Advanced Manufacturing of Allyl Benzenesulfonate: Technical Breakthroughs and Commercial Scalability
Advanced Manufacturing of Allyl Benzenesulfonate: Technical Breakthroughs and Commercial Scalability
The global demand for high-purity sulfonate esters, particularly Allyl Benzenesulfonate, has surged due to their critical role as selective allylation reagents and intermediates in complex organic synthesis. A pivotal advancement in this domain is detailed in patent CN114409574A, which outlines a robust, environmentally friendly preparation method that addresses long-standing inefficiencies in sulfonate ester production. This technology leverages a precise low-temperature esterification protocol using benzenesulfonyl chloride and allyl alcohol, mediated by liquid caustic soda under strictly controlled pH conditions. Unlike conventional approaches that often suffer from thermal instability and difficult purification, this novel route ensures product purity exceeding 98.5% and yields surpassing 97.5%. For R&D directors and procurement strategists, understanding the mechanistic nuances and operational advantages of this patent is essential for securing a reliable supply chain of this valuable fine chemical intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for allyl sulfonates frequently rely on harsh reaction conditions or expensive catalysts that complicate downstream processing. Historically, the esterification of sulfonyl chlorides with allylic alcohols has been plagued by the high reactivity of the sulfonyl chloride group, which is prone to rapid hydrolysis in the presence of moisture or excessive base. Conventional methods often utilize organic bases like pyridine or triethylamine to scavenge the generated hydrogen chloride; however, these reagents are costly, toxic, and difficult to remove completely from the final product, leading to impurity profiles that are unacceptable for pharmaceutical applications. Furthermore, the exothermic nature of the reaction, if not meticulously managed, can lead to thermal runaway, causing the decomposition of the sensitive allyl group and the formation of polymeric byproducts. These factors collectively result in lower overall yields, increased waste generation, and significant challenges in scaling up production to meet industrial demands without compromising safety or quality standards.
The Novel Approach
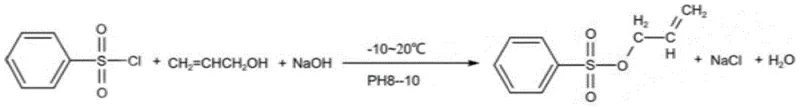
The methodology disclosed in patent CN114409574A represents a paradigm shift by replacing expensive organic bases with inexpensive liquid caustic soda (NaOH) while implementing rigorous thermal and pH controls. The core innovation lies in the dropwise addition of the alkali at temperatures ranging from -10°C to 20°C, which effectively manages the exotherm and suppresses the hydrolysis of the starting sulfonyl chloride. By maintaining the reaction system's pH between 8 and 10, the process ensures that the generated HCl is neutralized immediately to drive the equilibrium forward, without creating an overly alkaline environment that would trigger the degradation of the sulfonyl chloride into sulfonic acid. This approach not only simplifies the workup procedure—allowing for the easy filtration of sodium chloride byproduct—but also facilitates the recovery and reuse of excess allyl alcohol. The result is a streamlined, cost-effective process that delivers high-purity products suitable for sensitive applications in agrochemicals and pharmaceuticals, marking a significant improvement over legacy technologies.
Mechanistic Insights into Low-Temperature Esterification
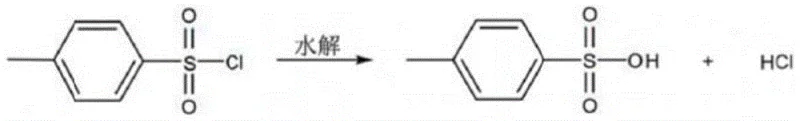
The success of this synthesis hinges on the delicate balance between nucleophilic attack and hydrolytic degradation. The reaction proceeds via a nucleophilic substitution mechanism where the oxygen atom of the allyl alcohol attacks the sulfur atom of the benzenesulfonyl chloride. This step generates a tetrahedral intermediate which subsequently collapses to release the chloride ion and form the sulfonate ester bond. However, the presence of water, either from the liquid alkali or the environment, introduces a competing pathway where hydroxide ions attack the sulfur center, leading to the formation of benzenesulfonic acid and sodium benzenesulfonate. This side reaction is thermodynamically favorable and kinetically fast, especially at elevated temperatures or high pH levels. The patent's strategy of operating at sub-ambient temperatures (-10°C to 20°C) drastically reduces the kinetic energy available for the hydrolysis pathway, thereby kinetically favoring the desired esterification. Additionally, the slow, dropwise addition of the base ensures that the local concentration of hydroxide ions never becomes high enough to overwhelm the system, preserving the integrity of the electrophilic sulfonyl chloride throughout the reaction duration.
Furthermore, the control of pH is critical in mitigating the formation of specific impurities that can compromise the utility of the final intermediate. As illustrated in the side reaction mechanisms, if the pH rises too high, the sulfonyl chloride undergoes rapid hydrolysis to form the corresponding sulfonic acid, which then converts to its sodium salt under alkaline conditions. This byproduct is difficult to separate from the desired ester and significantly lowers the isolated yield. The patent specifies maintaining a pH of 8-10, which is sufficiently basic to neutralize the stoichiometric HCl produced but not so basic as to accelerate the hydrolysis rate excessively. This precise window allows for the quantitative conversion of the starting material while minimizing the generation of acidic impurities. The subsequent refining steps, involving washing and vacuum dehydration at temperatures below 40°C, further ensure that no thermal decomposition occurs during isolation, preserving the labile allyl double bond and ensuring the structural fidelity of the molecule for downstream coupling reactions.
How to Synthesize Allyl Benzenesulfonate Efficiently
The synthesis protocol described in the patent offers a reproducible framework for manufacturing high-quality allyl benzenesulfonate. The process begins with the dissolution of the sulfonyl chloride in allyl alcohol, followed by the controlled addition of 30% liquid caustic soda under cooling. The reaction mixture is then warmed gently to complete the conversion, followed by a straightforward separation of the inorganic salt byproduct. The organic layer is washed to remove residual salts and base, and finally, the product is isolated via vacuum distillation to remove excess alcohol and water. This sequence is designed to maximize throughput while minimizing solvent usage and energy consumption. For detailed operational parameters and specific stoichiometric ratios validated by experimental data, please refer to the standardized synthesis guide below.
- Dissolve benzenesulfonyl chloride in allyl alcohol and cool the mixture to between -10°C and 20°C.
- Dropwise add liquid caustic soda (NaOH) while maintaining pH between 8 and 10 to neutralize generated HCl without causing hydrolysis.
- Filter off sodium chloride byproduct, wash the organic layer, and perform vacuum dehydration at temperatures below 40°C to recover the pure ester.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers substantial strategic benefits for procurement managers and supply chain leaders seeking to optimize their sourcing of fine chemical intermediates. The primary advantage lies in the drastic simplification of the raw material profile; by utilizing commodity chemicals like liquid caustic soda and allyl alcohol instead of specialized organic bases, the direct material costs are significantly reduced. Moreover, the ability to recover and recycle unreacted allyl alcohol from the aqueous waste stream creates a closed-loop system that minimizes raw material waste and lowers the effective cost per kilogram of the final product. This efficiency translates into a more competitive pricing structure for buyers without sacrificing the stringent purity requirements necessary for pharmaceutical and agrochemical synthesis. The process eliminates the need for complex chromatographic purification or extensive solvent exchanges, further reducing the operational expenditure associated with manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive organic amine catalysts and the use of aqueous sodium hydroxide fundamentally alters the cost structure of production. Since the byproduct is simple sodium chloride, which is easily filtered off, the downstream processing costs are minimized compared to methods that generate soluble amine hydrochloride salts requiring aqueous extraction. Additionally, the recovery of excess allyl alcohol means that the effective consumption of this reagent is lowered, contributing to substantial long-term savings. The mild reaction conditions also reduce energy consumption for heating and cooling, making the overall process more economically viable for large-scale production runs.
- Enhanced Supply Chain Reliability: The reliance on widely available, commodity-grade raw materials such as benzenesulfonyl chloride and allyl alcohol ensures a stable and resilient supply chain. Unlike processes dependent on niche reagents that may face availability fluctuations, the inputs for this synthesis are produced globally in high volumes. The robustness of the reaction, characterized by its tolerance to standard industrial equipment and lack of hazardous high-pressure steps, further guarantees consistent production schedules. This reliability is crucial for downstream manufacturers who require just-in-time delivery of high-purity intermediates to maintain their own production timelines without interruption.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at atmospheric pressure and moderate temperatures, removing the engineering barriers often associated with high-pressure hydrogenation or cryogenic reactions. The environmental footprint is significantly reduced as the process avoids the generation of toxic organic waste streams associated with amine bases. The primary waste products are saline water and recovered alcohol, both of which are manageable within standard wastewater treatment protocols. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making it an attractive option for companies aiming to reduce their environmental impact while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of allyl benzenesulfonate. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details helps in evaluating the feasibility of integrating this intermediate into your specific synthesis workflows.
Q: Why is temperature control critical in the synthesis of allyl benzenesulfonate?
A: Strict temperature control between -10°C and 20°C during the addition of alkali is essential to prevent the thermal decomposition of the sulfonyl chloride and to minimize the hydrolysis side reaction that forms sulfonic acid byproducts.
Q: How does this process improve yield compared to traditional methods?
A: By utilizing a controlled dropwise addition of liquid alkali and maintaining a specific pH range of 8-10, the process ensures complete conversion of the starting material while suppressing side reactions, resulting in yields exceeding 97.5%.
Q: Can the excess allyl alcohol be recovered in this manufacturing process?
A: Yes, the process includes a dedicated recovery step where the aqueous layer is neutralized and distilled, allowing for the efficient recycling of unreacted allyl alcohol, which significantly reduces raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Benzenesulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and supply continuity for key intermediates like Allyl Benzenesulfonate. As a leading CDMO and manufacturer, we have successfully integrated advanced synthetic methodologies, including the low-temperature esterification techniques described in recent patents, into our production facilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project phase. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art GC and HPLC systems to guarantee that every batch meets the >98.5% purity threshold required for sensitive pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for this essential building block. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient manufacturing processes can lower your total cost of ownership. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us be your partner in driving innovation and efficiency in your chemical synthesis projects.
