Advanced Glycosylation Technology for Scalable Production of Complex Pharmaceutical Intermediates
Introduction to Next-Generation Glycosylation Technology
The landscape of carbohydrate chemistry is undergoing a significant transformation driven by the urgent need for more efficient and stable methods to construct glycosidic bonds, a cornerstone in the synthesis of bioactive oligosaccharides and glycoconjugates. Patent CN114891049A introduces a groundbreaking approach utilizing ortho-alkynyl benzyl ether-based glycosyl donors that address longstanding challenges in stability and activation. This technology enables the efficient generation of target glycosides through the reaction of these specialized donors with various nucleophiles under mild catalytic conditions. The innovation lies not only in the high efficiency of the glycosidic bond formation but also in the dual functionality of the leaving group, which serves as a robust terminal protecting group during synthetic sequences. 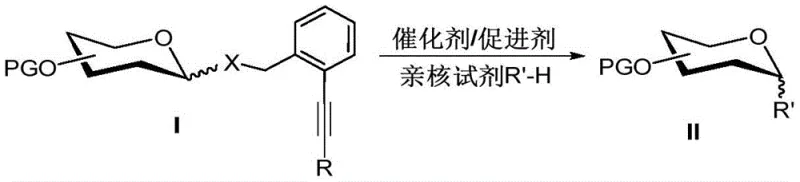 For R&D directors and process chemists, this represents a paradigm shift away from transient, unstable intermediates toward durable building blocks that streamline the overall synthetic route. By facilitating latent-active synthesis strategies, this method significantly accelerates the preparation of complex oligosaccharides, offering a reliable pathway for producing high-purity pharmaceutical intermediates required for drug development and biological research.
For R&D directors and process chemists, this represents a paradigm shift away from transient, unstable intermediates toward durable building blocks that streamline the overall synthetic route. By facilitating latent-active synthesis strategies, this method significantly accelerates the preparation of complex oligosaccharides, offering a reliable pathway for producing high-purity pharmaceutical intermediates required for drug development and biological research.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional glycosylation strategies, while foundational, suffer from inherent drawbacks that complicate large-scale manufacturing and process optimization. Methods such as the trichloroacetimidate ester approach, though widely used, involve leaving groups that are notoriously unstable and difficult to store for extended periods, leading to batch-to-batch variability and potential safety hazards. Similarly, the thioglycoside method, known for its stability, typically requires stoichiometric amounts of activators and involves precursors like thiols and thiophenols that possess unpleasant odors, posing significant environmental and occupational health challenges in a production facility. Furthermore, conventional approaches often necessitate a tedious sequence of installing a temporary terminal protecting group, removing it, and then introducing the leaving group immediately prior to glycosylation. This multi-step protection and deprotection cycle inevitably extends the synthetic route, reduces overall yield, and increases waste generation, making cost reduction in pharmaceutical intermediates manufacturing increasingly difficult to achieve with legacy technologies.
The Novel Approach
In stark contrast, the ortho-alkynyl benzyl ether methodology presented in the patent offers a streamlined solution that integrates stability with reactivity. The leaving group moiety in this system is exceptionally stable, allowing it to function effectively as a terminal protecting group that withstands standard synthetic manipulations without premature decomposition. This stability eliminates the need for repetitive protection and deprotection steps, thereby shortening the synthetic timeline and improving material throughput. Crucially, despite its stability, the group can be efficiently activated under the influence of various inexpensive catalysts or promoters, such as trifluoromethanesulfonic acid or metal triflates, to react with a broad spectrum of nucleophiles. 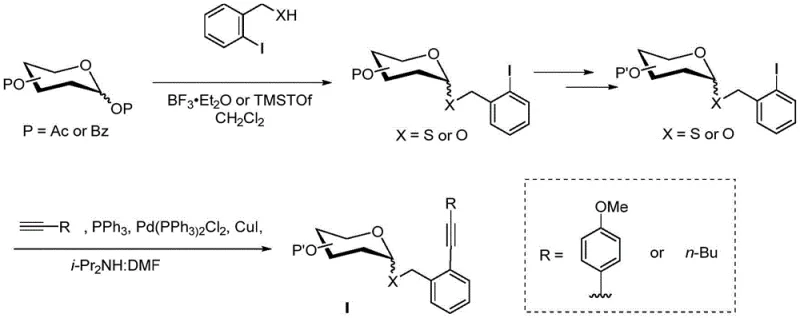 This dual nature supports a latent-active strategy, where the donor remains inert until specifically triggered, enabling the rapid assembly of oligosaccharides. For supply chain heads, this translates to a more robust inventory of intermediates that do not degrade over time, ensuring supply continuity and reducing the risk of production delays caused by reagent instability.
This dual nature supports a latent-active strategy, where the donor remains inert until specifically triggered, enabling the rapid assembly of oligosaccharides. For supply chain heads, this translates to a more robust inventory of intermediates that do not degrade over time, ensuring supply continuity and reducing the risk of production delays caused by reagent instability.
Mechanistic Insights into Catalytic Glycosidic Bond Formation
The mechanistic elegance of this glycosylation method lies in the unique electronic properties of the ortho-alkynyl benzyl ether system. Under the action of a Lewis acid catalyst or a promoter system like N-iodosuccinimide (NIS) combined with trifluoromethanesulfonic acid (TfOH), the alkyne moiety interacts with the electrophilic species to facilitate the cleavage of the glycosidic bond. This activation generates a reactive oxocarbenium ion intermediate while simultaneously stabilizing the departing leaving group through intramolecular interactions involving the ortho-substituent. The result is a highly controlled reaction environment that favors the formation of the desired glycosidic linkage with excellent stereoselectivity. Unlike traditional methods that might require harsh conditions or cryogenic temperatures to control selectivity, this process often proceeds efficiently at room temperature, as evidenced by the experimental data where reactions were completed within 0.5 to 24 hours under ambient conditions. This mildness is critical for preserving sensitive functional groups on complex carbohydrate scaffolds, ensuring the integrity of the final high-purity glycosyl donors.
Furthermore, the versatility of the catalyst system allows for fine-tuning the reactivity to match the nucleophilicity of the acceptor. Whether the acceptor is a hindered tertiary alcohol or a less reactive secondary hydroxyl group on a sugar ring, the choice of catalyst—ranging from mercury triflate to copper triflate—can be adjusted to optimize conversion rates. The patent details successful couplings with diverse nucleophiles, including sulfur- and nitrogen-containing compounds, demonstrating the broad substrate scope. This adaptability minimizes the formation of side products and simplifies downstream purification, a key factor in maintaining high purity specifications required for regulatory compliance in pharmaceutical applications. The ability to use catalytic amounts of activators rather than stoichiometric quantities also reduces the burden on waste treatment systems, aligning with modern green chemistry principles and environmental compliance standards.
How to Synthesize Ortho-Alkynyl Benzyl Ether Glycosyl Donors Efficiently
The synthesis of these advanced donors follows a logical and scalable sequence that begins with readily available starting materials such as o-iodobenzyl alcohol and peracetylated sugars. The process involves the initial conversion of the alcohol to a reactive halide or thioester, followed by glycosylation to install the sugar moiety, and finally, a palladium-catalyzed Sonogashira coupling to introduce the critical alkyne functionality. This modular approach allows for the late-stage diversification of the donor by simply changing the terminal alkyne, providing flexibility in designing libraries of glycosylating agents. The detailed standardized synthesis steps outlined below reflect the optimized conditions found in the patent examples, ensuring reproducibility and high yields suitable for commercial scale-up of complex oligosaccharides.
- Preparation of o-iodobenzyl thioacetate: React o-iodobenzyl alcohol with PBr3 in ether, followed by substitution with potassium thioacetate in methanol to yield compound 2.
- Glycosylation with sugar acceptor: React the thiol derivative with peracetylated mannose using BF3·Et2O in DCM to form the thioglycoside intermediate (compound 4).
- Sonogashira Coupling: Introduce the alkyne moiety via Pd-catalyzed coupling with terminal alkynes (e.g., 1-hexyne or 4-ethynylanisole) to finalize the ortho-alkynyl benzyl ether donor.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this ortho-alkynyl benzyl ether technology offers substantial strategic benefits that go beyond mere chemical efficiency. The inherent stability of the donors means that raw materials can be stocked in larger quantities without the fear of degradation, effectively decoupling production schedules from the immediate availability of fresh reagents. This stability significantly enhances supply chain reliability, allowing manufacturers to maintain consistent inventory levels and respond rapidly to fluctuating market demands for glycosylated APIs or research compounds. Moreover, the elimination of malodorous thiols and the reduction in stoichiometric activator usage simplify the manufacturing environment, reducing the need for specialized containment equipment and lowering operational overheads associated with hazardous material handling.
- Cost Reduction in Manufacturing: The streamlined synthetic route directly contributes to cost reduction in pharmaceutical intermediates manufacturing by minimizing the number of unit operations. By serving as both a leaving group and a protecting group, the ortho-alkynyl benzyl ether moiety removes the need for separate protection and deprotection steps, which traditionally consume significant amounts of reagents, solvents, and time. Additionally, the use of catalytic activation systems rather than stoichiometric promoters reduces the consumption of expensive activators and simplifies the workup procedure, leading to lower raw material costs and reduced waste disposal expenses. The high yields reported in the patent examples, often exceeding 90% for key coupling steps, further maximize material efficiency and reduce the cost per gram of the final product.
- Enhanced Supply Chain Reliability: The robustness of the intermediates ensures enhanced supply chain reliability by mitigating the risks associated with reagent instability. Traditional glycosyl donors often require cold chain logistics or have short shelf lives, creating vulnerabilities in the supply network. In contrast, these stable donors can be stored under standard conditions, simplifying logistics and reducing transportation costs. The broad substrate scope also means that a single donor platform can be adapted for multiple projects, reducing the need to source and qualify diverse sets of specialized reagents. This flexibility allows procurement teams to consolidate suppliers and negotiate better terms, ensuring a steady flow of high-quality materials for continuous production runs.
- Scalability and Environmental Compliance: The process is designed with scalability and environmental compliance in mind, featuring reactions that proceed at room temperature in common organic solvents like dichloromethane or DMF. The absence of extreme temperatures or pressures simplifies reactor requirements, making the transition from laboratory benchtop to pilot plant and full commercial production more straightforward. Furthermore, the reduction in hazardous waste generation, particularly from avoiding stoichiometric heavy metal activators and malodorous sulfur compounds, aligns with increasingly stringent environmental regulations. This compliance reduces the regulatory burden and potential liabilities associated with waste management, making the technology a sustainable choice for long-term manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this glycosylation technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the practical application of ortho-alkynyl benzyl ether donors in industrial settings. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the stability advantages of ortho-alkynyl benzyl ether donors compared to trichloroacetimidates?
A: Unlike trichloroacetimidates which are unstable and difficult to store long-term, ortho-alkynyl benzyl ether donors exhibit high stability and can function as terminal protecting groups, surviving conventional protection and deprotection operations without premature activation.
Q: Can this glycosylation method be used for both sugar and non-sugar acceptors?
A: Yes, the method is highly versatile. The patent demonstrates efficient glycosylation with carbohydrate primary or secondary alcohols as well as non-carbohydrate nucleophiles including primary, secondary, or tertiary alcohols, and sulfur- or nitrogen-containing nucleophiles.
Q: Does this method require stoichiometric amounts of activators?
A: No, a key advantage is the ability to operate under catalytic conditions. The leaving group can be activated by various catalysts such as triflic acid, copper triflate, or promoters like NIS/TfOH, often requiring only catalytic loading rather than stoichiometric reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ortho-Alkynyl Benzyl Ether Glycosyl Donors Supplier
As the demand for complex carbohydrate-based therapeutics continues to rise, having a partner with deep technical expertise and scalable manufacturing capabilities is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced synthetic methodologies like the ortho-alkynyl benzyl ether glycosylation to deliver superior results. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trial materials to full-scale market supply. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity glycosyl donors meets the exacting standards required for pharmaceutical applications, providing you with confidence in the quality and consistency of your supply chain.
We invite you to collaborate with us to explore how this innovative technology can optimize your specific synthesis routes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, demonstrating exactly how switching to this stable donor system can improve your bottom line. Please contact us today to request specific COA data for our available intermediates and to discuss route feasibility assessments for your target molecules. Let us help you accelerate your development timeline and secure a reliable supply of critical pharmaceutical intermediates for your future success.
