Advanced Aromatic Methyl Ether Production: High-Purity Pharma Intermediates via Recycled Monochloromethane
The chemical industry is currently witnessing a paradigm shift towards sustainable synthesis routes that prioritize both atomic economy and environmental compliance, a transition vividly exemplified by the technological breakthroughs detailed in patent CN103012074A. This specific intellectual property outlines a robust method for preparing aromatic methyl ether compounds, which serve as critical building blocks in the manufacturing of complex active pharmaceutical ingredients (APIs) and fine chemicals. By leveraging a phase transfer catalysis system coupled with a unique raw material recycling strategy, this process addresses the longstanding challenges of waste emission and energy inefficiency inherent in traditional etherification methods. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential, as it offers a pathway to secure a reliable pharma intermediates supplier status while drastically reducing the environmental footprint of production facilities. The core innovation lies not merely in the reaction conditions but in the integration of waste gas recovery from 1,2-benzo-isothiazolin-3-one (BIT) production, creating a closed-loop system that transforms a hazardous byproduct into a high-value reagent. This report delves deep into the mechanistic and commercial implications of this technology, providing a comprehensive analysis for stakeholders aiming to optimize their supply chains for high-purity aromatic methyl ethers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of aromatic methyl ether compounds has relied heavily on methods that pose significant safety hazards and environmental burdens, primarily utilizing dimethyl sulfate or methyl halides under extreme conditions. Traditional processes often require reaction temperatures exceeding 190°C to 200°C, as documented in early chemical literature, which necessitates expensive high-pressure equipment and consumes substantial energy resources, thereby inflating the overall cost reduction in pharma intermediates manufacturing. Furthermore, the use of sodium metal or liquefied ammonia in older methodologies introduces severe safety risks due to the pyrophoric nature of the reagents and the potential for runaway reactions, making these routes unsuitable for modern, large-scale commercial operations. Another critical drawback of conventional techniques is the poor atom economy and the generation of toxic waste streams, particularly when using methylating agents that produce stoichiometric amounts of inorganic salts which are difficult and costly to dispose of in compliance with stringent environmental regulations. The low selectivity and yield often associated with these high-temperature processes also result in complex purification steps, further eroding profit margins and extending the lead time for high-purity aromatic methyl ethers delivery to end-users. Consequently, the industry has been in urgent need of a milder, safer, and more economically viable alternative that does not compromise on the quality or scalability of the final product.
The Novel Approach
The methodology disclosed in CN103012074A represents a significant technological leap by introducing a moderate-temperature, phase-transfer catalyzed reaction system that operates efficiently between 60°C and 100°C under pressures of 0.5 MPa to 2.5 MPa. This novel approach fundamentally changes the economic equation by utilizing monochloromethane recovered from the exhaust gas of 1,2-benzo-isothiazolin-3-one (BIT) production, effectively turning a waste management liability into a valuable raw material asset. 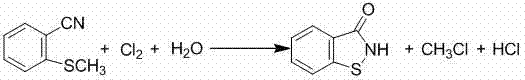 By integrating this recycling loop, the process not only solves the problem of exhaust emission in BIT manufacturing but also secures a low-cost source of methylating agent, significantly enhancing the supply chain reliability for downstream chemical synthesis. The addition of a phase transfer catalyst, such as tetrabutyl ammonium bromide, facilitates the interaction between the aqueous alkali phase and the organic phenol phase, dramatically increasing the reaction conversion ratio and selectivity without the need for extreme thermal energy. This results in a streamlined production workflow where the aromatic methyl ether compound purity can reach more than 99.5% with reaction yields consistently ranging from 95% to 97%, demonstrating very high value in industrialization. The ability to operate under such moderate conditions while achieving superior yields makes this method a prime candidate for the commercial scale-up of complex polymer additives and pharmaceutical intermediates.
By integrating this recycling loop, the process not only solves the problem of exhaust emission in BIT manufacturing but also secures a low-cost source of methylating agent, significantly enhancing the supply chain reliability for downstream chemical synthesis. The addition of a phase transfer catalyst, such as tetrabutyl ammonium bromide, facilitates the interaction between the aqueous alkali phase and the organic phenol phase, dramatically increasing the reaction conversion ratio and selectivity without the need for extreme thermal energy. This results in a streamlined production workflow where the aromatic methyl ether compound purity can reach more than 99.5% with reaction yields consistently ranging from 95% to 97%, demonstrating very high value in industrialization. The ability to operate under such moderate conditions while achieving superior yields makes this method a prime candidate for the commercial scale-up of complex polymer additives and pharmaceutical intermediates.
Mechanistic Insights into Phase Transfer Catalyzed Etherification
At the heart of this synthesis lies a sophisticated phase transfer catalysis (PTC) mechanism that overcomes the kinetic barriers typically associated with gas-liquid-solid multiphase reactions. In this system, the quaternary ammonium salt catalyst acts as a molecular shuttle, transporting hydroxide ions from the aqueous caustic alkali phase into the organic phase where the aromatic phenol substrate resides. This transport mechanism generates a highly reactive phenoxide anion in the organic layer, which then readily undergoes nucleophilic substitution with the monochloromethane gas dissolved in the medium. The efficiency of this process is heavily dependent on the specific structure of the phenol substrate, as defined by the general formula where R1 through R5 can represent hydrogen, halogen atoms, or various alkyl chains. 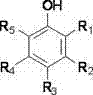 The versatility of this mechanism allows for the successful methylation of a wide range of substrates, including phenol, various cresols, and halophenols, without significant degradation of the catalyst or the formation of unwanted byproducts. The presence of water in the reaction mixture, maintained at a specific weight ratio to the caustic alkali, is crucial for maintaining the ionic conductivity and facilitating the phase transfer cycle, ensuring that the reaction proceeds smoothly to completion. This mechanistic understanding is vital for R&D teams aiming to adapt this process for new derivatives, as it highlights the importance of catalyst selection and phase ratios in optimizing the commercial scale-up of complex aromatic methyl ethers.
The versatility of this mechanism allows for the successful methylation of a wide range of substrates, including phenol, various cresols, and halophenols, without significant degradation of the catalyst or the formation of unwanted byproducts. The presence of water in the reaction mixture, maintained at a specific weight ratio to the caustic alkali, is crucial for maintaining the ionic conductivity and facilitating the phase transfer cycle, ensuring that the reaction proceeds smoothly to completion. This mechanistic understanding is vital for R&D teams aiming to adapt this process for new derivatives, as it highlights the importance of catalyst selection and phase ratios in optimizing the commercial scale-up of complex aromatic methyl ethers.
Controlling the impurity profile in this synthesis is achieved through the precise regulation of reaction parameters, specifically the molar ratio of the aromatic phenol compound to the caustic alkali and monochloromethane, which is maintained at 1:(1.0-2.0):(1.0-5.0). By strictly adhering to these stoichiometric boundaries, the process minimizes the formation of poly-alkylated byproducts and ensures that the unreacted phenol is kept to a negligible level, simplifying the downstream purification process. The moderate temperature range of 60°C to 100°C further contributes to impurity control by preventing thermal decomposition of the sensitive ether linkage or the catalyst itself, which is a common issue in high-temperature conventional methods. Additionally, the use of recycled monochloromethane requires careful pre-treatment to remove acidic impurities like hydrogen chloride, which is achieved through absorption in sodium hydroxide solution before the gas enters the reactor, thereby protecting the catalyst from deactivation. This rigorous attention to detail in the reaction design ensures that the final product meets the stringent purity specifications required for pharmaceutical applications, where even trace impurities can have significant regulatory implications. The result is a robust process capable of delivering high-purity aromatic methyl ethers with a consistent quality profile that satisfies the most demanding global compliance standards.
How to Synthesize Aromatic Methyl Ether Efficiently
Implementing this synthesis route requires a systematic approach to reactor setup and process control to fully realize the benefits of the phase transfer catalysis system. The detailed standardized synthesis steps involve charging a stainless steel autoclave with the phenol substrate, caustic alkali solution, and the phase transfer catalyst, followed by the controlled introduction of monochloromethane gas under pressure. Operators must maintain strict vigilance over the temperature and pressure profiles to ensure the reaction stays within the optimal window of 60°C to 100°C and 0.5 MPa to 2.5 MPa for the designated duration of 2 to 8 hours.
- Prepare the reaction mixture by combining aromatic phenol compounds with caustic alkali and water in a stainless steel autoclave equipped with agitation.
- Introduce the phase transfer catalyst, such as tetrabutyl ammonium bromide, to facilitate the gas-liquid two-phase reaction efficiency.
- Pressurize the system with monochloromethane gas and maintain the temperature between 60°C to 100°C for 2 to 8 hours to achieve high conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers substantial strategic advantages that extend far beyond simple chemical transformation. The primary benefit is the drastic simplification of raw material sourcing, as the monochloromethane is derived from the recycling of industrial waste gas, effectively decoupling the production cost from the volatile market prices of virgin methylating agents. This integration creates a symbiotic relationship between BIT production and ether synthesis, ensuring a stable and continuous supply of key reagents that enhances supply chain reliability and reduces the risk of production stoppages due to raw material shortages. Furthermore, the elimination of toxic reagents like dimethyl sulfate and the reduction of hazardous waste streams significantly lower the operational costs associated with environmental compliance and waste disposal, contributing to overall cost reduction in pharma intermediates manufacturing. The moderate reaction conditions also translate to lower energy consumption and reduced wear and tear on production equipment, extending the lifespan of capital assets and minimizing maintenance downtime. These factors combined create a highly competitive cost structure that allows manufacturers to offer high-purity aromatic methyl ethers at more attractive price points while maintaining healthy profit margins.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the utilization of recycled monochloromethane, which effectively negates the purchase cost of this primary raw material and transforms a waste treatment expense into a revenue-generating opportunity. By eliminating the need for expensive and hazardous methylating agents like dimethyl sulfate, the process removes the associated costs of specialized handling, storage, and neutralization of toxic byproducts, leading to significant operational savings. The high reaction yield of 95% to 97% ensures that raw material utilization is maximized, reducing the amount of feedstock required per unit of product and minimizing the volume of waste that requires disposal. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further lowering the utility costs associated with large-scale production runs. These cumulative savings allow for a more aggressive pricing strategy in the market while preserving the financial health of the manufacturing operation.
- Enhanced Supply Chain Reliability: The integration of waste gas recycling from BIT production creates a self-sustaining raw material loop that insulates the supply chain from external market fluctuations and availability issues common with commodity chemicals. Since the monochloromethane is generated on-site or sourced from a dedicated partner facility as a byproduct, the risk of supply disruption due to upstream manufacturing issues or logistics bottlenecks is significantly mitigated. This vertical integration ensures a consistent flow of materials, allowing for more accurate production planning and the ability to meet tight delivery deadlines for critical pharmaceutical intermediates. The robustness of the phase transfer catalysis system also means that the process is less sensitive to minor variations in raw material quality, further stabilizing the production output. For supply chain heads, this reliability translates into reduced safety stock requirements and improved cash flow management, as the need for emergency sourcing is virtually eliminated.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard stainless steel autoclaves and moderate pressure ratings that are common in existing chemical infrastructure, facilitating easy commercial scale-up of complex aromatic methyl ethers without massive capital expenditure. The zero-emission production technology achieved through the effective recycling of monochloromethane and the absorption of acidic gases ensures full compliance with increasingly stringent global environmental regulations, avoiding potential fines and production bans. The absence of heavy metal catalysts or persistent organic pollutants in the waste stream simplifies the wastewater treatment process, reducing the environmental footprint of the facility. This alignment with green chemistry principles not only satisfies regulatory bodies but also enhances the brand reputation of the manufacturer among eco-conscious clients and investors. The combination of easy scalability and environmental safety makes this technology a future-proof solution for long-term industrial growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this aromatic methyl ether synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: How does this method improve upon traditional dimethyl sulfate etherification?
A: This method eliminates the use of toxic dimethyl sulfate and high-temperature conditions, utilizing recycled monochloromethane instead for a safer, zero-emission process with yields exceeding 95%.
Q: What is the source of the monochloromethane used in this synthesis?
A: The monochloromethane is recovered from the waste gas emissions of 1,2-benzo-isothiazolin-3-one (BIT) production, turning a pollutant into a valuable raw material.
Q: What purity levels can be achieved with this phase transfer catalysis method?
A: The process consistently achieves aromatic methyl ether purity greater than 99.5% with reaction yields between 95% and 97%, suitable for high-specification pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Methyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies like the one described in CN103012074A to maintain competitiveness in the global fine chemicals market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs that guarantee every batch of aromatic methyl ether meets the highest international standards for pharmaceutical and agrochemical applications. We understand that the successful implementation of this phase transfer catalysis method requires precise engineering and process control, areas where our technical team excels through years of hands-on experience with complex gas-liquid reactions. By partnering with us, clients gain access to a supply chain that is not only cost-effective but also resilient and environmentally responsible, aligning with the modern demands of sustainable manufacturing.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this recycled monochloromethane process for your specific product line. We encourage you to reach out for specific COA data and route feasibility assessments to verify the compatibility of this technology with your existing infrastructure and quality requirements. Our team is ready to provide the technical support and commercial flexibility needed to optimize your supply chain and secure a competitive advantage in the market for high-purity aromatic methyl ethers.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
