Advanced Synthesis of Fucosylated Chondroitin Sulfate Oligosaccharide Clusters for Anticoagulant Drug Development
The pharmaceutical industry is constantly seeking more reliable sources for complex carbohydrate intermediates, particularly those with potent biological activities like anticoagulation. Patent CN108794653B introduces a groundbreaking method for synthesizing fucosylated chondroitin sulfate oligosaccharide clusters, addressing the critical limitations of natural extraction. This technology utilizes chondroitin sulfate A salt as a cost-effective raw material, transforming it through a series of precise chemical modifications including acidolysis, protecting group manipulation, and glycosylation. The ultimate goal is to create multivalent glycoclusters with a general formula Q structure, which simulate the activity of natural glycosaminoglycans while offering superior structural certainty. 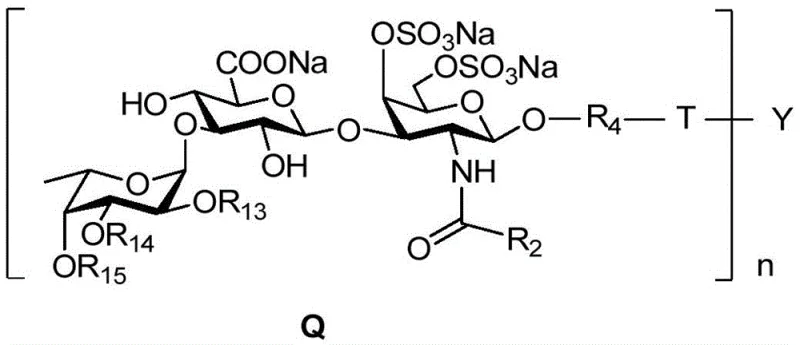 . For procurement managers and R&D directors alike, this represents a significant shift towards high-purity pharmaceutical intermediates that can be consistently manufactured, ensuring a stable supply chain for next-generation anticoagulant therapies.
. For procurement managers and R&D directors alike, this represents a significant shift towards high-purity pharmaceutical intermediates that can be consistently manufactured, ensuring a stable supply chain for next-generation anticoagulant therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, fucosylated chondroitin sulfate has been obtained directly from sea cucumbers or through degradation of natural polysaccharides. However, these conventional methods suffer from inherent microscopic inhomogeneity, meaning the resulting oligosaccharides vary in structure and sulfation patterns from batch to batch. This variability poses severe challenges for quality control and regulatory approval, as it is difficult to establish clear structure-activity relationships. Furthermore, natural extracts often carry the risk of contamination and have been associated with side effects such as thrombocytopenia. The reliance on biological sources also introduces supply chain vulnerabilities, where availability fluctuates with environmental factors and harvesting conditions. Consequently, the translation of these natural products into clinically viable drugs has been significantly hindered by the inability to guarantee purity and consistent biological performance across large-scale production runs.
The Novel Approach
The novel synthetic approach detailed in the patent overcomes these hurdles by constructing the oligosaccharide clusters chemically from well-defined building blocks. By starting with chondroitin sulfate A salt, the process ensures a consistent backbone structure before introducing specific fucose branches and sulfation patterns. This method allows for the precise adjustment of the degree of sulfation and the molecular weight by changing the valence of the sugar cluster scaffold. The use of click chemistry to assemble the final multivalent structures ensures high efficiency and specificity, minimizing byproducts. This level of control enables researchers to explore the exact impact of different sulfation modes on anticoagulant activity without the noise of structural heterogeneity. For manufacturers, this translates to a robust process capable of producing high-purity fucosylated chondroitin sulfate intermediates that meet stringent regulatory standards for medicinal use.
Mechanistic Insights into Multivalent Glycocluster Assembly
The synthesis begins with the acid hydrolysis of chondroitin sulfate M salt to yield a disaccharide backbone, which is then esterified and fully acylated to protect reactive hydroxyl and amino groups. A key mechanistic step involves the formation of an oxazoline intermediate under acid catalysis, which serves as a highly reactive glycosyl donor. This intermediate reacts with an azido-alcohol linker to install the handle necessary for subsequent cluster assembly. The process continues with selective deprotection strategies to expose specific hydroxyl groups for fucosylation, ensuring the fucose branch is attached at the correct 3-position of the uronic acid. The stereochemistry is tightly controlled throughout these glycosylation steps to maintain the biological integrity of the molecule. Finally, the sulfation pattern is introduced, which is critical for anticoagulant activity, before the monomeric units are ready for clustering.
The final assembly leverages the power of Cu(I)-catalyzed click chemistry to link the functionalized trisaccharide azides to multifunctional dendritic scaffolds. 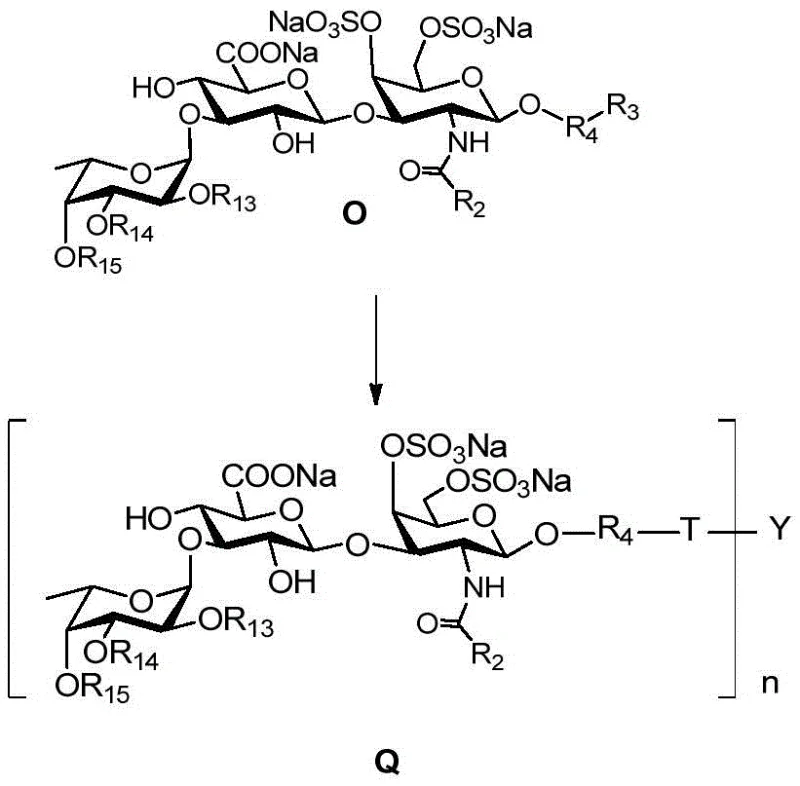 . This reaction is highly orthogonal, meaning it proceeds efficiently even in the presence of other functional groups like esters and sulfates, which simplifies the purification process significantly. The resulting glycoclusters exhibit a "glycocluster effect," where the multivalent presentation of the sugar moieties enhances binding affinity to biological targets compared to monomeric sugars. This mechanistic precision ensures that the final product not only has the correct primary structure but also the optimal spatial arrangement for therapeutic efficacy. Understanding this pathway is crucial for R&D teams aiming to replicate or scale this synthesis for commercial API intermediate production.
. This reaction is highly orthogonal, meaning it proceeds efficiently even in the presence of other functional groups like esters and sulfates, which simplifies the purification process significantly. The resulting glycoclusters exhibit a "glycocluster effect," where the multivalent presentation of the sugar moieties enhances binding affinity to biological targets compared to monomeric sugars. This mechanistic precision ensures that the final product not only has the correct primary structure but also the optimal spatial arrangement for therapeutic efficacy. Understanding this pathway is crucial for R&D teams aiming to replicate or scale this synthesis for commercial API intermediate production.
How to Synthesize Fucosylated Chondroitin Sulfate Efficiently
The synthesis protocol outlined in the patent provides a comprehensive roadmap for producing these complex molecules, starting from bulk commodity chemicals. The process is designed to be modular, allowing for the interchange of different dendritic scaffolds to produce clusters of varying valences (n=2 to 15). Detailed operational parameters, such as the use of specific Lewis acids for glycosylation and precise temperature controls for sulfation, are critical for success. The following guide summarizes the standardized synthesis steps derived from the patent data, serving as a foundational reference for process development teams looking to implement this technology. For the complete step-by-step experimental procedure and specific reagent quantities, please refer to the detailed guidelines below.
- Hydrolyze chondroitin sulfate A salt in mineral acid to obtain the disaccharide backbone, followed by esterification and acylation protection.
- Convert the protected disaccharide into an oxazoline intermediate and react with an azido-alcohol linker to introduce the click chemistry handle.
- Perform selective deprotection, fucosylation, and sulfation to generate the functionalized trisaccharide azide, then assemble into clusters via Cu(I)-catalyzed click chemistry.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthetic route offers profound advantages for supply chain stability and cost management in the production of anticoagulant intermediates. By shifting away from unpredictable natural extraction, manufacturers can secure a consistent supply of raw materials that are not subject to seasonal or ecological variations. The use of chondroitin sulfate A salt as a starting point leverages an abundant and inexpensive feedstock, significantly lowering the entry barrier for production. Furthermore, the chemical synthesis allows for the elimination of complex purification steps often required to remove biological impurities from natural extracts, streamlining the manufacturing workflow. This efficiency gain directly contributes to cost reduction in anticoagulant manufacturing, making the final therapeutic candidates more economically viable.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive and variable natural sourcing, replacing it with scalable chemical processes. By utilizing robust reactions like acid hydrolysis and click chemistry, the process minimizes waste and maximizes yield consistency. The removal of transition metal catalysts in certain steps or the use of recoverable reagents further optimizes the cost structure. Additionally, the ability to produce specific sulfation patterns on demand reduces the need for extensive fractionation, leading to substantial cost savings in downstream processing and quality control operations.
- Enhanced Supply Chain Reliability: Relying on chemical synthesis decouples production from the volatility of marine resource harvesting. This ensures a continuous and predictable supply of high-purity intermediates, which is critical for maintaining drug development timelines. The modular nature of the synthesis allows for rapid scaling from laboratory to commercial quantities without re-engineering the core process. This reliability mitigates the risk of supply disruptions, providing procurement managers with greater confidence in long-term planning and inventory management for critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard organic synthesis techniques that are easily transferred to large-scale reactors. The waste streams generated are primarily chemical solvents and salts, which are easier to treat and manage compared to the complex biological waste from extraction methods. This simplifies compliance with environmental regulations and reduces the burden on waste treatment facilities. The efficient atom economy of the click chemistry step also aligns with green chemistry principles, enhancing the overall sustainability profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these fucosylated chondroitin sulfate clusters. The answers are derived directly from the technical specifications and beneficial effects described in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility and value proposition of integrating this technology into their existing pipelines. For further technical discussions or specific data requests, our team is available to provide deeper insights.
Q: Why is synthetic fucosylated chondroitin sulfate preferred over natural extraction?
A: Natural extraction yields heterogeneous mixtures with micro-inhomogeneity and potential contamination risks, making quality control difficult. Synthetic methods provide defined structures and high purity essential for clinical applications.
Q: What is the significance of the glycocluster effect in this technology?
A: Assembling oligosaccharides into multivalent clusters mimics natural glycosaminoglycan activity more effectively. It allows for the adjustment of molecular weight and sulfation patterns to optimize anticoagulant activity while reducing side effects.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes inexpensive chondroitin sulfate A salt as a starting material and employs robust chemical transformations like acid hydrolysis and click chemistry, which are amenable to large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fucosylated Chondroitin Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this synthetic technology in advancing anticoagulant drug discovery. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop to market. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of fucosylated chondroitin sulfate intermediates meets the highest international standards. We understand the complexities of carbohydrate chemistry and are equipped to handle the precise protection and sulfation steps required for this synthesis.
We invite you to collaborate with us to leverage this innovative platform for your next-generation therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your development timeline while optimizing your budget. Let us be your partner in turning this complex chemistry into a commercial reality.
