Advanced Convergent Synthesis of GLP-1 Analogues for Commercial Scale API Manufacturing
Introduction to Advanced GLP-1 Analogue Manufacturing
The escalating global demand for glucagon-like peptide-1 (GLP-1) receptor agonists, particularly for the management of type 2 diabetes and obesity, has necessitated the development of robust, high-yield synthetic routes for complex peptides like Liraglutide and Semaglutide. Patent CN112912390A introduces a groundbreaking convergent solid-phase synthesis method that addresses critical bottlenecks in traditional peptide manufacturing, specifically focusing on achieving superior purity and yield through strategic fragment condensation. This technology leverages the unique properties of pseudoproline dipeptides to mitigate aggregation and racemization, two persistent challenges in long-chain peptide synthesis. By implementing a pooled synthesis strategy where a protected N-terminal pseudoproline fragment is condensed with a solid-supported C-terminal fragment, manufacturers can overcome the solubility issues and low coupling efficiencies that often plague linear approaches. This report analyzes the technical merits of this invention, providing R&D and procurement leaders with a clear roadmap for integrating this high-efficiency process into their supply chains for reliable pharmaceutical intermediate sourcing.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing long peptides such as Liraglutide often rely on sequential solid-phase peptide synthesis (SPPS) or standard fragment condensation techniques that suffer from significant drawbacks regarding product quality and process efficiency. In conventional fragment-based synthesis, the activation of the C-terminal carboxyl group of a peptide fragment is a critical step that frequently leads to the formation of activated esters and oxazolones, which are prone to racemization under alkaline coupling conditions. This results in crude peptide mixtures containing difficult-to-separate isomeric impurities that possess retention times very similar to the target molecule, thereby complicating downstream purification and drastically reducing overall yield. Furthermore, long peptide chains are highly susceptible to intermolecular aggregation and intramolecular folding, phenomena that severely hinder the efficiency of amino acid coupling and deprotection steps, often leading to truncated sequences or complete synthesis failure. The low water solubility of protected fragments, particularly the N-terminal segments, further exacerbates these issues, making the final coupling steps kinetically sluggish and industrially unfeasible without specialized solubilizing strategies.
The Novel Approach
The innovative methodology described in the patent circumvents these historical limitations by employing a convergent strategy that utilizes a C-terminal pseudoproline reactive site on the N-terminal fragment (Fragment A). This approach fundamentally alters the physicochemical properties of the peptide segment during synthesis, effectively disrupting the secondary structures that lead to aggregation and ensuring that the C-terminal activation proceeds without significant racemization. By preparing Fragment A on a solid support and cleaving it while maintaining alpha-amino and side-chain protection, the process ensures that the fragment remains soluble and reactive for the final condensation with the resin-bound C-terminal Fragment B. This pooled synthesis allows for the independent optimization of both fragments, ensuring that the challenging lipidated C-terminal sequences are assembled with high fidelity before the final union. The result is a crude peptide with a markedly improved impurity profile, facilitating easier purification and delivering the final API in high overall yield, which is a transformative advantage for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pseudoproline-Mediated Cyclization
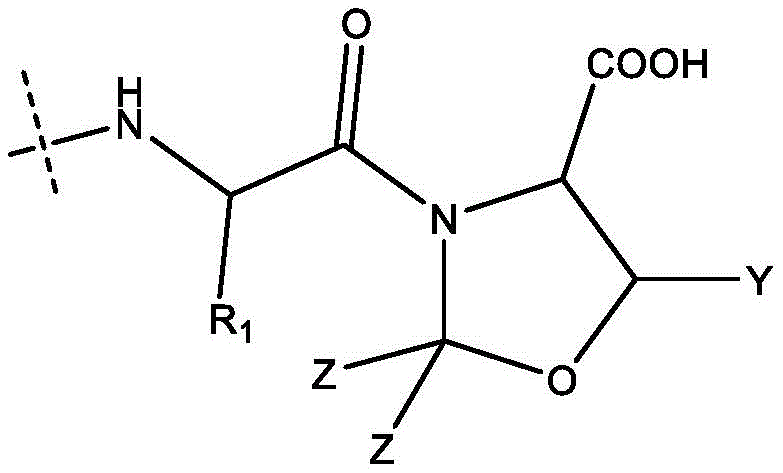
The core technical breakthrough of this synthesis lies in the strategic incorporation of pseudoproline dipeptides, which are oxazolidine derivatives formed from serine or threonine residues, acting as temporary structural disruptors within the peptide backbone. Mechanistically, the introduction of the pseudoproline moiety introduces a kink in the peptide chain that prevents the formation of stable beta-sheet structures, which are the primary drivers of peptide aggregation and insolubility during SPPS. This structural distortion ensures that the growing peptide chain remains accessible to coupling reagents, thereby maintaining high reaction kinetics even for long sequences. Furthermore, the cyclic nature of the pseudoproline protects the adjacent amide bond from participating in side reactions, and upon final acidolytic cleavage, it cleanly reverts to the native serine or threonine residue without leaving trace impurities. The specific geometry of the oxazolidine ring, as defined by the variables Y and Z in the patent structures, allows for fine-tuning of the steric environment to maximize solubility and minimize epimerization at the activation site.
In terms of impurity control, the use of pseudoproline at the C-terminal end of Fragment A effectively suppresses the formation of oxazolone intermediates that typically cause racemization during fragment condensation. The electron-withdrawing nature of the oxazolidine oxygen and the steric bulk of the dimethyl group stabilize the transition state during activation, favoring the desired nucleophilic attack by the N-terminal amine of Fragment B over the intramolecular cyclization that leads to racemization. This mechanism ensures that the stereochemical integrity of the peptide bond is preserved throughout the coupling process, resulting in a crude product with significantly lower levels of diastereomeric impurities. For R&D directors, this implies a more predictable and robust process where the burden on analytical QC is reduced, as the chromatographic separation of the target peptide from closely related isomers becomes far more manageable, directly translating to higher recovery rates and reduced solvent consumption during purification.
How to Synthesize GLP-1 Analogues Efficiently
The practical implementation of this convergent synthesis route involves a series of well-defined steps that leverage standard solid-phase equipment while introducing specific modifications to accommodate the pseudoproline chemistry. The process begins with the preparation of Fragment A, where a suitable C-terminal pseudoproline dipeptide is loaded onto a CTC resin, followed by the sequential addition of protected amino acids to build the N-terminal sequence. Crucially, the final cleavage of Fragment A is performed under mild acidic conditions that remove the peptide from the resin but leave the side-chain and alpha-amino protecting groups intact, rendering the fragment ready for solution-phase or on-resin coupling. Simultaneously, Fragment B is assembled on a Wang or MBH resin, incorporating the complex lipidated lysine moiety essential for albumin binding, with careful attention paid to capping unreacted sites to prevent deletion sequences. The detailed standardized synthesis steps, including specific reagent ratios, solvent systems, and reaction times optimized for this pathway, are outlined in the guide below.
- Prepare Fragment A by loading a C-terminal pseudoproline dipeptide onto CTC resin, extending the chain via SPPS, and cleaving while maintaining side-chain protection.
- Synthesize Fragment B on Wang or MBH resin by coupling C-terminal sequences, including the lipidated lysine moiety, ensuring the alpha-amino group is deprotected prior to final coupling.
- Condense Fragment A and Fragment B in the presence of coupling agents and detergents, followed by global deprotection, cleavage from resin, and HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this pseudoproline-mediated convergent synthesis offers substantial strategic benefits that extend beyond mere technical elegance, directly impacting the cost of goods sold (COGS) and supply security. The primary economic driver is the significant reduction in downstream processing costs; because the crude peptide generated by this method possesses a superior impurity profile with minimal racemization, the burden on preparative HPLC purification is drastically lowered. This translates to reduced consumption of expensive chromatography media and organic solvents, shorter cycle times for purification batches, and higher overall recovery of the active pharmaceutical ingredient, all of which contribute to a leaner and more cost-effective manufacturing operation. Furthermore, the enhanced solubility of the pseudoproline-containing fragments eliminates the need for exotic solubilizing additives or harsh conditions that can degrade equipment, thereby extending the lifespan of production assets and reducing maintenance downtime.
- Cost Reduction in Manufacturing: The elimination of difficult-to-remove isomeric impurities means that the purification process requires fewer passes and less solvent, leading to substantial cost savings in raw materials and waste disposal. By avoiding the formation of racemized by-products, the process maximizes the yield of the desired enantiomer, ensuring that expensive starting materials like protected amino acids and lipidated building blocks are not wasted on unusable side products. Additionally, the use of robust coupling conditions with common reagents like DIC and OxymaPure reduces the reliance on costly proprietary coupling agents, further optimizing the bill of materials for large-scale production runs.
- Enhanced Supply Chain Reliability: The convergent nature of this synthesis allows for the parallel production of Fragment A and Fragment B, effectively halving the critical path time for the synthesis of the full-length peptide compared to linear methods. This parallelization capability enhances supply chain resilience by allowing manufacturers to stockpile key intermediates and rapidly respond to fluctuations in market demand without compromising on quality. Moreover, the use of commercially available pseudoproline dipeptides and standard resins ensures that the supply of raw materials is stable and not subject to the bottlenecks often associated with custom-synthesized specialty reagents, guaranteeing continuity of supply for long-term contracts.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial feasibility in mind, utilizing standard solid-phase reactors that can be easily expanded from pilot to commercial scale without fundamental changes to the chemistry. The reduction in solvent usage and the higher atom economy resulting from improved yields align with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals. The ability to perform the final coupling in the presence of non-ionic detergents further improves the environmental profile by enabling the use of more benign solvent mixtures, reducing the toxic load of the effluent and simplifying waste treatment protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method, derived directly from the experimental data and claims presented in the patent documentation. These insights are intended to clarify the operational parameters and quality expectations for partners considering this technology for their API manufacturing portfolios. Understanding these nuances is critical for aligning R&D capabilities with commercial production targets and ensuring that the transition from lab-scale optimization to multi-ton production is seamless and compliant with regulatory standards.
Q: How does the pseudoproline strategy improve the purity of GLP-1 analogues?
A: The incorporation of a pseudoproline dipeptide at the C-terminal reactive site of Fragment A disrupts secondary structure aggregation and prevents the formation of oxazolones during activation, significantly reducing racemization and isomeric impurities.
Q: What resins are recommended for the fragment condensation process?
A: The patent specifies using acid-sensitive resins such as Wang resin or MBH resin for Fragment B to facilitate final cleavage, while 2-chlorotrityl chloride (CTC) resin is preferred for Fragment A to allow mild cleavage without removing side-chain protecting groups.
Q: Can this method be scaled for industrial production of Liraglutide?
A: Yes, the convergent solid-phase approach is designed for industrial feasibility, offering high overall yields and simplified purification steps compared to linear synthesis, making it suitable for large-scale API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GLP-1 Analogue Supplier
As the global market for GLP-1 analogues continues to expand, the ability to deliver high-purity intermediates and APIs with consistent quality and competitive pricing has become a decisive factor for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring advanced synthetic routes like the one described in CN112912390A to fruition. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications that ensure every batch of Liraglutide or Semaglutide intermediate meets the highest international pharmacopoeia standards. We understand the complexities of peptide chemistry, particularly the challenges of handling lipidated sequences and pseudoproline derivatives, and our technical team is dedicated to optimizing these processes for maximum efficiency and yield.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of how our implementation of this convergent synthesis can reduce your overall production costs and accelerate your time to market. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a secure and high-quality supply chain for your next-generation diabetes and obesity therapeutics.
