Advanced Solid Acid Catalysis for High-Purity Indane Diamine Manufacturing
Advanced Solid Acid Catalysis for High-Purity Indane Diamine Manufacturing
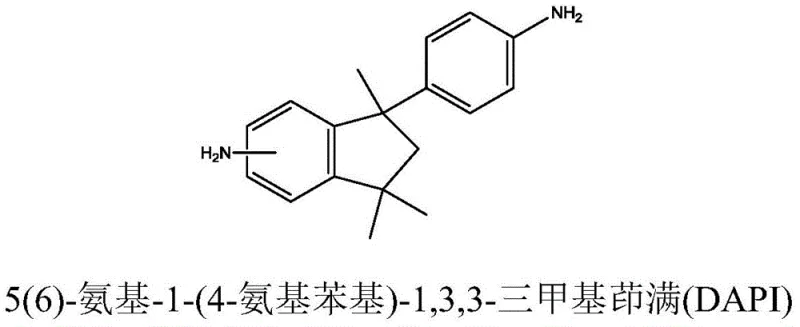
The chemical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing high-performance polymer monomers, and patent CN111484412A presents a groundbreaking methodology for producing compounds containing an indane structure. This patent specifically details the synthesis of 5(6)-amino-1-(4-aminophenyl)-1,3,3-trimethylindane, commonly known as DAPI, which serves as a critical diamine monomer for specialized polyimides and an exceptional curing agent for epoxy resins. The structural integrity of the indane ring within this molecule imparts superior thermal stability, oxidation resistance, and electrical properties, making it highly desirable for advanced material applications where performance under stress is paramount. Furthermore, the presence of methyl groups in the structure induces a dipole moment in the benzene ring, significantly enhancing compatibility with various high polymer matrices. By leveraging a novel solid acid catalytic system, this technology overcomes the severe limitations of traditional mixed-acid nitration, offering a scalable solution for the production of high-purity intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
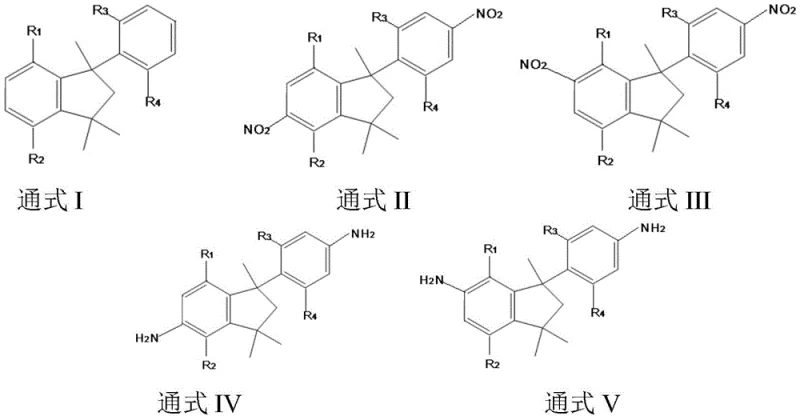
Historically, the preparation of indane-structured compounds like DAPI has relied heavily on starting materials such as 1-phenyl-1,3,3-trimethylindane, subjected to nitration via the traditional nitro-sulfur method followed by catalytic reduction. This conventional homogeneous reaction pathway, while capable of achieving relatively high conversion rates, is plagued by significant operational and environmental drawbacks that hinder modern manufacturing efficiency. The primary issue lies in the extensive use of mixed acid, where concentrated sulfuric acid gradually dilutes due to water generation during the reaction, leading to severe equipment corrosion and drastically increased maintenance costs for reactor vessels and piping systems. Moreover, the post-reaction processing involves complicated separation steps, including extraction and waste acid washing, to handle the large volume of spent mixed acid, which creates substantial pollution burdens and escalates production costs due to waste treatment requirements. Additionally, the conventional nitro-sulfur method lacks selectivity, typically yielding a mixture of 5-nitro and 6-nitro isomers in a roughly 45:55 ratio, necessitating difficult and costly purification processes to isolate the desired product specifications.
The Novel Approach
In stark contrast to the corrosive and wasteful traditional methods, the novel approach disclosed in the patent utilizes a heterogeneous solid acid catalyst to facilitate the nitration reaction, fundamentally transforming the process economics and environmental footprint. By employing catalysts such as super strong solid acids, sulfonic acid resins, or mesoporous molecular sieves, the reaction proceeds under milder conditions with enhanced selectivity, effectively limiting the free rotation of the substrate within the catalyst's mesoporous channels. This unique catalytic environment not only improves the positional selectivity of the nitro group but also simplifies the post-treatment workflow, as the solid catalyst can be easily separated via filtration and regenerated for reuse, eliminating the generation of large quantities of acidic waste liquid. The subsequent reduction step utilizes a palladium on carbon catalyst under controlled hydrogen pressure, ensuring high conversion rates and yielding the final white solid powder products with exceptional purity levels suitable for high-end electronic and aerospace applications.
Mechanistic Insights into Solid Acid-Catalyzed Nitration and Reduction
The core innovation of this synthesis route lies in the mechanistic behavior of the solid acid catalyst during the nitration phase, which differs significantly from the free-proton mechanism observed in liquid mixed acids. The solid acid catalysts, particularly the super strong varieties like SO4 2-/TiO2-ZrO2 or mesoporous molecular sieves like MCM-41, possess specific pore structures that impose steric constraints on the indane substrate molecules. This confinement effect restricts the free rotation of the 1-phenyl-1,3,3-trimethylindane molecule, thereby directing the electrophilic attack of the nitronium ion to specific positions on the aromatic ring with greater precision than is possible in a homogeneous solution. This shape-selective catalysis results in a more favorable ratio of the desired nitro-isomers, reducing the burden on downstream purification units and increasing the overall atom economy of the process. Furthermore, the solid nature of the catalyst allows for a clear phase separation between the catalyst and the reaction medium, preventing the catalyst deactivation issues often associated with homogeneous acid systems.
Following the nitration, the reduction mechanism employs a palladium on carbon (Pd/C) catalyst under a hydrogen atmosphere, which facilitates the transfer of hydrogen atoms to the nitro groups to form the corresponding amines. The reaction is conducted at temperatures ranging from 40°C to 120°C and hydrogen pressures between 1.0 MPa and 3.0 MPa, conditions that are optimized to maximize the reduction conversion rate to approximately 97% while minimizing side reactions. The use of Pd/C ensures that the reduction proceeds cleanly to the diamine stage without affecting the sensitive indane ring structure, preserving the thermal and mechanical properties inherent to the core scaffold. Impurity control is further enhanced by the initial high purity of the nitro-intermediates obtained from the selective solid acid nitration, ensuring that the final DAPI product meets stringent quality standards with purity levels exceeding 99%, which is critical for its application in high-performance polyimide resins.
How to Synthesize 5(6)-amino-1-(4-aminophenyl)-1,3,3-trimethylindane Efficiently
The synthesis of this high-value diamine involves a streamlined two-step process that begins with the precise nitration of the indane precursor using a selected solid acid catalyst, followed by a catalytic hydrogenation step to reduce the nitro groups to amines. The process is designed to be robust and scalable, utilizing standard chemical engineering unit operations such as jacketed reactors, filtration units, and rotary evaporators, making it highly adaptable for commercial scale-up of complex aromatic diamines. Operators must carefully control the滴加 rate of the molten indane原料 into the catalyst-acid mixture and maintain strict temperature profiles to ensure optimal selectivity and yield throughout the reaction course. For a detailed breakdown of the specific operational parameters, reagent ratios, and safety protocols required to execute this synthesis successfully, please refer to the standardized guide below.
- Perform nitration reaction on indane compounds using a solid acid catalyst (e.g., super strong solid acid or mesoporous molecular sieve) at 40-110°C to obtain nitro-intermediates.
- Separate the solid catalyst via filtration and purify the nitro-intermediates through solvent extraction and recrystallization.
- Conduct reduction reaction on the nitro-intermediates using a palladium on carbon catalyst under hydrogen pressure (1.0-3.0 MPa) to yield the final amino compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid acid catalytic route offers transformative advantages that directly address the pain points of cost volatility and supply continuity in the specialty chemicals sector. By shifting away from the consumption of large volumes of corrosive mixed acids, manufacturers can significantly reduce the capital expenditure associated with corrosion-resistant equipment and the operational expenditure linked to hazardous waste disposal and neutralization. The ability to recover and reuse the solid acid catalyst multiple times without significant loss of activity creates a closed-loop material flow that drastically lowers the raw material cost per kilogram of finished product, providing a competitive edge in pricing strategies for high-purity epoxy curing agents. Furthermore, the simplified workup procedure, which replaces complex extraction and washing steps with straightforward filtration, shortens the overall production cycle time, thereby enhancing the responsiveness of the supply chain to fluctuating market demands.
- Cost Reduction in Manufacturing: The elimination of sulfuric acid from the nitration process removes the need for expensive corrosion-resistant alloys in reactor construction and significantly lowers the cost of waste acid treatment, leading to substantial operational savings. Since the solid acid catalyst can be filtered, dried, and reactivated for repeated use, the recurring cost of catalyst consumption is minimized compared to the single-use nature of homogeneous mineral acids. Additionally, the higher selectivity of the reaction reduces the formation of unwanted isomers, which decreases the solvent and energy consumption required for purification, further driving down the total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: Utilizing stable solid acid catalysts reduces the dependency on the volatile supply chains associated with concentrated mineral acids and mitigates the risks of production stoppages due to equipment failure from corrosion. The robustness of the heterogeneous catalytic system ensures consistent batch-to-batch quality and yield, allowing for more accurate forecasting and inventory management of critical intermediates like DAPI. This reliability is crucial for maintaining uninterrupted production schedules for downstream customers in the electronics and aerospace industries who depend on timely delivery of high-performance polymer materials.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods, simplifying compliance with increasingly stringent environmental regulations and reducing the administrative burden of waste reporting and disposal. The straightforward scale-up potential of fixed-bed or slurry reactor systems using solid catalysts allows for seamless transition from pilot plant to full commercial production without the need for major process redesign. This scalability ensures that suppliers can rapidly increase output to meet surging demand for advanced polyimide monomers while maintaining a sustainable and eco-friendly manufacturing profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of indane-structured compounds, derived directly from the experimental data and beneficial effects described in the patent literature. These insights are intended to clarify the operational feasibility and quality advantages of the solid acid catalytic method for stakeholders evaluating this technology for integration into their supply chains. Understanding these specifics is essential for assessing the potential impact on product quality, production efficiency, and overall project viability in the context of modern chemical manufacturing.
Q: How does the solid acid catalyst improve regioselectivity compared to mixed acid?
A: The mesoporous channels of the solid acid catalyst limit the free rotation of the raw material indane compound, thereby imposing steric constraints that favor the formation of specific nitro-isomers over others, leading to improved selectivity.
Q: Can the solid acid catalyst be reused in this synthesis process?
A: Yes, the solid acid catalyst obtained after filtration can be repeatedly utilized after being cleaned, dried, and simply activated, maintaining high conversion efficiency even after multiple cycles.
Q: What purity levels can be achieved with this novel synthesis method?
A: The method allows for the production of compounds with a purity of approximately 99% or higher, specifically achieving yields of about 85% in the nitration step and 80% in the reduction step with minimal impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5(6)-amino-1-(4-aminophenyl)-1,3,3-trimethylindane Supplier
As the global demand for high-performance thermosetting resins continues to grow, the ability to source high-purity indane diamines from a technically proficient partner becomes a strategic imperative for success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality and volume. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of DAPI or related intermediate meets the exacting standards required for advanced epoxy curing and polyimide synthesis applications. We understand that the transition to new synthetic routes requires confidence in both the chemistry and the supply capability, which is why we prioritize transparency and technical support in every engagement.
We invite you to collaborate with our technical procurement team to explore how this innovative solid acid catalytic technology can optimize your material costs and enhance your product performance. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your production volume and application needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the development of next-generation polymer materials.
