Advanced Acenaphthyl Phosphine Ligands for High-Efficiency Palladium Catalysis and Commercial Scale-Up
The chemical industry continuously seeks advancements in catalytic efficiency, and patent CN110483571B introduces a groundbreaking class of bulky electron-deficient organophosphine ligands based on an acenaphthylene skeleton. These novel compounds, specifically (1-(substituted phenyl)acenaphthyl)-bis(3,5-bis(trifluoromethyl))phenylphosphine derivatives, address critical limitations in palladium-catalyzed cross-coupling reactions. Unlike traditional ligands that may suffer from instability or limited substrate scope, this innovation offers multiple modification sites and robust structural integrity. The ability to exist stably in air significantly reduces handling complexities, making them highly attractive for large-scale pharmaceutical and fine chemical manufacturing processes where reliability is paramount.
Furthermore, the synthesis method described ensures high purity and reproducibility, essential factors for maintaining consistent catalytic performance across different batches. By leveraging a larger conjugated system, these ligands facilitate stronger coordination with transition metals, thereby enhancing the overall reaction kinetics. This technological leap not only improves yield but also streamlines the production workflow, positioning these ligands as a cornerstone for next-generation catalytic systems in complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
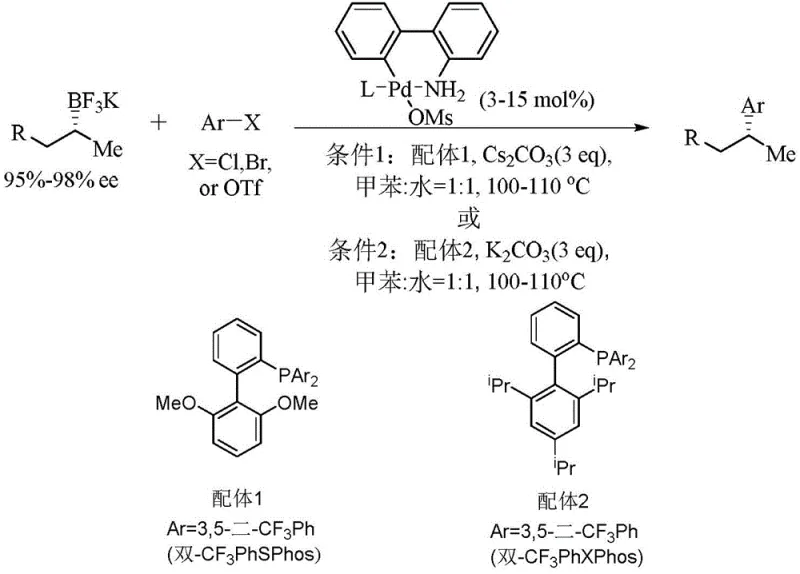
Historically, the development of organophosphine ligands has faced significant hurdles, particularly concerning steric hindrance and electronic properties. Conventional methods often rely on indenyl-based scaffolds, which, while effective in certain contexts, exhibit weaker conjugation effects and susceptibility to isomerization under alkaline conditions. This instability can lead to configuration changes during the reaction, resulting in inconsistent product quality and reduced yields. Additionally, traditional synthetic routes frequently involve lithiation steps that pose selectivity challenges, generating unwanted by-products that complicate purification and increase overall production costs.
Moreover, commercially available ligands like JackiePhos, though widely used, have demonstrated limitations in specific coupling scenarios, such as the construction of sp2-sp3 carbon bonds involving primary alkyl groups. The lower yields observed with these established ligands necessitate higher catalyst loadings or extended reaction times, which negatively impacts process economics. The inability to efficiently handle bulky substrates further restricts their applicability in synthesizing complex intermediates required for advanced drug discovery and material science applications.
The Novel Approach
The innovative approach detailed in the patent utilizes an acenaphthylene backbone to overcome these persistent challenges. By incorporating a larger conjugated system, the new ligands provide enhanced electronic communication with the palladium center, promoting efficient transmetallation and reductive elimination steps. This structural advantage translates directly into superior catalytic activity, as evidenced by significant improvements in coupling yields compared to prior art. The synthetic strategy prioritizes the introduction of the phosphine group before the aryl coupling, ensuring better control over reaction site selectivity and minimizing side reactions.
Crucially, this method avoids the pitfalls of lithiation-induced isomerization, guaranteeing structural fidelity throughout the synthesis. The resulting ligands are not only more active but also exhibit remarkable stability in air, simplifying storage and transportation logistics. This robustness is a game-changer for supply chain managers, as it reduces the risk of degradation during transit and ensures that the material arrives at the manufacturing site in optimal condition, ready for immediate use in sensitive catalytic processes.
Mechanistic Insights into Acenaphthylene-Based Phosphine Coordination
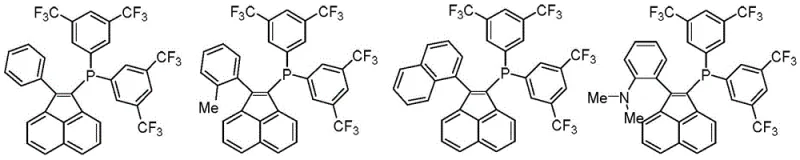
The mechanistic superiority of these ligands stems from their unique electronic and steric profile. The acenaphthylene core offers a rigid, planar structure that enforces a specific geometry around the phosphorus atom, optimizing the bite angle for palladium coordination. This geometric constraint facilitates the oxidative addition of aryl halides, a critical step in the catalytic cycle. Furthermore, the electron-deficient nature of the bis(3,5-bis(trifluoromethyl))phenyl groups withdraws electron density from the phosphorus, making the metal center more electrophilic and thus more reactive towards nucleophilic attack by organometallic species.
In terms of impurity control, the stability of the ligand under basic conditions is a decisive factor. Unlike indenyl analogues that may undergo base-catalyzed isomerization, the acenaphthylene framework remains intact, preventing the formation of inactive catalytic species. This stability ensures that the active catalyst concentration remains high throughout the reaction duration, leading to cleaner reaction profiles and easier downstream processing. For R&D directors, this means fewer variables to control and a more predictable outcome when scaling up from milligram to kilogram quantities.
How to Synthesize (1-(Substituted Phenyl)acenaphthyl) Phosphine Efficiently
The synthesis of these high-performance ligands follows a streamlined four-step protocol designed for scalability and safety. Starting from readily available acenaphthylene, the process involves controlled bromination, selective halogen exchange, Grignard addition, and a final palladium-catalyzed coupling. Each step is optimized to maximize yield and minimize waste, utilizing common solvents and reagents that are easily sourced globally. This accessibility is crucial for maintaining a steady supply chain and avoiding bottlenecks associated with exotic starting materials.
- Preparation of 1,2-dibromoacenaphthylene via radical bromination using NBS and benzoyl peroxide.
- Selective lithiation and iodination to form 1-bromo-2-iodo-acenaphthylene intermediate.
- Grignard reaction followed by phosphorylation to install the bis(3,5-bis(trifluoromethyl))phenylphosphine group.
- Final palladium-catalyzed cross-coupling with substituted phenylboronic acids to yield the target ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits beyond mere chemical performance. The use of readily available raw materials such as acenaphthylene and common boronic acids significantly reduces the cost of goods sold. By eliminating the need for specialized, hard-to-source precursors, manufacturers can secure a more resilient supply chain that is less vulnerable to market fluctuations. The simplified synthesis route also translates to shorter production cycles, allowing for faster response times to customer demands and reducing inventory holding costs.
Additionally, the air stability of the final product drastically lowers packaging and storage requirements. There is no need for expensive inert atmosphere containment during shipping, which reduces logistical expenses and carbon footprint. The high purity achievable through standard column chromatography means less solvent consumption for recrystallization, aligning with environmental compliance goals. These factors collectively contribute to substantial cost savings in manufacturing, making the final catalytic process more economically viable for large-scale industrial applications without compromising on quality.
- Cost Reduction in Manufacturing: The synthetic route avoids expensive transition metal removal steps often required with less stable ligands, as the robust structure minimizes metal leaching and decomposition. This inherent stability reduces the burden on downstream purification processes, leading to lower operational expenditures. Furthermore, the high catalytic efficiency allows for potentially lower catalyst loading in the final application, directly decreasing the amount of precious palladium required per batch of product.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward, as the key building blocks are commodity chemicals available from multiple global suppliers. This diversification mitigates the risk of single-source dependency, ensuring continuous production even during regional disruptions. The stability of the ligand also extends its shelf life, allowing for strategic stockpiling without the fear of degradation, which provides a buffer against sudden spikes in demand or supply chain delays.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing standard reaction conditions that are easily transferable from laboratory to pilot plant and full commercial production. The avoidance of hazardous lithiation reagents in the final steps and the use of greener solvents where possible support sustainability initiatives. Reduced waste generation and lower energy consumption due to milder reaction conditions contribute to a smaller environmental footprint, facilitating regulatory approval and enhancing corporate social responsibility profiles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these novel ligands in industrial settings. Understanding these aspects is vital for decision-makers evaluating the transition from existing catalytic systems to this advanced technology. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for professional assessment.
Q: Why are acenaphthylene-based ligands superior to indenyl analogues?
A: Acenaphthylene scaffolds offer a larger conjugated system compared to indenyl structures, providing stronger coordination with transition metals and enhanced stability under alkaline conditions, preventing isomerization.
Q: What is the primary advantage regarding reaction yield?
A: Experimental data demonstrates a significant increase in coupling yield, achieving up to 90% gas-phase yield in specific alkyl-aryl couplings compared to substantially lower yields with commercial alternatives like JackiePhos.
Q: Are these ligands stable for long-term storage?
A: Yes, the bulky electron-deficient nature combined with the rigid acenaphthylene backbone ensures the ligands can exist stably in air, simplifying handling and storage requirements for industrial users.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organophosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality ligands play in the success of modern chemical synthesis. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing floor is seamless. Our commitment to stringent purity specifications and rigorous QC labs guarantees that every batch of ligand meets the exacting standards required for sensitive pharmaceutical and fine chemical applications, providing you with the confidence to innovate.
We invite you to collaborate with our technical procurement team to explore how these advanced acenaphthyl phosphine ligands can optimize your specific processes. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits tailored to your production volume. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, paving the way for a more efficient and cost-effective manufacturing future.
