Advanced Chiral Titanium Catalysis for High-Purity Proton Pump Inhibitor Intermediates
Advanced Chiral Titanium Catalysis for High-Purity Proton Pump Inhibitor Intermediates
The pharmaceutical industry continuously seeks robust methodologies for the production of single-enantiomer drugs, particularly for the widely prescribed class of proton pump inhibitors (PPIs). Patent CN101429192A introduces a transformative approach to synthesizing chiral sulfoxide derivatives, specifically targeting the S-isomers or R-isomers of imidazole sulfoxide anti-ulcer compounds. This technology leverages a sophisticated chiral titanium complex system to selectively oxidize prochiral thioether precursors, addressing long-standing challenges in stereoselective synthesis. By utilizing dicumyl hydroperoxide as a specialized oxidant within a tailored catalytic environment, the process achieves exceptional optical purity exceeding 99% ee while maintaining high chemical purity. This breakthrough is pivotal for manufacturers aiming to produce high-value active pharmaceutical ingredients (APIs) such as Esomeprazole and Dexlansoprazole with superior efficiency.
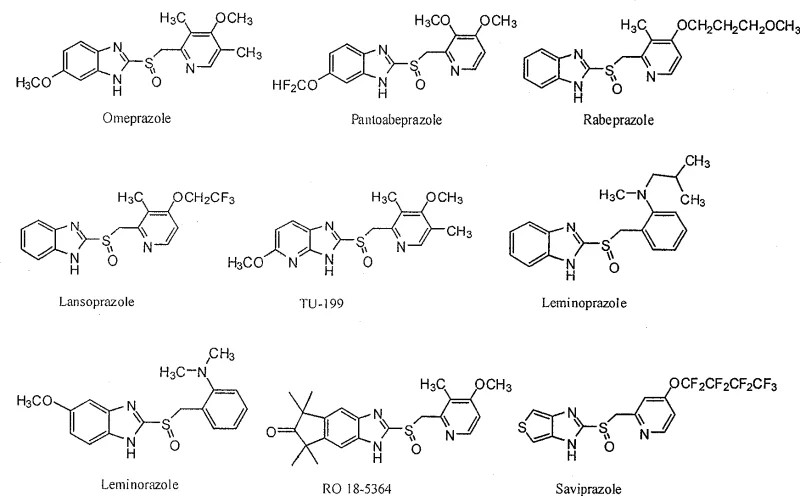
The structural complexity of these molecules, as illustrated in the provided chemical diagrams, underscores the necessity for precise stereocontrol during manufacturing. The presence of a chiral sulfur atom creates a pair of enantiomers with distinct physiological activities, making the selective production of the therapeutically active isomer a critical regulatory and commercial requirement. Traditional methods often struggle to differentiate between these mirror-image molecules without significant material loss. The methodology described in CN101429192A offers a direct route to the desired bioactive configuration, eliminating the need for inefficient resolution steps that plague older synthetic routes. For a reliable pharmaceutical intermediate supplier, adopting such advanced catalytic systems is essential to meet the stringent quality standards demanded by global health authorities and end-user patients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to obtaining single-isomer imidazole sulfoxides have been fraught with significant inefficiencies and economic drawbacks. Conventional chemical resolution methods, such as those involving diastereomeric salt formation or inclusion complexation, inherently suffer from a maximum theoretical yield of 50%, as the unwanted enantiomer is discarded or requires costly recycling processes. Furthermore, prior art stereoselective oxidation techniques, such as those disclosed in WO9602535 using cumene hydroperoxide, frequently encounter issues with over-oxidation. This side reaction generates sulfone impurities that possess physicochemical properties remarkably similar to the target sulfoxide, rendering purification extremely difficult and energy-intensive. Consequently, these legacy processes often result in suboptimal yields, sometimes as low as 29% after rigorous purification, and fail to consistently achieve the high enantiomeric excess required for modern drug registration.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally re-engineers the oxidation step to overcome these thermodynamic and kinetic barriers. By substituting traditional oxidants with dicumyl hydroperoxide (DCHP), the reaction profile is shifted towards a much milder and more controlled oxidation pathway. This specific choice of oxidant drastically suppresses the formation of sulfone by-products, thereby simplifying the downstream isolation of the target chiral sulfoxide. The process operates effectively within a temperature range of -20°C to 20°C, allowing for precise thermal management that further enhances selectivity. Unlike previous methods that yielded mixtures requiring complex chromatographic separation, this novel approach facilitates a streamlined workup procedure involving simple extraction and crystallization. This shift not only improves the overall mass balance of the synthesis but also aligns perfectly with the goals of cost reduction in API manufacturing by minimizing solvent usage and waste generation.
Mechanistic Insights into Chiral Titanium-Catalyzed Asymmetric Oxidation
The core of this technological advancement lies in the formation and function of the chiral titanium complex, which acts as the stereochemical director for the oxygen transfer event. The catalyst is generated in situ through the coordination of titanium tetraisopropoxide with chiral diethyl tartrate (DET) ligands, creating a rigid chiral pocket around the metal center. When the prochiral thioether substrate coordinates to this activated titanium species, the steric environment imposed by the tartrate ligands dictates the facial selectivity of the subsequent oxidation. The bulky dicumyl hydroperoxide molecule approaches the sulfur atom from a specific trajectory defined by the chiral catalyst, ensuring that oxygen is transferred exclusively to one face of the planar sulfide. This highly organized transition state is responsible for the observed enantiomeric excess values exceeding 99%, effectively distinguishing between the pro-R and pro-S pathways with remarkable precision.
Impurity control is another critical aspect governed by the mechanistic nuances of this catalytic system. In standard oxidation protocols, the initially formed sulfoxide is often more reactive towards further oxidation than the starting sulfide, leading to the accumulation of inactive sulfone impurities. However, the electronic and steric properties of the dicumyl hydroperoxide-titanium complex appear to modulate the reactivity such that the rate of the second oxidation step is significantly retarded. This kinetic discrimination prevents the over-oxidation of the valuable chiral sulfoxide product, preserving both chemical integrity and optical purity. Additionally, the use of bases like diisopropylethylamine helps to neutralize acidic by-products that could otherwise catalyze racemization of the sensitive sulfoxide bond. This comprehensive control over the reaction microenvironment ensures that the final product meets the rigorous specifications for high-purity proton pump inhibitors without the need for extensive recrystallization cycles.
How to Synthesize Chiral Imidazole Sulfoxides Efficiently
The practical implementation of this synthesis route involves a carefully orchestrated sequence of reagent addition and temperature control to maximize the efficiency of the chiral catalyst. Operators must first ensure the complete formation of the active titanium-tartrate species before introducing the oxidant, as premature addition can lead to non-selective background oxidation. The process typically begins with the suspension of the thioether precursor in an aromatic solvent like toluene, followed by the sequential addition of the chiral ligand and titanium source. Once the catalyst is activated, the reaction mixture is cooled to near-freezing temperatures to suppress thermal racemization and side reactions. This controlled environment allows for the slow, dropwise addition of the oxidant, ensuring that the concentration of reactive oxygen species remains low enough to favor mono-oxidation while maintaining high stereochemical fidelity throughout the batch.
- Prepare the chiral titanium catalyst by reacting titanium tetraisopropoxide with diethyl tartrate (DET) in toluene at elevated temperatures to form a transparent solution.
- Cool the reaction mixture to low temperatures (0-5°C) and add the prochiral thioether substrate along with a base such as diisopropylethylamine.
- Slowly add dicumyl hydroperoxide (DCHP) as the oxidant over several hours, maintaining strict temperature control to ensure high enantioselectivity and prevent over-oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits regarding cost structure and operational reliability. The elimination of inefficient resolution steps means that the theoretical yield ceiling is raised significantly, allowing manufacturers to produce more active ingredient from the same quantity of raw materials. This improvement in atom economy directly correlates to substantial cost savings in raw material procurement, as there is no longer a need to purchase double the amount of starting material to account for the discarded enantiomer. Furthermore, the simplified purification process reduces the consumption of expensive chromatography media and large volumes of organic solvents, lowering both direct material costs and environmental compliance expenses associated with waste disposal.
- Cost Reduction in Manufacturing: The transition to this asymmetric oxidation route removes the necessity for expensive chiral resolving agents and the associated recycling infrastructure required for racemic mixtures. By achieving high selectivity directly from the prochiral sulfide, the process minimizes the number of unit operations, such as repeated crystallizations or column chromatography, which are traditionally the most costly phases of API production. The use of commercially available reagents like dicumyl hydroperoxide and titanium tetraisopropoxide ensures that the input costs remain stable and predictable, avoiding the volatility associated with proprietary chiral auxiliaries. This streamlined workflow enables a leaner manufacturing model that significantly lowers the cost of goods sold (COGS) for high-volume PPI intermediates.
- Enhanced Supply Chain Reliability: Dependence on complex resolution technologies often introduces bottlenecks due to the limited availability of specific chiral acids or bases. In contrast, the reagents utilized in this novel method are commodity chemicals with robust global supply chains, ensuring consistent availability and reducing the risk of production stoppages. The high yield and purity achieved in the primary reaction step also mean that production schedules are more predictable, as there is less variability in batch outcomes that could delay downstream processing. This reliability is crucial for maintaining continuous supply to downstream formulation partners, especially for blockbuster drugs where market demand is constant and unforgiving of delays.
- Scalability and Environmental Compliance: The mild reaction conditions, operating between -20°C and 20°C, are easily manageable in standard stainless steel reactors without the need for specialized cryogenic equipment, facilitating straightforward scale-up from pilot to commercial production. The reduction in sulfone by-products and the avoidance of heavy metal catalysts or toxic resolving agents simplify the effluent treatment process, aligning the manufacturing footprint with increasingly strict environmental regulations. This green chemistry profile not only mitigates regulatory risk but also enhances the corporate sustainability metrics of the manufacturing entity, making the supply chain more resilient to future legislative changes regarding chemical waste and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and chemical principles involved. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. Engaging with these technical specifics allows stakeholders to make informed decisions about process validation and technology transfer.
Q: Why is dicumyl hydroperoxide preferred over cumene hydroperoxide in this synthesis?
A: Dicumyl hydroperoxide (DCHP) provides a milder oxidation environment that significantly reduces the formation of sulfone by-products, which are difficult to separate and lower the overall yield and optical purity of the final chiral sulfoxide.
Q: What represents the critical quality attribute for these proton pump inhibitor intermediates?
A: The critical quality attributes are high enantiomeric excess (ee > 99%) and high chemical purity (> 99%), as the biological activity of drugs like S-Omeprazole is strictly dependent on the specific stereochemistry of the sulfur center.
Q: How does this method improve scalability compared to traditional resolution methods?
A: Unlike chemical resolution which inherently wastes over 50% of the material as the unwanted enantiomer, this asymmetric oxidation converts the prochiral sulfide directly into the desired active isomer with high atom economy, simplifying downstream processing and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in the development of next-generation gastrointestinal therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate requirements of chiral titanium catalysis are met with precision at every batch size. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to verify that every shipment of Omeprazole or Pantoprazole intermediate meets the >99% ee benchmark required by global pharmacopeias. Our commitment to quality assurance ensures that your drug development timeline remains on track without the setbacks of failed purity tests.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-efficiency oxidation route for your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. By partnering with us, you secure a supply of high-purity pharmaceutical intermediates that are produced with both economic and environmental excellence in mind.
