Revolutionizing Gemifloxacin Production: A Safer, Scalable Route for Global API Manufacturers
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for the production of fourth-generation quinolone antibiotics, specifically targeting the synthesis of the Gemifloxacin main ring compound. Patent CN101899044B, published in July 2012, introduces a transformative methodology that addresses critical safety and efficiency bottlenecks inherent in traditional manufacturing. This technical insight report analyzes the novel approach of utilizing 2,6-dichloro-5-fluoronicotinic acid as a primary starting material, reacting it with sodium formylacetate ethyl ester under controlled CO2 pressure. Unlike legacy processes that depend on hazardous reagents, this invention leverages a streamlined two-step sequence involving acylation-amination followed by cyclization-hydrolysis. For R&D directors and procurement strategists, understanding this shift is vital, as it represents a move towards higher atom economy and reduced regulatory burden in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the Gemifloxacin core has been plagued by the use of chemically aggressive and environmentally unfriendly reagents. As illustrated in prior art methodologies, such as the route depicted in [图 1], manufacturers were forced to rely on triethyl orthoformate, a substance known for its facile hydrolysis and toxicological profile, which complicates waste treatment and poses significant environmental compliance challenges. Furthermore, alternative pathways often necessitated the use of sodium hydride (NaH), a pyrophoric solid that presents severe safety hazards during scale-up, requiring specialized inert atmosphere handling and increasing capital expenditure on safety infrastructure. Other routes involving acyl chlorides, while effective, generate stoichiometric amounts of corrosive byproducts and require rigorous purification steps to remove trace halogenated impurities that can persist through to the final API, jeopardizing patient safety and batch release specifications.
The Novel Approach
The patented method fundamentally重构 s the synthetic logic by bypassing these dangerous intermediates entirely. Instead of starting with activated acyl chlorides or unstable ketones, the process begins with the stable and commercially abundant 2,6-dichloro-5-fluoronicotinic acid. By introducing CO2 under mild pressure (1-2 atm) in the presence of sodium formylacetate ethyl ester, the reaction generates a key beta-keto ester intermediate in situ without the need for external activating agents like thionyl chloride. This intermediate is then directly subjected to amination with cyclopropylamine. The elegance of this approach lies in its telescoping potential; the filtration of salt byproducts after the CO2 step provides a clean filtrate ready for the next transformation. This not only simplifies the unit operations but also drastically reduces the generation of hazardous waste streams, aligning perfectly with modern green chemistry principles and reducing the total cost of ownership for the manufacturing facility.
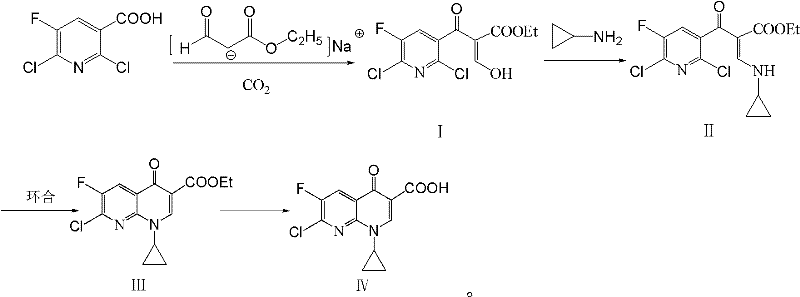
Mechanistic Insights into CO2-Mediated Acylation and Cyclization
The core innovation of this technology rests on the unique reactivity of the carboxylate anion under CO2 pressure. In the first stage, the 2,6-dichloro-5-fluoronicotinic acid reacts with the enolate of ethyl formylacetate. The introduction of CO2 likely facilitates the formation of a mixed anhydride or activates the carboxyl group towards nucleophilic attack by the enolate, leading to the formation of the beta-keto acid derivative (Compound I). This mechanism avoids the high-energy transition states associated with direct acylation using acid chlorides. Subsequently, the addition of cyclopropylamine triggers a nucleophilic substitution at the C-2 position of the pyridine ring, displacing the chlorine atom. This amination step is highly regioselective due to the electron-withdrawing nature of the adjacent carbonyl and the fluorine atom, ensuring that the cyclopropyl group installs exclusively at the desired position without affecting the C-6 chlorine, which is reserved for later functionalization in the full drug synthesis.
Following the isolation of Compound II, the cyclization step involves an intramolecular nucleophilic aromatic substitution (SNAr). In the presence of a mild base such as potassium carbonate (K2CO3) and a polar aprotic solvent like DMF or DMSO, the nitrogen of the cyclopropylamine moiety attacks the C-6 position, displacing the second chlorine atom and closing the quinolone ring system to form Compound III. This base-mediated cyclization is significantly safer than the NaH-mediated alternatives found in older patents, as K2CO3 is non-pyrophoric and easier to handle on a multi-ton scale. The final hydrolysis step cleaves the ethyl ester to yield the free carboxylic acid (Compound IV). The control of impurities here is critical; the mild conditions prevent the degradation of the sensitive fluorine substituent and minimize the formation of decarboxylated byproducts, ensuring a high-purity profile suitable for downstream coupling reactions.
How to Synthesize Gemifloxacin Main Ring Compound Efficiently
Implementing this synthesis requires precise control over reaction parameters, particularly temperature and gas pressure, to maximize yield and minimize side reactions. The process begins with the dissolution of the nicotinic acid derivative and the ester salt in a solvent like toluene or xylene, followed by cooling to 0-5°C before the introduction of CO2. Maintaining the pressure at 1-2 atm is crucial for driving the acylation to completion. Once the intermediate is formed and filtered, the amination is conducted under reflux conditions to ensure complete conversion. The subsequent cyclization requires heating to temperatures between 50°C and 125°C depending on the solvent choice, with reaction times ranging from 1 to 8 hours. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Dissolve 2,6-dichloro-5-fluoronicotinic acid and sodium formylacetate ethyl ester in an organic solvent, introduce CO2 under pressure, and filter to obtain the intermediate filtrate.
- Add cyclopropylamine to the filtrate to perform an amination reaction, yielding Compound II after separation and purification.
- Subject Compound II to cyclization using an acid binding agent like K2CO3 in a solvent like DMF or DMSO, followed by hydrolysis to obtain the final Gemifloxacin main ring compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented route offers compelling advantages that directly impact the bottom line and supply chain resilience. By shifting away from reagents like triethyl orthoformate and sodium hydride, manufacturers can eliminate the costs associated with specialized storage, hazardous waste disposal, and complex safety interlocks. The use of 2,6-dichloro-5-fluoronicotinic acid as a starting material is particularly advantageous because it is a commodity chemical with a stable global supply chain, reducing the risk of raw material shortages that often plague specialty reagent markets. Furthermore, the simplified workup procedures, which involve straightforward filtration and crystallization rather than complex chromatographic purifications, translate into shorter cycle times and higher throughput capacity for existing production assets.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous activating agents such as thionyl chloride or oxalyl chloride, which are typically required to convert the starting acid into an acyl chloride, results in substantial raw material cost savings. Additionally, replacing sodium hydride with potassium carbonate removes the need for expensive inert gas blanketing systems and reduces the insurance premiums associated with handling pyrophoric materials. The higher atom economy of this route means less waste is generated per kilogram of product, lowering the environmental levies and waste treatment costs that are increasingly significant in regulated markets.
- Enhanced Supply Chain Reliability: The reliance on stable, non-perishable starting materials ensures a consistent supply of the Gemifloxacin intermediate. Unlike methods that require freshly distilled or highly moisture-sensitive reagents which can degrade during transport or storage, the reagents in this process are robust and have long shelf lives. This stability allows procurement teams to purchase in larger bulk quantities to secure better pricing without the fear of material degradation, thereby smoothing out supply fluctuations and ensuring continuous production schedules for the final API.
- Scalability and Environmental Compliance: The process operates under mild pressure and uses common industrial solvents like toluene, DMF, and DMSO, which are easily recovered and recycled in standard distillation units. This compatibility with existing infrastructure means that scaling from pilot plant to commercial production (100 kgs to 100 MT) can be achieved with minimal capital investment in new equipment. Moreover, the avoidance of heavy metal catalysts and halogenated byproducts simplifies the environmental permitting process, facilitating faster regulatory approval for new manufacturing sites and reducing the risk of production shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the technology's fit within your current manufacturing portfolio. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: Why is this new synthesis route considered safer than conventional methods?
A: Conventional methods often rely on highly reactive and dangerous reagents such as sodium hydride (NaH) and flammable triethyl orthoformate. This patented process eliminates these hazards by utilizing stable 2,6-dichloro-5-fluoronicotinic acid and mild CO2 pressure, significantly reducing operational risks in large-scale manufacturing.
Q: What are the typical yields reported for this synthesis method?
A: According to the patent embodiments, the amination step (formation of Compound II) achieves yields around 90.2% when using toluene as a solvent. The subsequent cyclization and hydrolysis steps to form the final main ring compound (Compound IV) report yields ranging from 73.0% to 88.1%, demonstrating robust efficiency.
Q: Which solvents are compatible with this cyclization process?
A: The process offers flexibility in solvent selection for the cyclization step. Suitable organic solvents include ethanol, ethyl acetate, N,N-dimethylformamide (DMF), DMSO, acetonitrile, chloroform, carbon tetrachloride, and THF, allowing manufacturers to optimize based on cost and recovery capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemifloxacin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for maintaining competitiveness in the global pharmaceutical market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101899044B are fully realized in practical, GMP-compliant manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Gemifloxacin intermediate meets the highest international standards for impurity profiles and residual solvents.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your production of this vital antibiotic intermediate is both economically optimized and technically robust.
