Advanced Synthesis of Pioglitazone Related Substance D for Global Pharmaceutical Quality Control
The pharmaceutical industry's relentless pursuit of quality and safety has placed impurity profiling at the forefront of drug development, particularly for widely prescribed antidiabetic agents like Pioglitazone. Patent CN114031548A introduces a groundbreaking preparation method for Pioglitazone Related Substance D, addressing critical gaps in the availability of high-purity reference standards. This novel synthetic route leverages a sophisticated six-step sequence that transforms simple precursors into the complex target molecule with exceptional fidelity. By meticulously controlling reaction parameters such as temperature gradients ranging from -5°C to 80°C and optimizing reagent stoichiometry, the disclosed method achieves a level of purity that surpasses existing art. For global pharmaceutical manufacturers, this represents a pivotal advancement in ensuring the integrity of quality control protocols, as the availability of well-characterized impurities is mandatory for regulatory submissions and batch release testing.
The significance of this technology extends beyond mere compliance; it offers a robust framework for understanding the degradation pathways and potential toxicological profiles of thiazolidinedione derivatives. As regulatory bodies like the FDA and EMA tighten their guidelines on impurity thresholds, the ability to synthesize specific related substances on demand becomes a strategic asset. This patent details a pathway that not only enhances the structural elucidation of Pioglitazone impurities but also provides a scalable manufacturing process that can be adapted for the production of other structurally related analogues. Consequently, this innovation serves as a cornerstone for developing more stable and effective formulations, ultimately benefiting patient safety and therapeutic outcomes in the management of type 2 diabetes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Pioglitazone related substances has been plagued by inefficiencies that compromise both yield and purity. Traditional routes often suffer from non-selective reactions that generate a complex mixture of by-products, necessitating laborious and costly purification steps such as repeated recrystallization or preparative HPLC. These conventional methods frequently utilize harsh reaction conditions that can lead to the degradation of sensitive functional groups, resulting in low overall recovery rates. Furthermore, the reliance on unstable intermediates in older protocols poses significant challenges for scale-up, as minor deviations in temperature or mixing can lead to runaway reactions or the formation of polymeric tars. Such inconsistencies make it difficult for suppliers to guarantee the batch-to-batch reproducibility required for certified reference materials, creating bottlenecks in the supply chain for pharmaceutical quality control laboratories.
The Novel Approach
In stark contrast, the method disclosed in CN114031548A employs a rational design strategy that prioritizes chemoselectivity and operational simplicity. By introducing a controlled bromination step followed by a mild etherification, the process establishes the core carbon-oxygen backbone with minimal side reactions. The subsequent catalytic hydrogenation using 5% palladium carbon ensures the clean reduction of the nitro group without affecting other sensitive moieties, a common pitfall in older chemical reduction methods. The innovation truly shines in the diazotization and coupling sequence, where precise temperature control at -5°C prevents the decomposition of the diazonium salt, allowing for a highly efficient addition to ethyl acrylate. This systematic optimization of each synthetic step results in a streamlined workflow that drastically reduces the formation of impurities, thereby simplifying the final isolation and purification stages.
Mechanistic Insights into the Multi-Step Synthetic Cascade
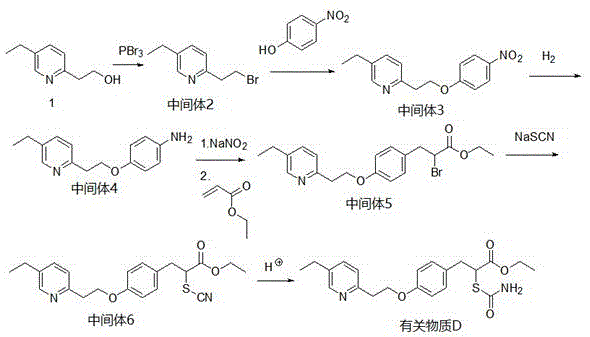
The chemical elegance of this synthesis lies in its sequential construction of the molecular architecture, beginning with the activation of 5-ethyl-2-pyridylethanol. In the initial step, phosphorus tribromide acts as a potent brominating agent, converting the hydroxyl group into a good leaving group (bromide) under cooling conditions to prevent elimination side reactions. This activated intermediate then undergoes a nucleophilic substitution with p-nitrophenol in the presence of potassium carbonate, forming the crucial ether linkage that connects the pyridine and phenyl rings. The choice of DMF as a solvent in this step is critical, as it solubilizes the polar intermediates and facilitates the SN2 reaction mechanism, ensuring high conversion rates. Following this, the nitro group is selectively reduced to an amine using hydrogen gas and a palladium catalyst, a transformation that is vital for the subsequent diazotization reaction.
The latter half of the synthesis involves the construction of the side chain characteristic of Related Substance D. The generated aniline derivative is subjected to diazotization using sodium nitrite and hydrochloric acid at sub-zero temperatures to form a reactive diazonium salt. This electrophilic species then reacts with ethyl acrylate, likely through a radical or ionic addition mechanism, to extend the carbon chain and introduce the ester functionality. The subsequent substitution of the halogen atom with a thiocyanate group using sodium thiocyanate introduces the sulfur atom, which is a key heteroatom in the final structure. Finally, acid-catalyzed hydrolysis and cyclization with concentrated sulfuric acid refine the molecule into the target Related Substance D. This intricate dance of functional group transformations is orchestrated to maximize yield while maintaining the structural integrity of the sensitive thiazolidinedione-like scaffold.
How to Synthesize Pioglitazone Related Substance D Efficiently
Executing this synthesis requires strict adherence to the optimized parameters outlined in the patent to ensure the highest quality output. The process begins with the careful addition of phosphorus tribromide to a cooled solution of the pyridine alcohol, followed by the sequential introduction of reagents for etherification and reduction. Each step demands precise monitoring of temperature and reaction time, particularly during the exothermic bromination and the sensitive diazotization phases. The detailed standardized synthetic steps provided below offer a comprehensive guide for laboratory and pilot-scale production, ensuring that operators can replicate the high purity and yield demonstrated in the patent examples.
- React 5-ethyl-2-pyridylethanol with phosphorus tribromide in dichloromethane at 0°C to form the brominated intermediate.
- Perform etherification by reacting the brominated intermediate with p-nitrophenol and potassium carbonate in DMF at 45°C.
- Reduce the nitro group using 5% palladium carbon and hydrogen in methanol to obtain the amino ether intermediate.
- Conduct diazotization with sodium nitrite and hydrochloric acid at -5°C, followed by reaction with ethyl acrylate.
- Substitute the halogen with sodium thiocyanate in DMF at 80°C to introduce the thiocyanate group.
- Treat the final intermediate with concentrated sulfuric acid, adjust pH to 7, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method translates into tangible strategic benefits that extend far beyond the laboratory bench. The streamlined nature of the reaction sequence eliminates the need for exotic or prohibitively expensive reagents, relying instead on commodity chemicals that are readily available in the global market. This reliance on standard raw materials significantly mitigates the risk of supply disruptions, ensuring a continuous flow of production inputs even during periods of market volatility. Furthermore, the high selectivity of the reaction pathway means that less material is wasted in the form of by-products, leading to a more efficient utilization of resources and a reduction in the overall cost of goods sold. By minimizing the complexity of the purification process, manufacturers can also reduce the consumption of solvents and stationary phases, contributing to a leaner and more sustainable operation.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its ability to bypass costly purification bottlenecks. Traditional methods often require extensive chromatographic separation to remove structurally similar impurities, a process that consumes vast amounts of silica gel and organic solvents. In contrast, the high intrinsic purity achieved by this new method allows for simpler workup procedures, such as extraction and crystallization, which are significantly less expensive to operate. Additionally, the use of catalytic hydrogenation rather than stoichiometric metal reductants reduces the generation of heavy metal waste, lowering the costs associated with waste disposal and environmental compliance. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final pharmaceutical product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the synthetic route, which tolerates minor variations in operating conditions without compromising product quality. The use of stable intermediates and common solvents like dichloromethane and methanol ensures that the process can be easily transferred between different manufacturing sites or scaled up without significant re-engineering. This flexibility allows suppliers to respond rapidly to fluctuations in demand, reducing lead times for high-purity pharmaceutical intermediates. Moreover, the elimination of hazardous or hard-to-source reagents simplifies logistics and storage requirements, further stabilizing the supply network against external shocks.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns with the principles of green chemistry by maximizing atom economy and minimizing waste generation. The process avoids the use of toxic heavy metals in the reduction step and employs aqueous workups that facilitate the recycling of organic solvents. This reduced environmental footprint not only lowers regulatory compliance costs but also enhances the corporate social responsibility profile of the manufacturer. The scalability of the process is evidenced by the use of standard reactor configurations and moderate temperature ranges, making it ideal for transition from kilogram-scale laboratory synthesis to multi-ton commercial production without the need for specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Pioglitazone Related Substance D. These insights are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity for stakeholders involved in pharmaceutical development and quality assurance. Understanding these nuances is essential for making informed decisions about sourcing and process implementation.
Q: What is the primary advantage of this synthesis method for Pioglitazone Related Substance D?
A: The primary advantage is the significantly improved purity and yield compared to conventional methods. By optimizing reaction conditions such as temperature control during diazotization (-5°C) and specific stoichiometric ratios, the process minimizes side reactions and polymerization, resulting in a high-quality impurity standard essential for regulatory compliance.
Q: Are the raw materials for this process readily available for large-scale production?
A: Yes, the synthesis utilizes commercially available and cost-effective starting materials such as 5-ethyl-2-pyridylethanol, p-nitrophenol, and ethyl acrylate. The solvents used, including dichloromethane, DMF, and methanol, are standard industrial chemicals, ensuring a robust and reliable supply chain for commercial manufacturing.
Q: How does this method address environmental and safety concerns in pharmaceutical manufacturing?
A: The process employs catalytic hydrogenation using 5% Pd/C, which is a cleaner alternative to stoichiometric metal reductions. Furthermore, the workup procedures involve standard extraction and pH adjustment techniques that facilitate efficient waste management. The elimination of exotic reagents simplifies the EHS (Environment, Health, and Safety) profile, making it suitable for green chemistry initiatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pioglitazone Related Substance D Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your pharmaceutical products depends on the quality of every component, down to the smallest impurity standard. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to perform comprehensive analyses, guaranteeing that every batch of Pioglitazone Related Substance D meets the highest international standards for identity and purity. We are committed to being a partner in your success, offering not just a product, but a guarantee of quality that supports your regulatory filings and patient safety initiatives.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient synthesis route can lower your total cost of ownership. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred choice for high-performance pharmaceutical intermediates. Together, we can ensure the continued availability of safe and effective antidiabetic therapies for patients worldwide.
