Advanced Direct Ethynylation Technology for High-Purity Alpha-Beta Unsaturated Alcohols and Commercial Scalability
Advanced Direct Ethynylation Technology for High-Purity Alpha-Beta Unsaturated Alcohols and Commercial Scalability
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes for alpha-beta unsaturated alcohols, which serve as critical intermediates for high-value fragrances like nerolidol and essential vitamin precursors such as isophytol. Patent CN1295201C introduces a groundbreaking methodology that shifts the paradigm from hazardous, high-energy traditional processes to a streamlined direct ethynylation technique. This innovation utilizes acetylene gas and solid strong alkali compounds in an organic solvent system to convert carbonyl-containing ketones or aldehydes directly into alpha-beta unsaturated alkynols. By operating under mild pressures of 0.1-0.25 MPa and avoiding extreme cryogenic conditions, this technology offers a reliable flavor and fragrance intermediate supplier pathway that drastically simplifies production workflows. The subsequent selective hydrogenation step further refines these alkynols into valuable enols, ensuring high purity and structural integrity essential for downstream applications in the synthesis of vitamins K1 and E.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of terpene alkyl allyl tertiary alcohols relied heavily on two problematic methodologies: the vinyl halide magnesium Grignard reagent addition and the metal acetylide reduction method involving liquid ammonia. The Grignard approach demands rigorously anhydrous environments and freshly prepared reagents, often leading to unstable reaction profiles and polymerization issues that suppress yields. Furthermore, the metal acetylide method typically requires liquefied ammonia at temperatures below -40°C within high-pressure sealed systems, creating significant safety hazards related to ammonia overflow and complex recycling infrastructure. These conventional routes impose severe operational constraints, requiring specialized equipment for low-temperature reflux and pressurized containment, which inherently drives up capital expenditure and operational complexity for manufacturers attempting cost reduction in vitamin intermediate manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a direct ethynylation strategy that operates effectively at normal or near-normal pressure conditions. By introducing acetylene gas directly into a mixture of strong alkali (such as potassium hydroxide) and organic solvents like toluene or tetrahydrofuran, the reaction proceeds smoothly without the need for pre-forming unstable metal acetylides in liquid ammonia. This method maintains the reaction system in a continuous acetylene atmosphere at 0.1-0.25 MPa, allowing for efficient addition to the carbonyl group. The elimination of cryogenic requirements and the use of common solid alkalis significantly lower the energy consumption and pollution load. This technological shift enables the commercial scale-up of complex polymer additives and fine chemicals with greater ease, providing a safer and more economically viable route for producing high-purity OLED material precursors and fragrance compounds.
Mechanistic Insights into Direct Ethynylation and Selective Hydrogenation
The core mechanism of this invention relies on the in-situ formation of acetylene salts within the organic phase. When acetylene gas is dissolved into the organic solvent containing a strong base, it reacts to form an acetylene salt which immediately undergoes nucleophilic addition with the ketone or aldehyde substrate. This dynamic equilibrium ensures that the reactive acetylene species are generated and consumed continuously, preventing the accumulation of unstable intermediates. The resulting alpha-beta unsaturated alkynol salt remains dissolved in the organic solvent, facilitating a homogeneous reaction environment that enhances conversion rates. Following the reaction, the addition of an acidic substance protonates the alkynol salt, releasing the free alpha-beta unsaturated alkynol which is then isolated via solid-liquid separation and washing. This mechanistic elegance avoids the side reactions common in Grignard chemistry, such as enolization or reduction, thereby preserving the carbon skeleton integrity required for high-value terpenoid synthesis.
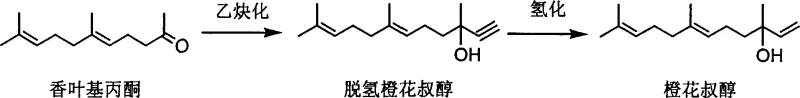
Following the ethynylation, the process employs a sophisticated selective hydrogenation mechanism to convert the triple bond into a double bond without over-reduction to a single bond. Using a solid-supported catalyst containing palladium or platinum (0.5%-10% mass percent), often modified with basic compounds like quinoline, the system achieves precise control over the hydrogenation degree. The catalyst, accounting for only 0.005%-1% of the product mass, facilitates the adsorption of hydrogen gas at mild pressures of 0.1-0.2 MPa. This selectivity is crucial for producing alpha-beta unsaturated enols like nerolidol and isophytol, where the preservation of the olefinic geometry is vital for biological activity and olfactory properties. The ability to tune the catalyst loading and reaction temperature between 20-100°C allows manufacturers to optimize the impurity profile, ensuring reducing lead time for high-purity alpha-beta unsaturated alcohols by minimizing downstream purification steps.
How to Synthesize Dehydronerolidol Efficiently
The synthesis of dehydronerolidol via this patented route involves a carefully controlled sequence of mixing, gas introduction, and dropwise addition to maximize yield and safety. The process begins with the preparation of a basic reaction medium, followed by the saturation of the system with acetylene to ensure immediate reaction upon substrate addition. Detailed operational parameters regarding solvent ratios, alkali equivalents, and distillation conditions are critical for reproducibility. For a comprehensive understanding of the specific stoichiometric ratios and temperature gradients required for optimal performance, please refer to the standardized synthesis protocol outlined below.
- Mix strong alkali (e.g., KOH) and organic solvent in a reactor, then introduce acetylene gas to maintain a pressure of 0.1-0.25 MPa.
- Dropwise add the carbonyl-containing ketone or aldehyde substrate while maintaining the acetylene atmosphere and stirring for 0.5-100 hours.
- Separate solid alkali, neutralize with acid, recover solvent via vacuum distillation, and optionally hydrogenate the alkynol to enol using a Lindlar catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this technology addresses several critical pain points associated with traditional terpene alcohol synthesis. The elimination of liquid ammonia and the reliance on standard solid alkalis like potassium hydroxide drastically simplify the raw material sourcing strategy, reducing dependency on specialized cryogenic gas suppliers. Furthermore, the ability to operate at near-atmospheric pressure removes the need for expensive high-pressure autoclaves, lowering the barrier to entry for contract manufacturing organizations. This operational simplicity translates directly into enhanced supply chain reliability, as the process is less susceptible to shutdowns caused by equipment failure or stringent safety inspections associated with hazardous high-pressure systems.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need for expensive and energy-intensive cryogenic cooling systems required for liquid ammonia processes. By utilizing common organic solvents that can be recovered and recycled via vacuum distillation at moderate temperatures (30-100°C), the overall solvent consumption is minimized. Additionally, the use of solid alkali instead of complex organometallic reagents reduces raw material costs, while the simplified workup procedure lowers labor and utility expenses associated with prolonged reaction times and difficult separations.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is essential for maintaining long-term supply contracts with major fragrance and pharmaceutical houses. Since the process does not rely on unstable Grignard reagents that require immediate use, the manufacturing schedule becomes more flexible and predictable. The reduced risk of safety incidents related to ammonia leaks or pyrophoric reagents further secures the production facility's operational continuity, ensuring that delivery timelines for critical intermediates like dehydroisophytol are met without interruption.
- Scalability and Environmental Compliance: This method is inherently scalable due to its reliance on standard unit operations such as gas sparging, filtration, and distillation. The environmental footprint is substantially reduced as the process avoids the release of volatile ammonia and minimizes the generation of heavy metal waste often associated with alternative reduction methods. The ability to recover solvents efficiently under reduced pressure not only improves economic efficiency but also aligns with increasingly stringent global environmental regulations, making it an ideal choice for sustainable chemical manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this direct ethynylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield expectations, safety protocols, and product specifications. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing production lines for fine chemical intermediates.
Q: How does this direct ethynylation method improve safety compared to traditional Grignard or liquid ammonia processes?
A: This method eliminates the need for cryogenic liquid ammonia (-40°C) and strictly anhydrous conditions required for Grignard reagents. By using solid alkali (KOH/NaOH) and acetylene gas at near-atmospheric pressure (0.1-0.25 MPa), it significantly reduces operational risks associated with high-pressure sealed systems and toxic ammonia overflow.
Q: What is the typical purity achievable for dehydronerolidol using this patented process?
A: According to the patent data, the crude product of dehydronerolidol can achieve a GC purity of approximately 96.69% with minimal unreacted geranyl acetone (1.85%). Further purification via vacuum distillation or rectification yields a colorless, high-purity product suitable for fine chemical applications.
Q: Can this process be scaled for industrial production of vitamin intermediates like isophytol?
A: Yes, the process is designed for scalability. It utilizes common organic solvents (toluene, THF) and standard solid-liquid separation techniques. The mild reaction conditions (30-100°C) and ability to recover solvents via vacuum distillation make it highly suitable for commercial scale-up of complex intermediates like dehydroisophytol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydronerolidol Supplier
NINGBO INNO PHARMCHEM stands at the forefront of adopting advanced synthetic technologies to deliver superior chemical solutions to the global market. Our expertise encompasses the translation of complex patent methodologies, such as the direct ethynylation process described in CN1295201C, into robust commercial manufacturing protocols. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs employ state-of-the-art analytical techniques, including GC-MS, to verify the identity and purity of every batch, guaranteeing that products like dehydronerolidol and isophytol are free from detrimental impurities.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific supply chain requirements. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your production volumes and quality needs. We encourage you to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and value in your manufacturing operations.
