Advanced Synthesis of 4-Benzyloxy Phenyl Ethyl N-Decanoate for Commercial Scale-Up
Advanced Synthesis of 4-Benzyloxy Phenyl Ethyl N-Decanoate for Commercial Scale-Up
The global demand for high-performance thermochromic materials, particularly for applications in erasable pen inks and smart packaging, necessitates robust and scalable synthetic routes for key additives. Patent CN110818560A introduces a significant technological breakthrough in the preparation of 4-benzyloxy phenyl ethyl n-decanoate, a critical thermochromic assistant. This innovation addresses long-standing challenges in ester synthesis by replacing hazardous and unstable acyl chloride intermediates with a direct, green esterification protocol. By leveraging p-toluenesulfonic acid catalysis combined with in-situ dehydration strategies, the disclosed method achieves high purity and yield while operating under remarkably mild thermal conditions. For R&D directors and procurement specialists seeking a reliable thermochromic additive supplier, this patent represents a pivotal shift towards more sustainable and cost-effective manufacturing paradigms in the specialty chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex esters like 4-benzyloxy phenyl ethyl n-decanoate has relied heavily on the Schotten-Baumann reaction or similar acylation protocols involving acid chlorides, specifically n-decanoyl chloride. While chemically feasible, these conventional pathways are plagued by significant operational drawbacks that hinder large-scale commercial viability. The background art indicates that reactions involving n-decanoyl chloride are often sluggish and prone to catalyst inactivation, leading to inconsistent batch quality and extended processing times. Furthermore, the handling of acid chlorides requires stringent safety measures due to their corrosive nature and the generation of hazardous hydrogen chloride byproducts, which complicates waste treatment and increases the overall environmental footprint of the manufacturing process. These factors collectively contribute to higher production costs and supply chain vulnerabilities for manufacturers of printing inks and functional additives.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent utilizes a direct Fischer esterification between 4-benzyloxy phenethyl alcohol and n-decanoic acid. This strategy eliminates the need for pre-activating the carboxylic acid into a chloride, thereby streamlining the raw material supply chain and reducing hazard exposure. The core innovation lies in the precise control of reaction parameters, specifically maintaining temperatures between 30-70°C and employing a solid desiccant such as anhydrous magnesium sulfate or calcium chloride. This combination effectively drives the equilibrium forward by removing water without subjecting the sensitive alcohol substrate to the high temperatures that typically induce intramolecular dehydration side reactions. 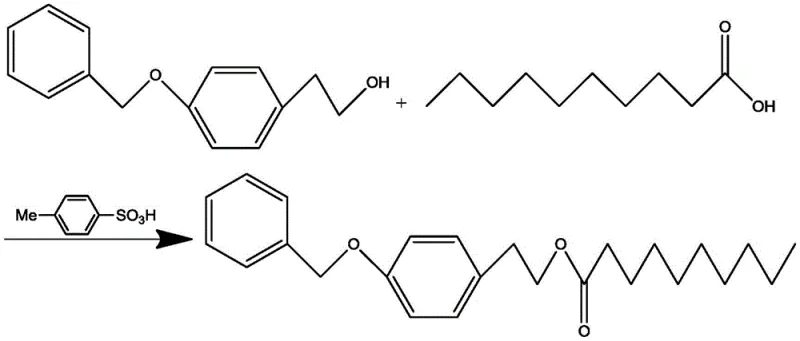
Mechanistic Insights into p-Toluenesulfonic Acid Catalyzed Esterification
The mechanistic elegance of this process relies on the dual function of the catalyst and the dehydrating agent to overcome thermodynamic barriers. P-toluenesulfonic acid (TsOH) acts as a strong organic soluble acid, protonating the carbonyl oxygen of the n-decanoic acid to increase its electrophilicity, thereby facilitating nucleophilic attack by the hydroxyl group of the 4-benzyloxy phenethyl alcohol. Unlike mineral acids which might promote charring or excessive side reactions, TsOH offers a balanced acidity profile suitable for sensitive substrates. Crucially, the presence of the desiccant serves to sequester the water molecule produced during the condensation, shifting the chemical equilibrium towards the product side according to Le Chatelier's principle. This ensures that the reaction proceeds to near-completion without requiring excessive excesses of reagents or harsh distillation conditions that could degrade the thermochromic properties of the final molecule.
Impurity control is another critical aspect where this mechanism excels, particularly regarding the suppression of olefin formation. Under standard acid-catalyzed conditions at elevated temperatures, benzylic alcohols are susceptible to elimination reactions, resulting in styrene-like impurities that can discolor the final ink formulation. By capping the reaction temperature at a maximum of 70°C and utilizing the desiccant to lower the effective activity of water, the kinetic pathway for elimination is effectively shut down in favor of esterification. This results in a cleaner crude product profile, which significantly reduces the burden on downstream purification steps such as recrystallization. For quality assurance teams, this translates to a more consistent impurity profile and higher confidence in the batch-to-batch reproducibility of the high-purity thermochromic additive.
How to Synthesize 4-Benzyloxy Phenyl Ethyl N-Decanoate Efficiently
The operational procedure for this synthesis is designed for straightforward implementation in standard glass-lined or stainless steel reactors commonly found in fine chemical facilities. The process begins with the charging of the alcohol and acid substrates into a solvent system, typically toluene, followed by the addition of the catalyst and the drying agent. The reaction is then heated gently to the optimized range of 40-70°C and maintained for a duration of approximately 12 hours to ensure full conversion. Detailed standardized synthesis steps, including specific workup procedures involving sodium carbonate washing and hexane recrystallization, are outlined below to guide process engineers in replicating these results.
- Charge the reactor with 4-benzyloxy phenethyl alcohol, n-decanoic acid, toluene solvent, p-toluenesulfonic acid catalyst, and a solid desiccant like anhydrous magnesium sulfate.
- Heat the reaction mixture to a mild temperature range of 40-70°C and maintain stirring for approximately 12 hours to ensure complete conversion while avoiding dehydration side reactions.
- Quench the reaction with saturated sodium carbonate solution, separate the organic layer, dry over anhydrous magnesium sulfate, remove solvent, and recrystallize the crude product using n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented methodology offers substantial benefits for procurement managers aiming to optimize the cost structure of specialty chemical manufacturing. By shifting from acid chlorides to free carboxylic acids, the process leverages n-decanoic acid, a commodity chemical with a stable and abundant global supply, rather than the more specialized and volatile n-decanoyl chloride. This substitution not only lowers the direct material costs but also mitigates the risks associated with the storage and transport of hazardous acylating agents. Furthermore, the simplified workflow reduces the number of unit operations required, leading to significant operational expenditure savings and a smaller physical footprint for production facilities.
- Cost Reduction in Manufacturing: The elimination of the acid chloride activation step removes the need for expensive chlorinating reagents and the associated scrubbing systems for hydrogen chloride gas. This qualitative shift in the synthetic route drastically simplifies the plant requirements and lowers the barrier to entry for production. Additionally, the use of recoverable solvents like toluene and the ability to recycle the desiccant or use low-cost inorganic salts further contributes to a leaner cost base, allowing for more competitive pricing in the thermochromic additive market.
- Enhanced Supply Chain Reliability: Relying on n-decanoic acid and 4-benzyloxy phenethyl alcohol ensures a robust supply chain, as these precursors are produced by multiple vendors globally, reducing dependency on single-source suppliers. The mild reaction conditions also imply less stress on equipment, leading to reduced maintenance downtime and higher asset utilization rates. For supply chain heads, this means a more predictable production schedule and the ability to scale output rapidly to meet fluctuating market demands for erasable ink components without facing raw material bottlenecks.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this process, such as atom economy and the avoidance of corrosive byproducts, align perfectly with modern environmental regulations. The absence of HCl emissions simplifies permitting and reduces the cost of waste neutralization and disposal. As production scales from pilot batches to multi-ton commercial runs, the safety profile remains favorable, minimizing the risk of thermal runaways and ensuring compliance with increasingly stringent industrial safety standards across different jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this thermochromic ester. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on process robustness and product specifications. Understanding these details is essential for technical teams evaluating the feasibility of integrating this material into their existing formulations or production lines.
Q: Why is p-toluenesulfonic acid preferred over traditional acyl chloride methods for this synthesis?
A: Traditional methods using n-decanoyl chloride often suffer from slow reaction rates and catalyst inactivation. The patented method utilizes direct esterification with n-decanoic acid under mild acidic conditions, which significantly simplifies the process and improves stability.
Q: How does the process prevent intramolecular dehydration side reactions?
A: The process strictly controls the reaction temperature between 30-70°C and employs a solid desiccant. This prevents the high-temperature conditions that typically trigger the unwanted dehydration of the benzyloxy phenethyl alcohol substrate.
Q: What are the key raw materials required for this thermochromic additive?
A: The primary feedstocks are 4-benzyloxy phenethyl alcohol and n-decanoic acid (capric acid), utilizing toluene as the solvent and p-toluenesulfonic acid as the catalyst, all of which are readily available industrial chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Benzyloxy Phenyl Ethyl N-Decanoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of end-user products like erasable pens and smart labels. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4-benzyloxy phenyl ethyl n-decanoate meets the exacting standards required for thermochromic applications, delivering consistency that your R&D team can trust.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and product quality.
