Advanced Microwave-Assisted Synthesis of Hypericin: A Technical Breakthrough for Commercial Scale-Up
Advanced Microwave-Assisted Synthesis of Hypericin: A Technical Breakthrough for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable routes for synthesizing complex bioactive molecules. Patent CN102126942A introduces a transformative methodology for the total synthesis of hypericin, a potent naphthodianthrone derivative known for its significant antiviral, antidepressant, and antitumor properties. Unlike traditional extraction methods which suffer from low natural abundance (approximately 0.04% in plant material) and ecological strain, this chemical synthesis pathway leverages modern microwave technology to achieve superior yields and purity. For R&D directors and procurement specialists, this patent represents a pivotal shift towards a more reliable hypericin supplier capability, decoupling production from agricultural volatility and establishing a foundation for consistent, high-volume manufacturing of this critical active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of hypericin has been plagued by significant inefficiencies inherent to both natural extraction and earlier synthetic attempts. Prior art, such as the method disclosed in Chinese patent application CN1827574A, relies on a cumbersome sequence involving pyridine, piperidine, and iron salts (FeSO4·7H2O) for the critical dimerization step. These conventional processes are characterized by prolonged reaction times, often extending over several hours under dark conditions, and generate substantial chemical waste due to the complexity of the reagent mixture. Furthermore, the purification of the final product from such complex reaction matrices is notoriously difficult, leading to lower overall recovery rates and inconsistent batch-to-batch quality. For supply chain heads, these factors translate into unpredictable lead times and elevated costs associated with waste disposal and extensive downstream processing, making traditional routes economically unviable for large-scale commercial adoption.
The Novel Approach
In stark contrast, the methodology outlined in CN102126942A streamlines the synthesis through a clever application of microwave-assisted organic synthesis (MAOS) and simplified catalysis. The core innovation lies in the replacement of the toxic and slow iron-catalyzed system with a rapid, base-mediated condensation using potassium tert-butoxide. By conducting this reaction in a microwave solid-liquid phase synthesis instrument at controlled temperatures between 130-150°C, the reaction time is compressed dramatically to merely 30-90 minutes. This approach not only simplifies the operational workflow by eliminating volatile amines like pyridine but also enhances the selectivity of the dimerization, thereby reducing the burden on purification units. This technological leap directly addresses the need for cost reduction in pharmaceutical intermediates manufacturing by minimizing energy consumption and maximizing throughput per unit time.
Mechanistic Insights into Microwave-Assisted Oxidative Dimerization
The synthetic route begins with the reduction of emodin to emodin anthrone, a crucial precursor that sets the stage for the subsequent dimerization. In this initial step, emodin is dissolved in glacial acetic acid and treated with stannous chloride dihydrate (SnCl2·2H2O) and concentrated hydrochloric acid at elevated temperatures of 100-125°C. This reduction converts the quinone moiety into the anthrone form, which is essential for the coupling reaction. The process is meticulously controlled under an argon atmosphere to prevent premature oxidation, ensuring high conversion rates. As illustrated in the reaction scheme below, this transformation is highly efficient, with reported yields reaching up to 93%, providing a robust starting material for the critical coupling step.
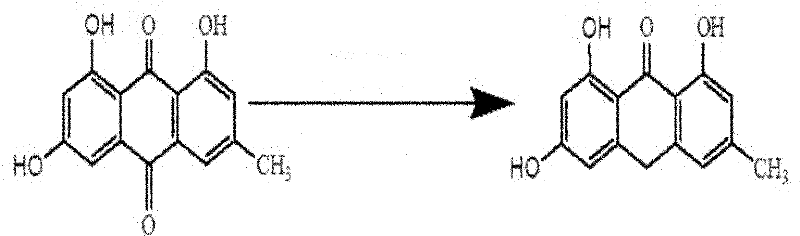
Following the formation of emodin anthrone, the process advances to the pivotal oxidative dimerization step, which constructs the complex naphthodianthrone skeleton of protohypericin. This step utilizes potassium tert-butoxide as a strong base catalyst in dimethylformamide (DMF) solvent under microwave irradiation. The microwave energy facilitates rapid dielectric heating, which significantly accelerates the kinetics of the condensation reaction compared to conventional conductive heating methods. The molar ratio of emodin anthrone to potassium tert-butoxide is optimized between 3-5:0.5-0.7, ensuring sufficient basicity to drive the coupling without excessive degradation. The reaction proceeds under inert gas protection at a power of 250-550W, yielding protohypericin with high selectivity. The structural evolution during this phase is depicted in the following diagram, highlighting the formation of the central biaryl bond.
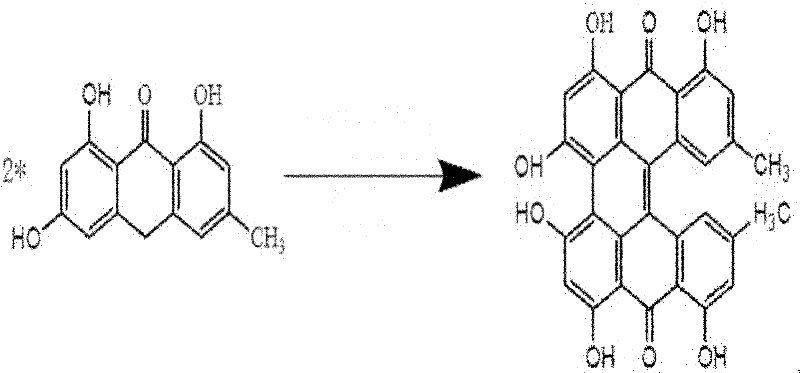
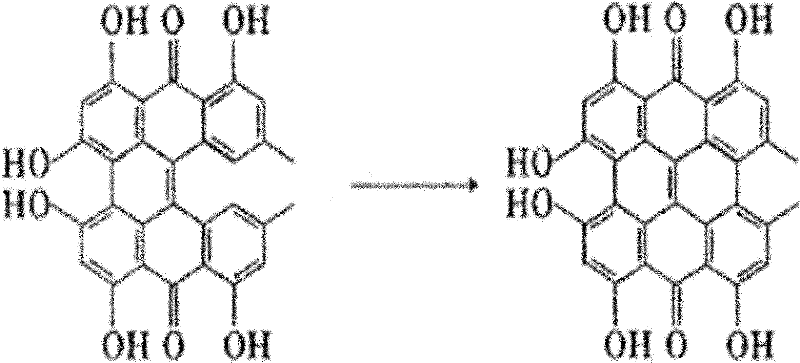
The final transformation involves the photo-oxidation of protohypericin to yield the target molecule, hypericin. This step capitalizes on the photosensitive nature of the proto-form, which, upon exposure to visible light in the presence of oxygen, undergoes dehydrogenation to form the fully conjugated aromatic system of hypericin. The protocol specifies dissolving protohypericin in acetone and irradiating the solution with a 500-watt halogen lamp for 15-24 hours under argon protection. This mild oxidative condition avoids the use of harsh chemical oxidants, thereby preserving the integrity of the sensitive hydroxyl groups on the periphery of the molecule. The resulting product is obtained with a remarkable yield of 95% and a purity of ≥98.5% after silica gel column chromatography, as shown in the final reaction scheme below, confirming the efficacy of this gentle yet effective oxidation strategy.
How to Synthesize Hypericin Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process is divided into three distinct stages: the acidic reduction of emodin, the microwave-mediated dimerization, and the final photo-oxidation. Each stage demands specific attention to temperature, stoichiometry, and atmospheric conditions to prevent side reactions. For laboratory and pilot-scale operations, adhering to the standardized protocols derived from the patent data is essential to replicate the high success rates reported. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety precautions required for handling reagents like concentrated hydrochloric acid and potassium tert-butoxide.
- Reduce emodin to emodin anthrone using SnCl2·2H2O and concentrated hydrochloric acid in glacial acetic acid at 100-125°C.
- Perform oxidative dimerization of emodin anthrone using potassium tert-butoxide in DMF under microwave irradiation at 130-150°C to form protohypericin.
- Convert protohypericin to hypericin via photo-oxidation using a halogen lamp in acetone for 15-24 hours.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this microwave-assisted synthesis route offers profound advantages for procurement managers and supply chain strategists looking to optimize their sourcing of high-purity hypericin. The elimination of expensive and toxic transition metal catalysts, such as the iron salts used in prior art, significantly reduces the raw material costs and simplifies the waste treatment protocols. Furthermore, the drastic reduction in reaction time—from several hours to under 90 minutes for the key step—enhances the overall equipment effectiveness (OEE) of the manufacturing facility, allowing for greater production capacity without additional capital expenditure. This efficiency gain translates directly into substantial cost savings and improved margin potential for downstream pharmaceutical applications.
- Cost Reduction in Manufacturing: The streamlined reagent profile eliminates the need for complex amine bases and heavy metal catalysts, which are not only costly but also require expensive removal processes to meet pharmaceutical purity standards. By utilizing common industrial chemicals like potassium tert-butoxide and DMF, the direct material costs are significantly lowered. Additionally, the high yield of the final photo-oxidation step (95%) minimizes material loss, ensuring that the maximum amount of input raw material is converted into saleable product, thereby optimizing the cost of goods sold (COGS) for the final API.
- Enhanced Supply Chain Reliability: Dependence on agricultural extraction for hypericin subjects the supply chain to seasonal variations, weather risks, and geopolitical instability in sourcing regions. This fully synthetic route decouples production from these external factors, ensuring a continuous and predictable supply of the active ingredient. The use of readily available starting materials like emodin, which can be sourced from multiple suppliers globally, further mitigates the risk of single-source bottlenecks, providing procurement teams with greater flexibility and security in their inventory planning.
- Scalability and Environmental Compliance: The simplicity of the workup procedures, primarily involving filtration and standard column chromatography, makes this process highly amenable to scale-up from kilogram to multi-ton production levels. Moreover, the reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the liability and cost associated with effluent treatment. The microwave technology itself is energy-efficient, contributing to a smaller carbon footprint for the manufacturing process, which is a critical metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is crucial for stakeholders evaluating its feasibility for commercial production. The following questions address common concerns regarding process safety, scalability, and quality control, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational advantages and assist technical teams in making informed decisions regarding the adoption of this technology.
Q: What are the primary advantages of the microwave-assisted method over traditional extraction?
A: Traditional extraction from Hypericum perforatum yields less than 1% hypericin and damages vegetation. This synthetic method offers high relative yield (up to 95% in the final step) and consistent purity (≥98.5%), ensuring a stable supply independent of agricultural variables.
Q: How does the novel dimerization step improve process efficiency?
A: By utilizing potassium tert-butoxide as a strong base catalyst under microwave irradiation (250-550W), the reaction time is drastically reduced to 30-90 minutes compared to hours in conventional methods, while eliminating complex reagents like pyridine and piperidine.
Q: What purification strategies are employed to ensure pharmaceutical grade quality?
A: The process utilizes silica gel column chromatography with specific eluent systems (petroleum ether:ethyl acetate:methanol) to remove impurities effectively, resulting in a final product with purity specifications suitable for rigorous pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hypericin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a robust and scalable supply chain for complex pharmaceutical intermediates like hypericin. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical partners. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of hypericin supplied adheres to the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this efficient route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us support your journey towards bringing high-quality antiviral and antidepressant therapies to the market.
