Revolutionizing p-Toluenesulfonyl Isocyanate Production: A Safe, High-Yield Commercial Pathway
Revolutionizing p-Toluenesulfonyl Isocyanate Production: A Safe, High-Yield Commercial Pathway
The global demand for high-quality pharmaceutical intermediates continues to surge, driven by the expanding market for type II diabetes treatments and advanced agrochemicals. At the forefront of this evolution is the critical intermediate p-toluenesulfonyl isocyanate, a cornerstone molecule for synthesizing sulfonylurea drugs such as glimepiride and gliclazide. A groundbreaking technical disclosure, detailed in patent CN114181120A, introduces a transformative preparation method that fundamentally alters the risk-reward profile of manufacturing this essential compound. By replacing the notoriously hazardous phosgene gas with a safer dimethyl carbonate-based chlorination strategy, this innovation addresses long-standing safety concerns while delivering exceptional chemical efficiency. For R&D directors and supply chain leaders, this patent represents not just a procedural update, but a strategic opportunity to secure a more resilient and compliant supply chain for complex organic synthesis.
This technical insight report delves deep into the mechanistic advantages and commercial implications of this novel pathway. The shift away from high-pressure phosgene reactors to a more manageable atmospheric or near-atmospheric process significantly lowers the barrier to entry for safe production. Moreover, the reported yields exceeding 95% and purities surpassing 98% demonstrate that safety improvements do not come at the expense of performance. As we analyze the specific reaction conditions and purification protocols outlined in the patent, it becomes clear that this method offers a robust solution for the commercial scale-up of complex sulfonylurea intermediates, ensuring consistent quality for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of p-toluenesulfonyl isocyanate has been plagued by significant safety and engineering challenges associated with the use of phosgene gas. Traditional protocols typically require the reaction of p-toluenesulfonamide with phosgene under high-pressure and high-temperature conditions, necessitating specialized, heavily reinforced reactor vessels and extensive safety containment systems. The inherent toxicity of phosgene poses a catastrophic risk in the event of a leak, requiring facilities to maintain rigorous, costly monitoring and emergency response protocols. Furthermore, the high-pressure environment limits the scalability of batch processes and increases the capital expenditure required for compliant manufacturing infrastructure. These factors collectively contribute to higher production costs and potential supply chain vulnerabilities, as fewer manufacturers are willing or able to meet the stringent regulatory requirements for handling bulk phosgene.
The Novel Approach
The innovative method disclosed in patent CN114181120A circumvents these dangers by utilizing dimethyl carbonate (DMC) as a precursor for the carbonyl source, effectively generating the reactive species in situ without the need for external phosgene storage. In this streamlined process, chlorine gas is introduced into dimethyl carbonate to form a reactive intermediate, which then couples with p-toluenesulfonamide dissolved in chlorobenzene. This reaction proceeds efficiently at reflux temperatures between 90-110°C, eliminating the need for high-pressure equipment entirely. The result is a process that is inherently safer, easier to control, and more adaptable to standard chemical manufacturing facilities. By removing the most hazardous step from the supply chain, this approach facilitates cost reduction in pharmaceutical manufacturing by lowering insurance premiums, safety compliance overhead, and equipment maintenance costs, all while maintaining superior reaction kinetics.
Mechanistic Insights into Dimethyl Carbonate Chlorination and Coupling
To fully appreciate the elegance of this synthesis, one must examine the stepwise transformation of the raw materials into the final isocyanate product. The process begins with the chlorination of dimethyl carbonate, where chlorine gas reacts with the carbonate ester to generate a highly reactive trichloromethyl species, functionally acting as a solid or liquid equivalent of phosgene within the reaction matrix. This in situ generation ensures that the concentration of toxic gas remains negligible, as it is immediately consumed in the subsequent step. The precise stoichiometry, utilizing a molar ratio of dimethyl carbonate to chlorine of approximately 1:6 to 1:6.6, is critical to driving the chlorination to completion without excessive side reactions. This controlled activation sets the stage for the nucleophilic attack by the sulfonamide nitrogen, facilitating the formation of the sulfonyl isocyanate bond with high atom economy.
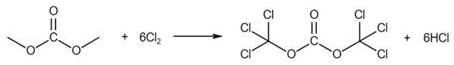
Following the activation of the carbonate, the p-toluenesulfonamide, dissolved in chlorobenzene, is introduced to the reaction kettle. The mechanism involves the nucleophilic attack of the sulfonamide nitrogen on the electrophilic carbonyl carbon of the activated intermediate. This step is conducted under reflux conditions at 90-110°C for 3 to 5 hours, ensuring complete conversion of the starting materials. The use of chlorobenzene as a solvent is particularly advantageous due to its high boiling point, which supports the necessary thermal energy for the reaction, and its ability to be easily recovered later. The reaction produces hydrogen chloride as a byproduct, which is managed within the closed system, preventing environmental release. The second reaction image below illustrates the stoichiometric coupling where three equivalents of the sulfonamide react with the activated carbonate species to yield three equivalents of the target isocyanate.
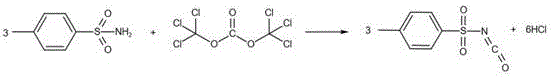
Impurity control is meticulously managed through the anhydrous nature of the reaction environment. Water is the enemy of isocyanates, as it leads to hydrolysis and the formation of unwanted ureas or amines, which degrade product quality. By maintaining an almost water-free system and using dry solvents, the patent ensures that hydrolysis is minimized, directly contributing to the reported high yields of over 95%. Furthermore, the final purification via high-vacuum distillation allows for the separation of the product from any remaining solvent or high-boiling byproducts. This physical separation method is superior to crystallization for this specific molecule, as it avoids the introduction of additional solvents that would require removal, thereby streamlining the workflow and enhancing the overall purity profile to exceed 98%.
How to Synthesize p-Toluenesulfonyl Isocyanate Efficiently
Implementing this synthesis route requires precise adherence to the reaction parameters defined in the patent to ensure reproducibility and safety. The process is divided into three distinct operational phases: the initial chlorination of the carbonate source, the coupling reaction with the sulfonamide, and the final purification via distillation. Operators must carefully monitor the temperature during chlorine introduction, keeping it between 30-90°C to prevent runaway exotherms, and maintain strict vacuum controls during the solvent recovery phase to maximize chlorobenzene recycling. The following guide outlines the standardized procedure derived from the patent examples, providing a roadmap for laboratory and pilot-scale execution.
- Perform chlorination reaction by introducing chlorine gas into dimethyl carbonate to generate the reactive carbonyl source.
- Conduct esterification by adding p-toluenesulfonamide solution and refluxing at 90-110°C to form the isocyanate bond.
- Execute refining via reduced pressure solvent recovery followed by high-vacuum distillation to isolate pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this phosgene-free methodology offers tangible strategic benefits that extend beyond simple chemical yield. The elimination of high-pressure phosgene reactors significantly reduces the capital intensity required for production facilities, allowing for a more distributed and resilient manufacturing network. This decentralization mitigates the risk of supply disruptions caused by regulatory shutdowns of high-hazard facilities, ensuring a more consistent flow of high-purity p-toluenesulfonyl isocyanate to downstream customers. Additionally, the ability to recycle the chlorobenzene solvent with high efficiency drastically reduces raw material consumption and waste disposal costs, contributing to a leaner and more sustainable cost structure that can be passed on to partners in the form of competitive pricing.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of equipment requirements and the optimization of raw material usage. By avoiding the need for specialized high-pressure vessels and complex gas handling systems, manufacturers can operate with lower fixed costs and reduced maintenance expenditures. Furthermore, the high recovery rate of the chlorobenzene solvent means that less fresh solvent needs to be purchased, and the volume of hazardous waste requiring treatment is substantially diminished. These operational efficiencies aggregate to create a significantly lower cost of goods sold (COGS), enabling suppliers to offer more attractive pricing tiers without compromising on margin or quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the stringent regulations surrounding toxic gases like phosgene. By adopting a synthesis route that relies on safer, more manageable reagents like dimethyl carbonate and chlorine, manufacturers can operate in a wider range of jurisdictions with fewer regulatory hurdles. This flexibility reduces the likelihood of production halts due to safety inspections or permit renewals, thereby reducing lead time for high-purity API precursors. The robustness of the reaction conditions also means that batch-to-batch variability is minimized, ensuring that procurement teams receive consistent quality shipments that meet strict specification sheets every time.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental risks, but this method is designed with green chemistry principles in mind. The near-anhydrous conditions prevent the generation of large volumes of acidic wastewater, a common issue in isocyanate synthesis. The closed-loop nature of the solvent recovery system ensures that volatile organic compounds (VOCs) are contained and reused rather than emitted. This alignment with environmental, social, and governance (ESG) goals makes the supply chain more attractive to multinational corporations seeking sustainable partners. The process is inherently scalable from pilot batches to multi-ton production runs without requiring fundamental changes to the reactor design, facilitating rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of p-toluenesulfonyl isocyanate using this advanced methodology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on yield expectations, safety protocols, and purification standards. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own synthetic workflows.
Q: Why is the dimethyl carbonate route safer than traditional phosgene methods?
A: Traditional methods utilize gaseous phosgene, which requires high-pressure equipment and poses severe toxicity risks. The patented method generates the reactive species in situ from dimethyl carbonate and chlorine, eliminating the need for storing or transporting bulk phosgene gas, thereby significantly reducing operational hazards and equipment complexity.
Q: What purity levels can be achieved with this new synthesis method?
A: By employing a rigorous high-vacuum distillation process after the reaction, the method consistently achieves product purity exceeding 98%. This high level of purity is critical for pharmaceutical applications, particularly in the synthesis of sulfonylurea drugs like glimepiride, where impurity profiles must be strictly controlled.
Q: How does this process impact environmental compliance and waste management?
A: The process operates in an almost anhydrous environment, preventing hydrolysis and minimizing wastewater generation. Furthermore, the reaction solvent, chlorobenzene, is recovered under reduced pressure and recycled back into the process, leading to high utilization rates and a drastic reduction in hazardous waste discharge compared to conventional aqueous workups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable p-Toluenesulfonyl Isocyanate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final pharmaceutical product depends on the quality of every intermediate in the chain. As a leading CDMO and supplier, we have extensively evaluated the technical merits of the phosgene-free synthesis route described in patent CN114181120A and possess the expertise to implement it at scale. Our facilities are equipped with the necessary corrosion-resistant reactors and high-vacuum distillation columns to execute this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need kilogram quantities for R&D or metric tons for commercial API manufacturing, our stringent purity specifications and rigorous QC labs guarantee a product that meets the highest global standards.
We invite you to collaborate with us to optimize your supply chain for sulfonylurea intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this safer, more efficient process can improve your bottom line. Contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us be your partner in delivering safe, high-quality chemical solutions that drive innovation in the pharmaceutical industry.
