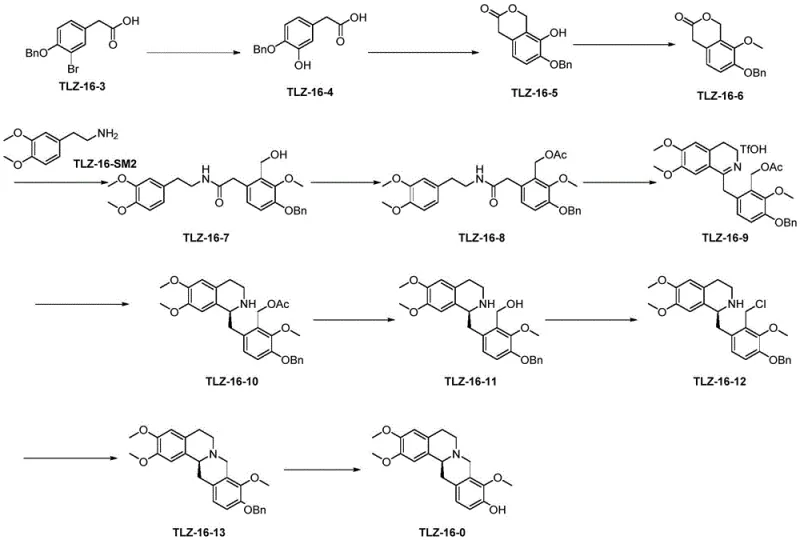
Scalable Synthesis of L-Corydalmine: Advanced Catalytic Routes for Commercial Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex alkaloids, and the recent disclosure in patent CN112812112A offers a transformative approach to producing L-corydalmine, a potent isoquinoline alkaloid with significant analgesic and anti-addiction properties. This technical breakthrough addresses long-standing challenges in the synthesis of this molecule, specifically focusing on the critical debenzylation and cyclization steps that have historically plagued yield and purity metrics. By leveraging a novel palladium hydroxide on carbon catalytic system, the disclosed method achieves exceptional selectivity, avoiding the common pitfall of demethylation that compromises product quality in conventional routes. The strategic implementation of trifluoromethanesulfonic anhydride for ring closure further streamlines the process, reducing impurity profiles and simplifying downstream purification. For R&D directors and procurement specialists alike, this patent represents a viable pathway to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials consistently. The following analysis dissects the chemical innovations within this patent, highlighting how these technical advancements translate into tangible supply chain stability and cost efficiency for commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-corydalmine and its precursors has been hindered by inefficient cyclization protocols and harsh debenzylation conditions that degrade the sensitive isoquinoline core. Traditional methods often rely on phosphorus oxychloride for ring closure, a reagent known to generate substantial side reaction products, reportedly around 20%, which necessitates complex and wasteful post-treatment procedures involving large quantities of quenching agents. Furthermore, conventional debenzylation strategies frequently employ Lewis acids or alcoholic solvents with hydrochloric acid, conditions that inadvertently cleave methyl ether groups essential for the biological activity of the final molecule. This lack of chemoselectivity results in yields often dropping below 70%, with significant product loss during the neutralization and purification stages. The accumulation of impurities not only lowers the overall throughput but also imposes a heavy burden on quality control laboratories, requiring extensive chromatographic separation to meet pharmacopeial standards. These technical bottlenecks have traditionally made the commercial scale-up of complex pharmaceutical intermediates like L-corydalmine economically challenging and operationally risky for manufacturers.
The Novel Approach
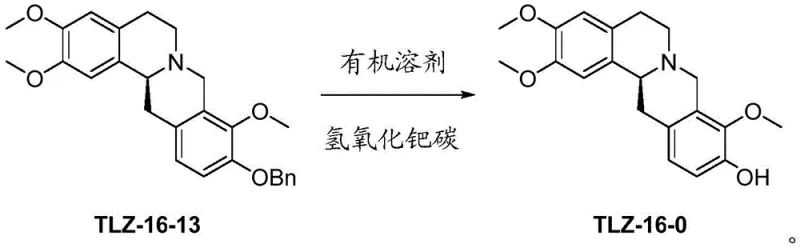
The methodology outlined in patent CN112812112A introduces a paradigm shift by utilizing palladium hydroxide on carbon as a highly selective hydrogenation catalyst for the final debenzylation step. This catalytic system operates effectively in a mixed solvent environment of ethyl acetate and alcohol, allowing for the clean removal of the benzyl protecting group without affecting the adjacent methoxy functionalities. The inventors have demonstrated that this specific catalyst loading, ranging from 5% to 30% by weight, facilitates a reaction that proceeds with yields exceeding 85% and purity levels surpassing 99.5%. Additionally, the substitution of phosphorus oxychloride with trifluoromethanesulfonic anhydride in the presence of a base reagent like 2-chloropyridine dramatically reduces the reaction temperature and time while minimizing byproduct formation. This dual innovation ensures that the process is not only chemically superior but also operationally simpler, as the catalyst can be filtered off and the product crystallized directly from the reaction solvent. Such improvements are critical for reducing lead time for high-purity alkaloids and ensuring a consistent supply of material for downstream drug development.
Mechanistic Insights into Pd-Catalyzed Debenzylation and Cyclization
The core of this synthetic advancement lies in the precise control of catalytic hydrogenation and electrophilic cyclization mechanisms. In the debenzylation step, the palladium hydroxide on carbon surface adsorbs hydrogen and the benzyl ether substrate, facilitating the cleavage of the C-O bond through a hydrogenolysis mechanism that is remarkably tolerant of other functional groups. Unlike acidic hydrolysis which attacks electron-rich methoxy groups, this catalytic surface reaction is specific to the benzylic position, preserving the integrity of the trimethoxy substitution pattern essential for L-corydalmine's pharmacological profile. The use of ethyl acetate as a co-solvent further modulates the solubility of the intermediate TLZ-16-13, ensuring homogeneous reaction conditions that prevent localized over-reduction or catalyst poisoning. This mechanistic specificity is what allows the process to achieve such high enantiomeric excess values, maintaining the chiral integrity of the molecule throughout the transformation. For technical teams, understanding this selectivity is key to replicating the success of this route in a GMP environment.
Regarding impurity control, the switch to trifluoromethanesulfonic anhydride for the cyclization of the amide precursor TLZ-16-8 to the iminium salt TLZ-16-9 is equally vital. The mechanism involves the activation of the amide oxygen by the trifling agent, followed by intramolecular electrophilic aromatic substitution to close the isoquinoline ring. The presence of 2-chloropyridine as a base scavenges the generated triflic acid, preventing acid-catalyzed degradation of the sensitive intermediate. This contrasts sharply with phosphorus-based reagents which generate phosphoric acid byproducts that are difficult to remove and can catalyze decomposition. The result is a cleaner reaction profile where the primary impurity is easily removed via crystallization rather than complex chromatography. This level of impurity management is crucial for meeting the stringent purity specifications required for API intermediates, ensuring that the final drug substance is free from genotoxic or persistent organic impurities that could delay regulatory approval.
How to Synthesize L-Corydalmine Efficiently
The synthesis of L-corydalmine via this patented route involves a sequence of well-defined chemical transformations that prioritize yield and purity at every stage. The process begins with the construction of the isoquinoline core through the cyclization of a substituted phenethylamine derivative, followed by stereoselective reduction and final deprotection. Detailed standard operating procedures for each step, including precise temperature controls, reagent addition rates, and workup protocols, are essential for reproducing the high yields reported in the patent literature. Operators must pay close attention to the catalyst activation and solvent drying steps, as moisture can significantly impact the efficiency of the triflic anhydride cyclization. The following guide outlines the critical operational parameters necessary to achieve the reported >85% yield and >99.5% purity in a production setting.
- Prepare the intermediate TLZ-16-13 via cyclization using trifluoromethanesulfonic anhydride and a base reagent like 2-chloropyridine.
- Conduct debenzylation of TLZ-16-13 using 5-30% Pd(OH)2/C catalyst in a mixed solvent system of ethyl acetate and alcohol.
- Purify the final product through crystallization and filtration to achieve purity exceeding 99.5% with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for specialty chemicals. The elimination of hazardous phosphorus reagents and the reduction in purification steps directly translate to a safer and more environmentally compliant manufacturing process, which is increasingly important for maintaining social license to operate in the fine chemical sector. By simplifying the workflow, manufacturers can reduce the operational complexity and the associated labor costs, leading to significant cost savings in API intermediate manufacturing without compromising on quality. The robustness of the catalytic system also means that the process is less sensitive to minor variations in raw material quality, enhancing supply chain reliability and reducing the risk of batch failures. This stability is paramount for ensuring continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive and hazardous reagents like phosphorus oxychloride, replacing them with more manageable alternatives that reduce waste disposal costs. By avoiding complex neutralization and extraction steps, the consumption of solvents and auxiliary chemicals is drastically simplified, leading to substantial cost savings in raw material procurement. Furthermore, the high yield of the final step minimizes the loss of valuable chiral intermediates, ensuring that the cost of goods sold remains competitive even at smaller production scales. The ability to crystallize the product directly from the reaction solvent further reduces energy consumption associated with solvent exchange and evaporation, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: The use of commercially available catalysts and solvents ensures that the supply chain is not dependent on scarce or specialized reagents that could cause bottlenecks. The robustness of the reaction conditions allows for flexible production scheduling, as the process is less prone to delays caused by sensitive parameter deviations. This reliability is critical for maintaining inventory levels and ensuring that downstream drug formulation teams have uninterrupted access to key intermediates. By partnering with a reliable pharmaceutical intermediate supplier who utilizes this technology, companies can mitigate the risk of supply disruptions and secure a steady flow of high-quality materials for their clinical and commercial programs.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing standard reactor equipment and filtration techniques that are easily transferable from pilot to commercial scale. The reduction in hazardous waste generation aligns with modern environmental regulations, reducing the burden on waste treatment facilities and lowering the overall environmental footprint of the manufacturing site. This compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is increasingly a factor in supplier selection criteria for major pharmaceutical companies. The ease of scale-up ensures that production capacity can be rapidly expanded to meet market demand without the need for significant capital investment in new specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of L-corydalmine using the methods described in patent CN112812112A. These answers are derived directly from the experimental data and technical disclosures within the patent, providing clarity on the feasibility and advantages of this specific synthetic route. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or commercial sourcing of this intermediate.
Q: What is the primary advantage of using Pd(OH)2/C for L-corydalmine synthesis?
A: The use of palladium hydroxide on carbon allows for highly selective debenzylation under mild conditions, avoiding the demethylation side reactions common with Lewis acids, thereby significantly improving yield and purity.
Q: How does the new cyclization method improve upon traditional phosphorus oxychloride routes?
A: Replacing phosphorus oxychloride with trifluoromethanesulfonic anhydride reduces side reaction products from approximately 20% to negligible levels, simplifying post-treatment and eliminating the need for extensive quenching procedures.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability with yields exceeding 85% and purity over 99.5% in kilogram-scale batches, utilizing readily available reagents and standard filtration techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Corydalmine Supplier
The technical potential of this synthetic route is immense, offering a clear path to high-quality L-corydalmine production that meets the rigorous demands of the global pharmaceutical market. NINGBO INNO PHARMCHEM, as a seasoned CDMO expert, possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the high enantiomeric excess and chemical purity demanded by this process. We understand the nuances of handling palladium catalysts and trifling agents safely and efficiently, ensuring that every batch delivered meets the highest standards of quality and consistency. Our commitment to technical excellence ensures that your supply chain is backed by scientific rigor and operational reliability.
We invite you to initiate a conversation about optimizing your supply chain for this critical intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments that will demonstrate how our implementation of this patented technology can benefit your project. By collaborating with us, you gain access to a partner dedicated to advancing your drug development timeline through superior chemical manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
