Advanced Titanium-Catalyzed Synthesis of Dexlansoprazole for Commercial API Production
Introduction to Patent CN103694225A and Technological Breakthrough
The pharmaceutical industry continuously seeks robust synthetic routes for proton pump inhibitors, specifically focusing on the production of high-purity dexlansoprazole intermediates. Patent CN103694225A introduces a groundbreaking methodology utilizing a novel titanium (IV) catalyst ligand, chemically identified as (1S,2S)-N-p-toluenesulfonic acid-1,2-diphenylethylenediamine. This innovation addresses critical stability issues found in previous generations of asymmetric catalysis, offering a pathway that consistently delivers enantiomeric excess values ≥99.2% and yields ≥80%. For R&D directors and procurement specialists, this represents a pivotal shift from unstable, low-yielding processes to a reliable, industrially viable protocol. The technology leverages a specific coordination chemistry between the sulfonamide-based ligand and titanium tetraisopropoxide, creating a catalytic environment that is far less sensitive to thermal variations than traditional tartrate-based systems. By integrating this advanced catalytic approach, manufacturers can achieve substantial improvements in both process efficiency and product quality, ensuring a steady supply of high-purity pharmaceutical intermediates for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of dexlansoprazole has been plagued by inefficiencies inherent in two primary methodologies: racemate resolution and early-generation asymmetric oxidation. The resolution pathway, often depicted in legacy literature, involves synthesizing the racemic mixture first and then separating the enantiomers through chemical or enzymatic means. 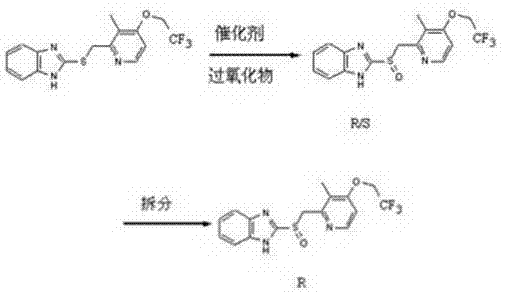 As detailed in the background art, chemical resolution methods frequently suffer from theoretical yield limitations, rarely exceeding 50%, with practical yields often dropping to 40-50% due to processing losses. Furthermore, enzymatic resolution, while selective, imposes harsh reaction conditions and prohibitive enzyme costs that render it economically unfeasible for large-scale operations. Similarly, earlier asymmetric catalytic attempts using titanium salts complexed with L-diethyl tartrate faced severe stability challenges; the Ti-O bonds in these complexes are notoriously fragile, leading to catalyst deactivation during the exothermic oxidation phase. This instability results in inconsistent optical purity and poor conversion rates, sometimes as low as 23.2%, creating significant waste and supply chain volatility for API manufacturers relying on these outdated techniques.
As detailed in the background art, chemical resolution methods frequently suffer from theoretical yield limitations, rarely exceeding 50%, with practical yields often dropping to 40-50% due to processing losses. Furthermore, enzymatic resolution, while selective, imposes harsh reaction conditions and prohibitive enzyme costs that render it economically unfeasible for large-scale operations. Similarly, earlier asymmetric catalytic attempts using titanium salts complexed with L-diethyl tartrate faced severe stability challenges; the Ti-O bonds in these complexes are notoriously fragile, leading to catalyst deactivation during the exothermic oxidation phase. This instability results in inconsistent optical purity and poor conversion rates, sometimes as low as 23.2%, creating significant waste and supply chain volatility for API manufacturers relying on these outdated techniques.
The Novel Approach
In stark contrast to these conventional limitations, the novel approach described in the patent utilizes a specialized diamine ligand that fundamentally alters the stability profile of the titanium catalyst. By employing (1S,2S)-N-p-toluenesulfonic acid-1,2-diphenylethylenediamine, the process establishes a robust Ti-N coordination bond that withstands the rigors of industrial oxidation conditions.  This structural integrity allows the reaction to proceed at moderate temperatures (50-60°C) without the rapid degradation observed in tartrate-based systems. The result is a dramatic enhancement in process performance, with conversion rates consistently surpassing 90% and isolated yields stabilizing above 80%. For a reliable dexlansoprazole intermediate supplier, this methodology eliminates the need for complex downstream purification steps associated with racemate splitting, thereby streamlining the entire manufacturing workflow. The ability to directly synthesize the desired R-enantiomer with high fidelity not only reduces raw material consumption but also minimizes the generation of hazardous waste, aligning perfectly with modern green chemistry principles and cost reduction in API manufacturing initiatives.
This structural integrity allows the reaction to proceed at moderate temperatures (50-60°C) without the rapid degradation observed in tartrate-based systems. The result is a dramatic enhancement in process performance, with conversion rates consistently surpassing 90% and isolated yields stabilizing above 80%. For a reliable dexlansoprazole intermediate supplier, this methodology eliminates the need for complex downstream purification steps associated with racemate splitting, thereby streamlining the entire manufacturing workflow. The ability to directly synthesize the desired R-enantiomer with high fidelity not only reduces raw material consumption but also minimizes the generation of hazardous waste, aligning perfectly with modern green chemistry principles and cost reduction in API manufacturing initiatives.
Mechanistic Insights into Ti-N Coordinated Asymmetric Oxidation
The core mechanistic advantage of this technology lies in the unique electronic and steric properties of the sulfonamide ligand when coordinated with titanium (IV). Unlike oxygen-donor ligands such as diethyl tartrate, which form labile Ti-O bonds susceptible to hydrolysis and thermal decomposition, the nitrogen-donor sulfonamide group forms a significantly more stable Ti-N bond. Under the alkaline conditions facilitated by N,N-diisopropylethylamine, the sulfonamide moiety likely undergoes deprotonation to form an anionic species that coordinates tightly with the titanium center. This enhanced stability prevents the premature collapse of the catalytic cycle, ensuring that the active oxidizing species remains available throughout the reaction duration. The rigid chiral backbone of the 1,2-diphenylethylenediamine scaffold further imposes a well-defined chiral environment around the metal center, effectively directing the approach of the oxidant (cumene hydroperoxide) to the sulfide substrate. This precise spatial arrangement is critical for achieving the high stereoselectivity observed, as it energetically favors the formation of the R-sulfoxide over the S-enantiomer. For technical teams, understanding this mechanism underscores the importance of maintaining strict stoichiometric control over the ligand-to-titanium ratio to maximize the population of the active catalytic species.
Furthermore, the inclusion of trace amounts of purified water plays a subtle yet vital role in the activation of the catalyst complex, potentially facilitating the formation of the active oxo-titanium species required for oxygen transfer. The reaction kinetics are carefully managed by the sequential addition of reagents: the catalyst components are pre-formed at elevated temperatures to ensure complete complexation before the introduction of the substrate and oxidant. This staged addition prevents side reactions and ensures that the oxidation occurs exclusively within the chiral pocket of the catalyst. The use of cumene hydroperoxide as the terminal oxidant is also advantageous, as it provides a clean oxygen transfer mechanism without generating excessive acidic byproducts that could degrade the acid-sensitive benzimidazole core. By optimizing the molar ratios of the ligand, titanium source, and base, the process achieves a balance between reaction rate and selectivity, mitigating the risk of over-oxidation to the sulfone impurity. This level of mechanistic control is essential for producing high-purity pharmaceutical intermediates that meet stringent regulatory specifications for impurity profiles.
How to Synthesize Dexlansoprazole Efficiently
Implementing this synthesis route requires precise adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the catalyst solution under an inert atmosphere, typically nitrogen, to prevent moisture interference prior to the controlled addition of water. Operators must carefully monitor the temperature ramp-up to 53.5°C ± 1°C, as this specific thermal window is critical for the proper formation of the active titanium-ligand complex. Following the addition of the sulfide substrate and the base, the reaction mixture is cooled to 5°C before the slow introduction of the oxidant, a step designed to manage the exotherm and maintain high enantioselectivity. The detailed standardized synthesis steps below provide a comprehensive guide for laboratory and pilot-scale execution, ensuring that all critical process parameters are maintained within the optimal range defined by the intellectual property.
- Prepare the catalyst complex by mixing the chiral ligand, tetraisopropyl titanate, and toluene at room temperature, then heating to 50-60°C.
- Add purified water and the sulfide substrate (2-[[[3-methyl-4-(2,2,2-trifluoroethoxy)-2-pyridyl]methyl]sulfanyl]-1H-benzimidazole) to the reaction mixture.
- Introduce N,N-diisopropylethylamine followed by cooling to 5°C and adding cumene hydroperoxide to initiate the asymmetric oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this titanium-catalyzed route offers profound advantages for procurement managers and supply chain heads focused on cost efficiency and continuity. The primary driver of value is the substantial increase in chemical yield compared to legacy resolution methods; by bypassing the 50% theoretical yield ceiling of racemate splitting, manufacturers can effectively double the output from the same quantity of starting materials. This efficiency gain translates directly into significant cost reduction in pharmaceutical intermediates manufacturing, as the burden of purchasing, handling, and disposing of the unwanted S-enantiomer is completely eliminated. Moreover, the enhanced stability of the catalyst complex reduces the risk of batch failures due to catalyst deactivation, a common issue with traditional tartrate systems that often leads to costly production delays and supply interruptions. The robustness of the process against minor temperature fluctuations further enhances supply chain reliability, allowing for more flexible scheduling and reduced need for expensive cryogenic cooling infrastructure.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the high conversion rate significantly lower the overall cost of goods sold by maximizing raw material utilization. By avoiding the complex derivatization and separation procedures required in chemical resolution, the process simplifies the workflow and reduces labor and utility costs associated with extended processing times. The high selectivity also minimizes the formation of difficult-to-remove impurities, reducing the load on downstream purification units and extending the lifespan of chromatography columns or crystallization equipment. These cumulative efficiencies create a leaner manufacturing model that is highly competitive in the global API market.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents, such as tetraisopropyl titanate and the specific diamine ligand, ensures that raw material sourcing is not a bottleneck for production. Unlike enzymatic methods that rely on specialized biocatalysts with limited shelf-life and supply, this chemical catalyst system can be prepared on-demand with standard industrial chemicals. The process stability ensures consistent batch-to-batch quality, reducing the likelihood of out-of-specification results that could disrupt inventory planning. This predictability allows supply chain planners to maintain lower safety stock levels while still meeting customer demand, optimizing working capital and storage capacity.
- Scalability and Environmental Compliance: The reaction conditions are amenable to large-scale reactor operations, utilizing standard solvents like toluene and manageable temperature ranges that do not require exotic equipment. The high atom economy of the asymmetric oxidation reduces the volume of chemical waste generated per kilogram of product, simplifying wastewater treatment and lowering environmental compliance costs. By avoiding the use of heavy metal catalysts or toxic resolving agents, the process aligns better with increasingly strict environmental regulations, mitigating regulatory risk and enhancing the sustainability profile of the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and chemical principles involved. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for procurement professionals assessing the long-term viability of the supply source.
Q: What is the primary advantage of the new titanium ligand over L-diethyl tartrate?
A: The new ligand forms a stable Ti-N bond compared to the unstable Ti-O bond in traditional methods, resulting in significantly higher process stability and enantiomeric excess (≥99.2%).
Q: How does this method improve yield compared to racemate resolution?
A: Unlike resolution methods which theoretically max out at 50% yield and often achieve only 40-50%, this asymmetric synthesis achieves yields ≥80% by directly producing the desired R-enantiomer.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent highlights that the catalyst is less sensitive to temperature fluctuations and uses readily available reagents, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dexlansoprazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the titanium-catalyzed oxidation described in CN103694225A can be seamlessly translated into robust manufacturing operations. We are committed to delivering high-purity dexlansoprazole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex chiral syntheses allows us to offer partners a secure and high-quality supply chain for this essential proton pump inhibitor building block.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages of switching to this catalytic method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring that your transition to this superior manufacturing process is smooth, compliant, and commercially successful.
