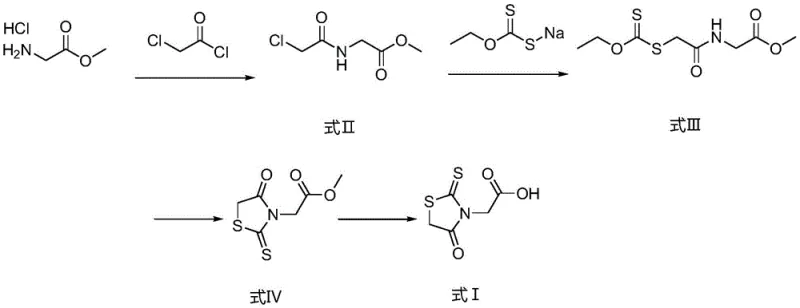
Advanced Manufacturing of Epalrestat Intermediate: A Safer, Scalable Synthetic Route
The pharmaceutical industry constantly seeks robust synthetic pathways that balance efficiency with stringent safety standards, particularly for critical diabetes management compounds. Patent CN114163396B introduces a transformative preparation method for an epalrestat intermediate, addressing long-standing safety and scalability issues inherent in traditional manufacturing. This technical breakthrough replaces the hazardous use of carbon disulfide with a safer, solid-state reagent system, fundamentally altering the risk profile of the supply chain. By leveraging nucleophilic substitution and cyclization reactions under controlled conditions, this method ensures that every intermediate generated is a solid, significantly enhancing quality control capabilities. For global procurement teams, this represents a shift towards more reliable sourcing of high-purity pharmaceutical intermediates, mitigating the regulatory and logistical burdens associated with volatile reagents. The strategic implementation of this technology underscores a commitment to sustainable and safe chemical manufacturing practices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of epalrestat intermediates has relied heavily on glycine, carbon disulfide, and alpha-methyl cinnamaldehyde as primary starting materials. The reliance on carbon disulfide presents a formidable challenge for industrial operations due to its extreme volatility, flammability, and explosive nature. These characteristics necessitate specialized transportation equipment and bulk storage facilities that are not only costly but also pose significant safety risks to personnel and infrastructure. Furthermore, the handling of such hazardous gases complicates the regulatory compliance landscape, often leading to delays in production schedules and increased insurance liabilities. The difficulty in realizing safe industrialization production using these conventional reagents has long been a bottleneck for manufacturers aiming to scale up efficiently. Consequently, the industry has faced persistent pressure to identify alternative synthetic routes that do not compromise on yield or purity while eliminating these inherent safety hazards.
The Novel Approach
The innovative methodology disclosed in the patent circumvents these dangers by utilizing sodium ethyl xanthate, a reagent that is easy to obtain, industrialized, and convenient to store. This substitution marks a pivotal shift from gas-phase hazards to solid-phase handling, drastically simplifying the operational workflow. The reaction equation demonstrates a clear, multi-step progression where safety is engineered into the chemistry itself. By avoiding inflammable and explosive reagents, the process opens new avenues for cost reduction in pharma manufacturing without sacrificing chemical integrity. The ability to manage reagents as solids rather than volatile gases allows for more precise dosing and reaction monitoring, which is critical for maintaining batch-to-batch consistency. This approach not only enhances the safety profile of the facility but also streamlines the supply chain by reducing the need for specialized hazard containment protocols.
Mechanistic Insights into Nucleophilic Substitution and Cyclization
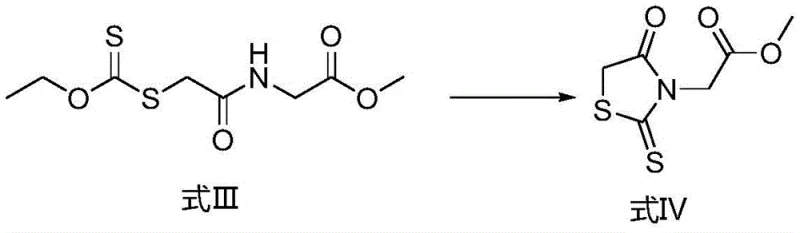
The core of this synthetic strategy lies in a meticulously orchestrated series of nucleophilic substitution reactions followed by a base-catalyzed cyclization. Initially, glycine methyl ester hydrochloride undergoes acylation with chloroacetyl chloride in the presence of an alkali base, typically sodium bicarbonate, to form the chloroacetamide derivative known as Formula II. This step is critical as it establishes the carbon backbone required for subsequent heterocyclic formation. The reaction is conducted in a biphasic system of dichloromethane and water, which facilitates the separation of the organic product from inorganic salts. Following this, the introduction of sodium ethyl xanthate triggers a second nucleophilic substitution, displacing the chloride atom to install the dithiocarbonate moiety found in Formula III. This transformation is conducted in an aqueous environment where the product precipitates as a solid, allowing for immediate isolation and purification. The final cyclization step employs potassium tert-butoxide in tetrahydrofuran to close the rhodanine ring, forming Formula IV, which is subsequently hydrolyzed to yield the target carboxylic acid.
Impurity control is inherently built into this mechanism through the physical state of the intermediates. Because Formula II, Formula III, and Formula IV are all isolated as solids, they can be subjected to rigorous washing and recrystallization processes that are impossible with oily or gaseous intermediates. This solid-state progression ensures that side products and unreacted starting materials are physically removed before they can propagate through the synthesis. For R&D directors focused on purity and impurity profiles, this means a cleaner final API with reduced downstream purification burden. The use of specific bases like potassium tert-butoxide versus sodium methoxide has been empirically optimized to minimize specific impurity peaks, as demonstrated in the patent's comparative data. Such mechanistic precision ensures that the commercial scale-up of complex pharmaceutical intermediates remains feasible and robust, meeting the stringent specifications required by regulatory bodies worldwide.
How to Synthesize Epalrestat Intermediate Efficiently
The standardized synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-efficiency route in a GMP environment. The process begins with the careful control of temperature and pH during the initial acylation to prevent hydrolysis of the ester group. Detailed operational parameters, including solvent ratios and addition rates, are critical for maximizing yield and minimizing byproduct formation. The subsequent substitution and cyclization steps require inert atmosphere protection to prevent oxidation of the sulfur-containing species. While the general flow is straightforward, strict adherence to the specified workup procedures, such as nitrogen pressure filtration and specific drying temperatures, is essential for maintaining product quality. The detailed standardized synthesis steps see the guide below for exact operational parameters.
- Perform nucleophilic substitution between glycine methyl ester hydrochloride and chloroacetyl chloride using sodium bicarbonate in a dichloromethane-water system to obtain Formula II.
- React Formula II with sodium ethyl xanthate in an aqueous environment to generate the solid intermediate Formula III through substitution.
- Execute ring closure on Formula III using potassium tert-butoxide in tetrahydrofuran to form Formula IV, followed by acid hydrolysis to yield the final product Formula I.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that extend beyond mere chemical elegance to impact the bottom line and supply chain resilience. The elimination of carbon disulfide removes a major logistical bottleneck, as sourcing and transporting this hazardous material often involves complex permitting and specialized logistics providers. By switching to sodium ethyl xanthate, manufacturers can leverage a more stable and widely available supply chain, reducing lead time for high-purity pharmaceutical intermediates. The fact that all intermediates are solids simplifies warehousing and inventory management, as there is no need for pressurized tanks or gas detection systems. This structural change in the manufacturing process translates directly into enhanced supply chain reliability, ensuring that production schedules are not disrupted by regulatory hurdles or safety incidents. Furthermore, the simplified purification process reduces solvent consumption and waste generation, aligning with modern environmental compliance standards.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous reagents like carbon disulfide significantly lowers the raw material costs associated with safety containment and disposal. Additionally, the ability to isolate intermediates as solids reduces the need for complex chromatographic purification steps, which are often the most expensive part of fine chemical manufacturing. The streamlined workflow minimizes labor hours and equipment downtime, leading to substantial cost savings over the lifecycle of the product. By optimizing the reaction conditions to favor solid precipitation, the process also reduces solvent usage, further driving down operational expenditures. These efficiencies collectively contribute to a more competitive pricing structure for the final intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing stable, solid reagents mitigates the risk of supply disruptions caused by transportation restrictions on hazardous goods. Sodium ethyl xanthate is a commodity chemical with a robust global supply network, ensuring consistent availability even during market fluctuations. The improved safety profile of the plant allows for higher throughput and reduced insurance premiums, which stabilizes the overall cost of goods sold. Moreover, the ease of handling solid intermediates facilitates faster turnaround times between batches, enabling manufacturers to respond more agilely to changes in demand. This reliability is crucial for long-term partnerships where continuity of supply is a primary contractual obligation.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The avoidance of toxic gases simplifies the environmental permitting process, allowing for faster deployment of new production lines. Waste streams are easier to treat since they do not contain volatile sulfur compounds, reducing the burden on effluent treatment plants. This alignment with green chemistry principles not only satisfies regulatory requirements but also enhances the corporate sustainability profile of the manufacturer. As global regulations tighten around emissions and worker safety, this scalable and compliant methodology positions the supply chain for future-proof operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this method for their own production lines. The clarity provided here aims to bridge the gap between academic innovation and industrial application.
Q: Why is the sodium ethyl xanthate route preferred over traditional carbon disulfide methods?
A: Traditional methods rely on carbon disulfide, which is highly volatile, flammable, and explosive, posing severe safety risks during transport and storage. The novel route utilizes sodium ethyl xanthate, a stable solid that eliminates these hazards while simplifying industrial handling and quality control.
Q: How does this process improve impurity control for pharmaceutical applications?
A: A key advantage of this methodology is that all intermediate products obtained throughout the multi-step reaction are solids. This physical state facilitates rigorous quality control, filtration, and washing, ensuring that impurities are effectively removed before proceeding to the next stage, resulting in higher purity final APIs.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial feasibility. By avoiding hazardous gases and utilizing stable solid intermediates, the reaction conditions are safer and more manageable at scale. The use of common solvents like dichloromethane and tetrahydrofuran further supports seamless integration into existing manufacturing infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epalrestat Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for vital pharmaceutical intermediates. Our team of expert chemists has extensively evaluated the methodology described in CN114163396B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the solid-state quality of every intermediate batch. We are committed to delivering high-purity epalrestat intermediate that meets the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this CS2-free process. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Together, we can build a more resilient and sustainable supply chain for diabetes therapeutics.
