Advanced Julolidine-Based Nonlinear Optical Chromophores for Next-Gen Optoelectronics
Advanced Julolidine-Based Nonlinear Optical Chromophores for Next-Gen Optoelectronics
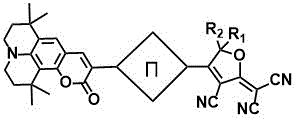
The rapid evolution of optoelectronic devices demands materials that combine exceptional optical nonlinearity with robust thermal and chemical stability. Patent CN109438459B introduces a groundbreaking class of organic second-order nonlinear optical chromophores featuring a sophisticated D-π-A (Donor-π-Acceptor) architecture. These novel compounds utilize tetramethyljulolidine derivatives as potent electron donors coupled with tricyanofuran-based electron acceptors, creating a highly polarized system capable of significant frequency conversion and signal processing. The strategic incorporation of steric hindrance groups within the julolidine framework effectively suppresses detrimental dipole-dipole interactions, a common bottleneck in high-concentration polymer doping. This innovation not only enhances the macroscopic electro-optic coefficient to impressive levels exceeding 76 pm/V but also ensures thermal decomposition temperatures surpassing 250°C, addressing critical reliability concerns in high-power laser and telecommunication applications.
For procurement specialists and supply chain managers seeking a reliable nonlinear optical chromophore supplier, this technology represents a paradigm shift towards cost-effective and scalable manufacturing. The synthetic routes described eliminate the need for exotic transition metal catalysts often associated with complex organometallic syntheses, relying instead on accessible organic bases and standard solvent systems. This simplification translates directly into cost reduction in electronic chemical manufacturing by minimizing purification steps and reducing the environmental footprint associated with heavy metal waste disposal. Furthermore, the modular nature of the synthesis allows for the facile adjustment of the conjugated electron bridge, enabling the production of a diverse library of high-purity optoelectronic materials tailored to specific refractive index and absorption requirements without compromising on yield or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing organic nonlinear optical materials often struggle with a trade-off between microscopic hyperpolarizability and macroscopic alignment stability. Conventional chromophores frequently suffer from strong intermolecular electrostatic interactions that lead to antiparallel pairing of dipoles, effectively canceling out the net nonlinearity in the poled polymer film. Additionally, many existing high-performance dyes lack sufficient thermal stability, decomposing at temperatures well below the processing thresholds required for modern device fabrication, typically around 200°C. This thermal instability limits the operational lifetime of electro-optic modulators and increases the failure rate in harsh environments. Moreover, older synthetic methodologies often involve multi-step sequences with low atom economy, requiring hazardous reagents and generating significant volumes of toxic waste, which complicates regulatory compliance and increases the total cost of ownership for manufacturers aiming for green chemistry standards.
The Novel Approach
The methodology outlined in the patent data overcomes these historical barriers through a rational molecular design that integrates steric isolation directly into the donor moiety. By employing tetramethyljulolidine derivatives, the synthesis introduces bulky methyl groups that physically separate the highly polar chromophore cores, thereby inhibiting aggregation and preserving the poling-induced order. This structural innovation is complemented by a versatile synthetic strategy that supports both direct condensation and bridge-extension pathways. The use of tricyanofuran acceptors provides a stronger electron-withdrawing capability compared to traditional cyano or nitro groups, significantly boosting the first-order hyperpolarizability (β value). This novel approach ensures that the resulting materials possess not only superior optical figures of merit but also the mechanical and thermal robustness necessary for the commercial scale-up of complex chromophores in high-volume optoelectronic device production lines.
Mechanistic Insights into D-π-A Charge Transfer and Steric Isolation
The exceptional performance of these materials stems from the efficient intramolecular charge transfer (ICT) facilitated by the conjugated π-system connecting the donor and acceptor. Upon exposure to an electric field or optical stimulus, electrons are readily displaced from the electron-rich julolidine nitrogen center through the conjugated bridge to the electron-deficient tricyanofuran ring. This displacement creates a large change in the dipole moment, which is the fundamental origin of the second-order nonlinear optical response. The efficiency of this process is maximized by optimizing the planarity and length of the conjugated bridge, which can be tuned using vinyl, thiophene, or furan linkers as depicted in the structural variations. Theoretical calculations support that this specific D-π-A arrangement lowers the energy gap between the ground and excited states, enhancing the resonance contribution to the hyperpolarizability without pushing the absorption edge into the visible spectrum, thus maintaining optical transparency in telecommunications bands.
Crucially, the mechanism also addresses the issue of chromophore-chromophore interaction through site isolation. In standard high-loading scenarios, chromophores tend to align antiparallel to minimize electrostatic energy, which reduces the net electro-optic effect. The tetramethyl groups on the julolidine ring act as a physical barrier, increasing the effective volume of the chromophore and preventing close approach of the dipolar ends. This steric hindrance allows for higher doping concentrations in amorphous polymer matrices like PMMA without inducing crystallization or phase separation. Consequently, the macroscopic electro-optic coefficient (r33) is significantly enhanced, as evidenced by experimental data showing values up to 76 pm/V. This mechanistic understanding is vital for R&D teams aiming to further optimize the refractive index and loss characteristics of the final waveguide devices.
How to Synthesize Julolidine-Tricyanofuran Chromophores Efficiently
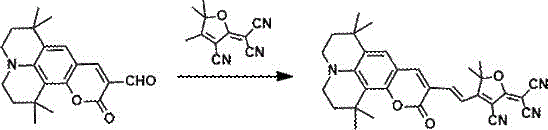
The patent details two primary synthetic pathways to access these high-value intermediates, offering flexibility depending on the desired conjugation length and bridge composition. The first method involves a direct Knoevenagel condensation between a julolidine-aldehyde derivative and a tricyanofuran acceptor in ethanol, catalyzed by organic bases such as triethylamine or piperidine at moderate temperatures of 50-80°C. This one-pot procedure is highly atom-economical and yields products with high purity after simple column chromatography. For applications requiring extended conjugation to red-shift the absorption or enhance nonlinearity, a second multi-step method is employed. This involves a Wittig reaction to install a thiophene or furan vinyl bridge, followed by Vilsmeier-Haack formylation to regenerate the aldehyde functionality, and finally condensation with the acceptor. Both routes utilize commercially available starting materials and avoid cryogenic conditions, facilitating easy technology transfer to pilot and production scales.
- Prepare the electron donor by synthesizing or sourcing tetramethyljulolidine coumarin derivatives, ensuring high purity for optimal optical performance.
- Select the conjugated electron bridge strategy: either direct condensation for simple structures or Wittig reaction followed by Vilsmeier-Haack formylation for extended thiophene/furan bridges.
- Perform the final Knoevenagel condensation between the aldehyde intermediate and tricyanofuran electron acceptor in ethanol with an organic base catalyst at 50-80°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic technology offers profound advantages in terms of raw material security and process safety. The reliance on ethanol and 1,2-dichloroethane as solvents, along with common organic bases, means that the supply chain is not vulnerable to the geopolitical fluctuations often associated with rare earth metals or specialized organometallic catalysts. This stability ensures consistent lead times and reduces the risk of production stoppages due to reagent shortages. Furthermore, the high thermal stability of the final product (>250°C) reduces the need for stringent temperature controls during downstream device fabrication, lowering energy consumption and equipment wear. For procurement managers, this translates into a more predictable cost structure and reduced total cost of ownership over the lifecycle of the optoelectronic component.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of high-yield condensation reactions significantly lower the direct material costs. The simplified purification process, often requiring only standard silica gel chromatography rather than preparative HPLC or recrystallization from exotic solvents, reduces solvent consumption and waste treatment expenses. Additionally, the high atom economy of the Knoevenagel condensation minimizes the generation of byproduct waste, aligning with green chemistry initiatives that can lead to regulatory incentives and lower disposal fees.
- Enhanced Supply Chain Reliability: The starting materials, including julolidine derivatives and tricyanofuran precursors, are synthesized from commodity chemicals with established global supply networks. This reduces dependency on single-source suppliers for niche intermediates. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without significant yield loss, ensures consistent batch-to-batch quality. This reliability is critical for maintaining just-in-time inventory models and meeting the rigorous qualification standards of multinational electronics manufacturers.
- Scalability and Environmental Compliance: The synthetic routes are inherently scalable, having been demonstrated effectively in multi-gram laboratory settings with yields exceeding 80%. The absence of heavy metals simplifies the environmental compliance landscape, making it easier to obtain permits for large-scale production facilities. The ability to produce high-purity materials with minimal impurity profiles reduces the burden on quality control laboratories and accelerates the time-to-market for new optoelectronic devices, providing a competitive edge in the fast-paced display and telecommunications sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis, stability, and application of these advanced nonlinear optical materials. Understanding these details is essential for integrating these chromophores into your specific device architectures and ensuring optimal performance under operating conditions.
Q: What is the thermal stability of these julolidine-based chromophores?
A: According to patent CN109438459B, the synthesized chromophores exhibit excellent thermal stability, with decomposition temperatures reaching above 250°C for specific derivatives containing thiophene bridges, ensuring robustness in device operation.
Q: How does the D-π-A structure improve electro-optic performance?
A: The Donor-π-Acceptor architecture facilitates high electron delocalization and charge transfer. The julolidine donor provides strong electron-donating capability, while the tricyanofuran acceptor ensures strong electron-withdrawing properties, resulting in electro-optic coefficients up to 76 pm/V.
Q: Can these chromophores be scaled for industrial production?
A: Yes, the synthesis methods utilize common solvents like ethanol and 1,2-dichloroethane with standard catalysts such as triethylamine. The reactions proceed at moderate temperatures (50-90°C) and offer high atom economy, making them suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Second-Order Nonlinear Optical Chromophore Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance materials play in the advancement of next-generation optoelectronics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of chromophore meets the exacting standards required for electro-optic modulation and frequency conversion applications. Our commitment to quality assurance means that you receive materials with verified electro-optic coefficients and thermal stability profiles, ready for immediate integration into your polymer formulations.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development cycles. By leveraging our expertise in fine chemical synthesis, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Let us help you engineer the future of light with materials that deliver unmatched performance and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
