procurement team to索取 specific COA data and route feasibility assessments, ensuring that your transition to this superior manufacturing method is seamless and scientifically sound. Let us collaborate to bring safer and more effective treatments to patients worldwide.
