Scalable Production of Optically Pure Omega-Hydroxy Acid Derivatives for Pharma
Scalable Production of Optically Pure Omega-Hydroxy Acid Derivatives for Pharma
The pharmaceutical and fine chemical industries constantly seek efficient routes to chiral building blocks, particularly those featuring specific stereochemical arrangements like chiral methyl side chains. Patent CN100548965C presents a significant technological advancement in this domain by detailing a robust method for synthesizing ω-hydroxy acid derivatives with chiral methyl side chains. This innovation leverages optically pure starting materials such as 4R-4-methylvalerolactone and 3R-3-methylbutyrolactone, which are conveniently derived from the oxidative degradation of steroidal sapogenins. The general structural framework of these valuable intermediates is depicted below, showcasing the versatility of the R and X groups which allow for diverse downstream functionalization.
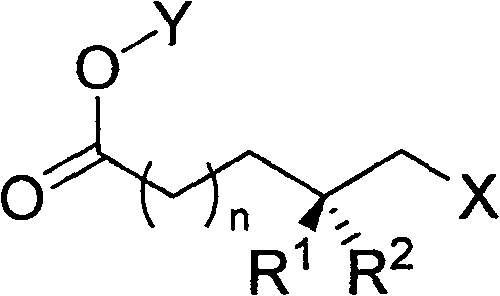
These compounds serve as critical precursors for the synthesis of high-value targets including Vitamin E, Vitamin K, insect sex pheromones, and various bioactive natural products. By establishing a reliable supply chain for these medium-length bifunctional chiral synthons, manufacturers can significantly streamline the production of complex active pharmaceutical ingredients (APIs) and agrochemical intermediates. The ability to access both R and S configurations with high optical purity addresses a longstanding bottleneck in asymmetric synthesis, offering a practical solution for large-scale industrial applications where cost and availability are paramount concerns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral synthetic building blocks characterized by bifunctional structures with chiral methyl side chains has relied heavily on asymmetric synthesis techniques or the modification of natural products. Common laboratory methods involve the introduction of chiral auxiliaries such as Evans reagents or S(R)-AMP, or direct reactions with chiral substrates like L-tartrate derivatives and BINOL-Ti complexes. While effective on a small scale, these approaches suffer from severe limitations when translated to commercial manufacturing. The synthetic routes are often excessively long, requiring multiple protection and deprotection steps that drastically reduce overall yield. Furthermore, the reagents employed, particularly chiral ligands and catalysts, are prohibitively expensive, rendering the final product cost-prohibitive for bulk commodity chemicals. Additionally, biotechnological methods, while greener, often struggle with substrate specificity, limiting the range of carbon chain lengths that can be effectively produced, particularly for medium-length chains greater than three carbons but less than seven.
The Novel Approach
The methodology outlined in CN100548965C offers a transformative alternative by utilizing readily available, renewable resources as the chiral pool. Specifically, it capitalizes on the F-ring portion of steroidal sapogenins like diosgenin and sisal sapogenin, which are typically discarded as waste during the production of steroid drugs. Through an established oxidative degradation process using hydrogen peroxide instead of toxic chromic anhydride, optically pure methyl valerolactone and methyl butyrolactone are obtained. 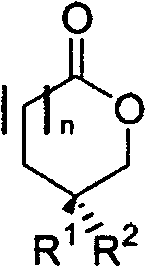 The core innovation lies in the subsequent stabilization of these lactones. Since the raw lactones are prone to polymerization and difficult to store, the patent describes converting them into stable, chain-like compounds. This approach not only valorizes industrial waste but also provides a direct, short route to medium-length chiral building blocks that were previously difficult to access economically. The resulting derivatives possess the necessary bifunctionality for further elaboration while maintaining the critical stereochemical integrity required for high-performance end products.
The core innovation lies in the subsequent stabilization of these lactones. Since the raw lactones are prone to polymerization and difficult to store, the patent describes converting them into stable, chain-like compounds. This approach not only valorizes industrial waste but also provides a direct, short route to medium-length chiral building blocks that were previously difficult to access economically. The resulting derivatives possess the necessary bifunctionality for further elaboration while maintaining the critical stereochemical integrity required for high-performance end products.
Mechanistic Insights into Acid-Catalyzed Lactone Ring-Opening
The chemical transformation at the heart of this technology involves the regioselective ring-opening of the cyclic ester (lactone) functionality. In the presence of an alcohol solvent and an acid catalyst, such as hydrobromic acid or sulfuric acid, the lactone ring undergoes nucleophilic attack by the alcohol molecule. This reaction proceeds through a protonated carbonyl intermediate, which increases the electrophilicity of the carbonyl carbon. The alcohol then attacks, leading to the cleavage of the acyl-oxygen bond and the formation of a linear hydroxy ester. This process is highly efficient, as demonstrated in Example 1, where refluxing 4R-4-methylvalerolactone in anhydrous isopropanol with catalytic HBr yields the corresponding isopropyl ester in excellent conversion.
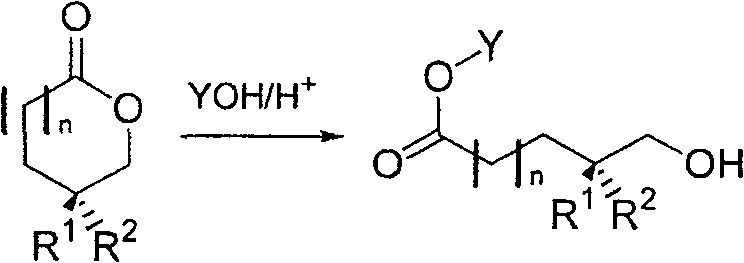
Beyond simple alcoholysis, the patent elucidates mechanisms for introducing diverse functional groups at the terminal position. By employing nucleophiles such as hydrogen halides (HX) or thiols (HSR) in inert solvents or alcohols, the ring opening can be directed to install halogen or sulfide functionalities directly. For instance, treating the lactone with HCl gas in methanol results in the formation of a chloro-ester, effectively installing a leaving group for subsequent substitution reactions. This mechanistic flexibility allows chemists to tailor the terminal functionality (X group) to specific synthetic needs, whether it be an alcohol for oxidation, a halide for coupling, or a protected ether for stability. The preservation of the chiral center at the methyl branch during these acidic or nucleophilic conditions is crucial, ensuring that the optical purity of the starting sapogenin-derived material is retained in the final product.
How to Synthesize Chiral Methyl Building Blocks Efficiently
The synthesis of these valuable intermediates follows a logical progression from raw material selection to functional group manipulation. The process begins with the isolation of the specific optical isomer of the methyl lactone, followed by the chosen ring-opening protocol. Depending on the desired terminal functionality, operators can select between alcoholysis for hydroxy esters or nucleophilic attack for halo/sulfide derivatives. The reaction conditions are mild, typically ranging from room temperature to reflux, and utilize standard laboratory glassware or industrial reactors. Detailed standard operating procedures regarding stoichiometry, temperature control, and workup protocols are essential for maintaining high purity and yield.
- Select optically pure starting materials such as 4R-4-methylvalerolactone or 3R-3-methylbutyrolactone derived from steroidal sapogenin degradation.
- Perform ring-opening reactions using either alcoholic solvents with acid/base catalysis for hydroxy esters, or nucleophiles like HX/HY for halo/sulfide derivatives.
- Purify the resulting medium-length bifunctional compounds through standard extraction, drying, and distillation or column chromatography techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology represents a strategic opportunity to optimize costs and secure supply continuity. The shift from synthetic chiral catalysts to bio-based feedstocks fundamentally alters the cost structure of producing these intermediates. By sourcing raw materials from the waste stream of the steroid industry, manufacturers can achieve substantial cost reductions compared to purchasing expensive chiral reagents or resolving racemic mixtures. This circular economy approach not only lowers the direct material costs but also mitigates the price volatility associated with specialty chemical markets. Furthermore, the simplicity of the reaction workup, which often involves basic extraction and distillation rather than complex chromatography, reduces processing time and solvent consumption, contributing to a leaner manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and chiral ligands removes a significant cost driver from the bill of materials. Traditional asymmetric synthesis often requires stoichiometric or near-stoichiometric amounts of expensive chiral auxiliaries that must be recovered and recycled, adding complexity and expense. In contrast, this process uses commodity chemicals like alcohols and mineral acids as reagents. The high yields reported in the patent examples indicate minimal material loss, further enhancing the economic viability. Additionally, the avoidance of cryogenic conditions or high-pressure equipment reduces capital expenditure and energy costs, making the process attractive for large-scale production facilities aiming to improve their bottom line through operational efficiency.
- Enhanced Supply Chain Reliability: Reliance on steroidal sapogenins, which are produced in massive quantities globally for the hormone industry, ensures a stable and abundant supply of the chiral precursor. Unlike niche chiral reagents that may have single-source suppliers and long lead times, the starting materials for this process are widely available commodities. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Moreover, the stability of the final ester and halo-ester products allows for inventory buffering, enabling manufacturers to stockpile key intermediates without significant degradation, thereby smoothing out demand fluctuations and ensuring consistent delivery to downstream customers.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, utilizing unit operations that are standard in the fine chemical industry. The replacement of chromium-based oxidants with hydrogen peroxide in the upstream preparation of the lactones aligns with modern green chemistry principles, reducing hazardous waste generation. Downstream, the use of simple aqueous workups minimizes the volume of organic waste streams. This environmental profile simplifies regulatory compliance and permitting for new production lines. The robustness of the chemistry means that scaling from pilot plant to commercial tonnage does not require fundamental re-engineering of the process, allowing for rapid capacity expansion to meet market demand for high-purity pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these aspects is vital for R&D teams evaluating the feasibility of integrating these building blocks into their existing pipelines. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of using steroidal sapogenin waste for chiral synthesis?
A: Utilizing waste streams from steroidal sapogenin degradation provides a renewable, cost-effective source of optically pure methyl lactones, bypassing the need for expensive asymmetric catalysts or resolution steps typically required in traditional chiral synthesis.
Q: How does this patent address the stability issues of methyl lactones?
A: The patent describes converting highly polymerizable and unstable optical pure methyl lactones into stable, medium-length chain compounds with bifunctional groups (hydroxy, halo, thio), making them suitable for long-term storage and downstream synthetic applications.
Q: Can these intermediates be scaled for industrial production?
A: Yes, the described methods utilize common reagents (alcohols, mineral acids, simple nucleophiles) and standard unit operations (reflux, extraction, distillation), which are inherently scalable from laboratory to multi-ton commercial manufacturing without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Methyl Building Blocks Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our omega-hydroxy acid derivatives meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance means that you can rely on us for consistent material performance, batch after batch.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the technical feasibility of a new route, our technical procurement team is ready to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your target molecules. Together, we can drive efficiency and innovation in the production of complex chiral pharmaceuticals.
