Advanced Synthesis of 2-Substituted Halopyridines: Enhancing Purity and Commercial Viability
Advanced Synthesis of 2-Substituted Halopyridines: Enhancing Purity and Commercial Viability
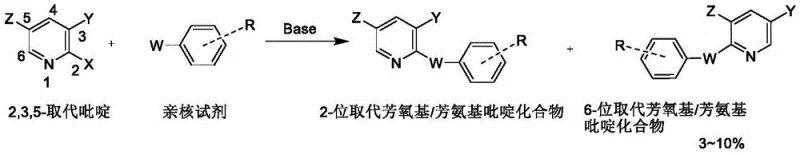
The pharmaceutical and agrochemical industries rely heavily on the availability of high-purity heterocyclic building blocks, among which 2-substituted halopyridine compounds stand out as critical intermediates for liquid crystal materials, pesticides, and active pharmaceutical ingredients. A significant technological breakthrough in this domain is documented in Chinese Patent CN110894188B, which details a novel preparation method that fundamentally alters the economic and technical landscape of producing these valuable molecules. This patent introduces a sophisticated etherification strategy using 2,3-dihalopyridine compounds as raw materials, reacting them with nucleophilic reagents in a uniquely engineered mixed solvent system. By shifting away from traditional single-solvent approaches, this innovation effectively suppresses the formation of unwanted 6-position substitution isomers, a persistent challenge that has long plagued manufacturers. The result is a streamlined process that delivers products with greater than 96% content directly from the reaction mixture, boasting yields exceeding 88% and eliminating the need for costly and yield-reducing recrystallization steps. For global supply chain leaders and R&D directors, this represents a pivotal shift towards more sustainable and cost-effective manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted halopyridine derivatives has been fraught with selectivity issues that directly impact commercial viability and production costs. In standard etherification reactions involving 2,3-dihalopyridines, the nucleophilic attack does not exclusively occur at the desired 2-position; instead, a significant portion of the reaction proceeds at the 6-position of the pyridine ring. Literature and prior art, such as methods reported by Chen Qiang and Tan Chengxia, indicate that these processes typically generate between 5% to 10% of substitution position-transfer isomers. To meet the stringent purity requirements of the pharmaceutical market, which often demand content levels above 96%, manufacturers are forced to employ rigorous purification techniques. The industry standard has been repeated recrystallization, a process that is not only solvent-intensive and time-consuming but also devastating to overall material throughput. Data indicates that after a single recrystallization, product yields can plummet to approximately 75%, and when pushing for ultra-low impurity profiles (below 1%), final yields often degrade to a mere 50-60%. This substantial loss of material translates directly into inflated raw material costs and increased waste disposal burdens, creating a significant bottleneck for large-scale production.
The Novel Approach
The methodology outlined in patent CN110894188B offers a transformative solution by re-engineering the reaction medium itself to control regioselectivity at the molecular level. Instead of relying solely on polar aprotic solvents which fail to discriminate effectively between the 2 and 6 positions, this novel approach employs a binary solvent system comprising a polar aprotic solvent and a specific lower alcohol. The introduction of secondary or tertiary alcohols, such as isopropanol or tert-butanol, into the reaction matrix creates a unique microenvironment that favors the desired transformation. Experimental results demonstrate that this mixed solvent strategy drastically inhibits the generation of the 6-position substituted isomer, controlling its content to below 1% without the need for downstream purification. Consequently, the reaction mixture, after simple filtration and concentration, yields a product with purity exceeding 96%, ready for direct market sale. This elimination of the recrystallization step not only preserves the high reaction yield of over 88% but also simplifies the equipment requirements and reduces the operational complexity, marking a distinct advantage over legacy synthetic routes.
Mechanistic Insights into Mixed-Solvent Regioselective Etherification
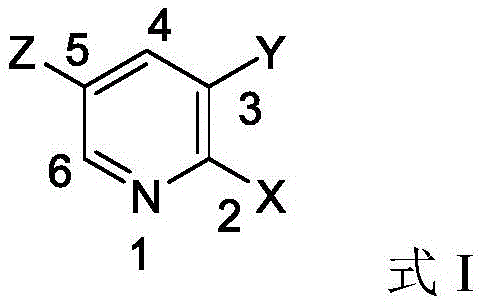
Understanding the mechanistic underpinnings of this improved synthesis is crucial for R&D directors aiming to replicate or adapt this technology for diverse substrates. The core innovation lies in the interaction between the solvent system and the electrophilic pyridine ring. In traditional polar aprotic media, the electron-withdrawing effects of the nitrogen atom and the halogen substituents activate both the 2 and 6 positions for nucleophilic aromatic substitution, leading to a mixture of regioisomers. However, the patent proposes that the addition of lower alcohols introduces a competitive hydrogen-bonding network. It is hypothesized that the hydroxyl group of the alcohol solvent forms a transient hydrogen bond complex with the halogen atom or the nitrogen center on the pyridine ring. This solvation shell effectively increases the steric bulk around the 6-position or modulates the electron density distribution, thereby raising the activation energy barrier for nucleophilic attack at that specific site. By kinetically favoring the attack at the 2-position, the reaction pathway is steered almost exclusively towards the desired product. This subtle yet powerful manipulation of solvent-solute interactions ensures that the nucleophile, whether an aryloxy or arylamino compound, reacts with high fidelity, minimizing the formation of difficult-to-separate structural analogues.
Furthermore, the control of water content and the specific choice of alcohol play pivotal roles in maintaining this high selectivity. The process mandates a rigorous dehydration step prior to the addition of the dihalopyridine, heating the base and nucleophile mixture to 80-120°C under vacuum. This step is critical because residual water can lead to hydrolysis of the sensitive halopyridine starting material, generating phenolic or pyridinol byproducts that complicate the impurity profile. Additionally, the selection of secondary or tertiary alcohols with fewer than six carbon atoms is deliberate; these solvents are sufficiently bulky to provide the necessary steric influence but lack the primary hydroxyl reactivity that might lead to competing etherification of the solvent itself. This precise balancing act between solvent polarity, hydrogen bonding capability, and steric hindrance creates a robust reaction window where the thermodynamic and kinetic drivers align to produce high-purity 2-substituted halopyridines consistently, providing a reliable foundation for scaling up complex heterocyclic syntheses.
How to Synthesize 2-Substituted Halopyridine Efficiently
The practical implementation of this patented technology involves a carefully sequenced three-stage protocol designed to maximize yield while minimizing impurity formation. The process begins with the preparation of the nucleophilic species in a dehydrated environment, followed by the controlled addition of the electrophile in the presence of the regioselectivity-enhancing alcohol co-solvent. This sequence ensures that the reactive intermediates are generated under optimal conditions before being exposed to the sensitive dihalopyridine substrate. The detailed operational parameters, including specific temperature ramps, vacuum levels, and molar ratios, are critical for achieving the reported >96% purity and >88% yield. For process chemists looking to adopt this route, adherence to the solvent dehydration and temperature control specifications is paramount to replicating the suppression of the 6-position isomer. The following guide outlines the standardized synthesis steps derived from the patent examples, serving as a foundational reference for laboratory and pilot-scale operations.
- System Dehydration: Heat a mixture of polar aprotic solvent, inorganic base, and nucleophile to 80-120°C under vacuum to remove water, then cool to 30-50°C.
- Etherification Reaction: Add a lower alcohol solvent (secondary or tertiary), heat to 50-90°C, and dropwise add the 2,3-dihalopyridine compound, maintaining reaction at 60-90°C for 3-12 hours.
- Post-Processing: Filter the reaction liquid, wash the filter cake, concentrate the filtrate, and perform water/alkali washing to obtain the product with over 96% content without recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible strategic benefits that extend far beyond simple chemical yield. The most immediate impact is the drastic simplification of the downstream processing workflow. By eliminating the recrystallization step, manufacturers can significantly reduce the consumption of organic solvents, which are often expensive and subject to volatile market pricing and environmental regulations. This reduction in solvent usage directly correlates to a decrease in waste generation, lowering the costs associated with hazardous waste disposal and environmental compliance. Furthermore, the removal of a time-intensive purification stage shortens the overall batch cycle time, allowing production facilities to increase throughput and respond more agilely to market demand fluctuations. The patent explicitly notes that industrial production costs can be reduced by 15%, a figure driven by the combined effects of higher material efficiency, lower utility consumption, and reduced labor hours per kilogram of finished product. This cost structure provides a competitive edge in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily due to the elimination of yield losses associated with purification. In conventional processes, achieving high purity often sacrifices more than 40% of the theoretical yield through multiple crystallization cycles. By securing a direct yield of over 88% with high purity, the effective cost per kilogram of the active ingredient is substantially lowered. Additionally, the avoidance of transition metal catalysts in some variations of this etherification further reduces raw material costs and eliminates the need for expensive heavy metal scavenging steps. The qualitative reduction in processing steps means less energy is required for heating, cooling, and drying, contributing to a leaner and more cost-efficient manufacturing operation that aligns with modern principles of green chemistry and economic sustainability.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthetic route enhances reliability and continuity of supply. The reliance on readily available commodity chemicals such as DMF, DMSO, and common alcohols like isopropanol ensures that raw material sourcing is not a bottleneck. Unlike processes dependent on exotic catalysts or specialized reagents that may face supply disruptions, this method utilizes a stable and accessible chemical inventory. The simplified process flow also reduces the risk of batch failures; fewer unit operations mean fewer opportunities for human error or equipment malfunction. This stability allows suppliers to offer more consistent lead times and maintain safety stocks with greater confidence, ensuring that downstream pharmaceutical and agrochemical manufacturers receive their critical intermediates on schedule, thereby safeguarding their own production timelines against upstream volatility.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often exposes hidden inefficiencies, but this method is inherently designed for industrial scalability. The exothermic nature of the etherification is manageable within standard reactor configurations, and the absence of difficult filtration steps (associated with crystallization) simplifies the hardware requirements. Environmentally, the process aligns with increasingly strict regulatory frameworks by minimizing solvent waste and avoiding the generation of complex mother liquors that are difficult to treat. The ability to produce high-purity material without extensive purification reduces the facility's overall environmental footprint, a key metric for corporate social responsibility and regulatory compliance. This makes the technology not just chemically superior but also strategically sound for companies aiming to future-proof their manufacturing assets against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 2-substituted halopyridine synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in patent CN110894188B, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios. The answers highlight the specific advantages in terms of impurity control, yield optimization, and operational simplicity that define this innovative approach.
Q: How does the new method suppress the formation of 6-position isomers?
A: The method utilizes a mixed solvent system of polar aprotic solvents and lower alcohols. The alcohol solvent forms hydrogen bond complexes with the halogen on the pyridine ring, sterically and electronically hindering nucleophilic attack at the 6-position, thereby reducing isomer content to below 1%.
Q: What are the yield improvements compared to conventional recrystallization methods?
A: Conventional methods often require multiple recrystallizations to remove 5-10% isomer impurities, dropping yields to 50-60%. This novel approach achieves over 88% yield directly with >96% purity, eliminating the need for yield-lossing recrystallization steps.
Q: Which solvents are critical for this etherification reaction?
A: The process requires a specific combination: a polar aprotic solvent (like DMF or DMSO) for the initial dehydration and base activation, followed by the addition of a secondary or tertiary alcohol (like isopropanol or tert-butanol) to control regioselectivity during the etherification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted Halopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patented laboratory methods to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN110894188B can be realized on an industrial scale. Our facilities are equipped with state-of-the-art reactors capable of handling the precise temperature and vacuum controls required for this dehydration and etherification process. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-substituted halopyridine meets the exacting standards of the global pharmaceutical and agrochemical industries. Our commitment to quality ensures that the high purity and low isomer content achieved in the lab are consistently delivered in every shipment.
We invite potential partners to engage with our technical procurement team to explore how this advanced synthesis route can optimize your supply chain. By leveraging our expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. Whether you require high-purity intermediates for clinical trials or bulk quantities for commercial drug manufacturing, NINGBO INNO PHARMCHEM is positioned to be your reliable partner in delivering cost-effective, high-quality chemical solutions that drive your business forward.
