Advancing Chiral Pharmaceutical Intermediates Production with Novel BaSDR1 Carbonyl Reductase Technology
The landscape of chiral synthesis for pharmaceutical intermediates is undergoing a transformative shift towards greener, more efficient biocatalytic solutions, as exemplified by the technological breakthroughs detailed in patent CN110129382B. This pivotal intellectual property discloses a highly stereoselective method for synthesizing chiral ortho-halogenated-α-phenethyl alcohols, which serve as critical building blocks for a wide array of high-value therapeutic agents. These compounds are indispensable precursors in the manufacture of complex drugs, including α7nChR agonists like JN403 and potential treatments for Alzheimer's disease such as γ-secretase inhibitors. The patent introduces the application of a specific carbonyl reductase, designated as BaSDR1, or engineered bacteria containing this enzyme, to perform the asymmetric reduction of latent chiral ortho-halogenated acetophenones. By leveraging this biological catalyst, manufacturers can access a pathway that not only ensures exceptional optical purity but also aligns with the rigorous environmental and safety standards demanded by modern regulatory bodies. The structural versatility of the substrates accepted by this system is broad, encompassing various halogen substitutions that are often challenging to handle with traditional chemical methods.
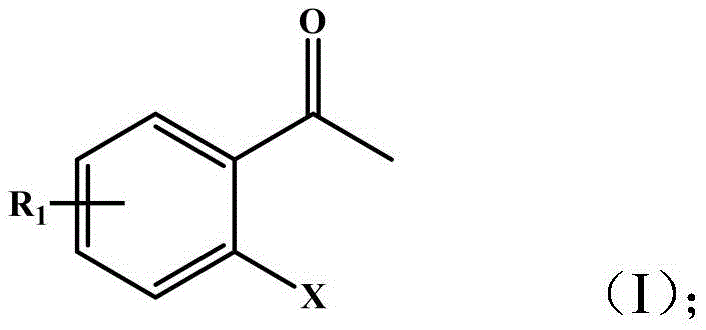
For R&D directors and process chemists evaluating synthetic routes, understanding the limitations of conventional methodologies is crucial for appreciating the value proposition of this novel biocatalytic approach. Traditional chemical asymmetric reduction methods, while theoretically capable of high yields, frequently suffer from significant practical drawbacks that hinder their scalability and economic viability. These chemical processes often rely on expensive and toxic transition metal catalysts, such as ruthenium or rhodium complexes, which necessitate stringent removal steps to meet residual metal specifications in final drug substances. Furthermore, chemical reductions typically require harsh reaction conditions, including high pressures of hydrogen gas or the use of hazardous hydride reagents, which introduce substantial safety risks and operational complexities in a manufacturing plant. The stereoselectivity of chemical catalysts can also be inconsistent across different substrate analogues, often leading to mixtures of enantiomers that require costly and yield-loss-inducing recrystallization or chromatographic separation steps to achieve the necessary optical purity.
In stark contrast, the novel approach utilizing carbonyl reductase BaSDR1 offers a paradigm shift in how these chiral alcohols are manufactured, addressing the core pain points of the legacy chemical routes. This biocatalytic method operates under remarkably mild conditions, typically within a temperature range of 20°C to 55°C and a pH window of 5.5 to 10.5, thereby eliminating the need for extreme pressure or temperature control systems. The enzyme exhibits extraordinary stereoselectivity, consistently delivering products with an enantiomeric excess (ee) of up to 99%, specifically favoring the (S)-enantiomer configuration which is often the pharmacologically active form. This high level of selectivity drastically simplifies the downstream purification process, as the formation of unwanted isomers is virtually negligible. Moreover, the use of engineered E. coli whole cells as the biocatalyst source provides a robust and cost-effective alternative to isolated enzymes, facilitating easier handling and recycling of the biocatalyst in industrial settings. The process utilizes benign co-substrates like glucose for cofactor regeneration, further enhancing the green chemistry profile of the synthesis.
Mechanistic Insights into BaSDR1-Catalyzed Asymmetric Reduction
To fully appreciate the technical sophistication of this invention, one must delve into the mechanistic underpinnings of the BaSDR1 carbonyl reductase derived from the strain Bacillus aryabhattai NWPU-1801. This enzyme functions as a highly specific biocatalyst that facilitates the transfer of a hydride ion from the reduced cofactor NADH or NADPH to the carbonyl carbon of the ortho-halogenated acetophenone substrate. The active site of the BaSDR1 enzyme is structurally configured to bind the pro-chiral ketone in a precise orientation that sterically favors the attack on one specific face of the carbonyl group, thereby enforcing the formation of the (S)-configured alcohol with near-perfect fidelity. This intrinsic stereocontrol is a result of millions of years of evolutionary optimization, which chemical catalysts struggle to mimic without extensive and costly ligand design. The reaction mechanism avoids the formation of radical intermediates that are common in metal-catalyzed reductions, thus preventing side reactions such as dehalogenation, which is a critical concern when working with ortho-halo substrates containing sensitive carbon-halogen bonds.
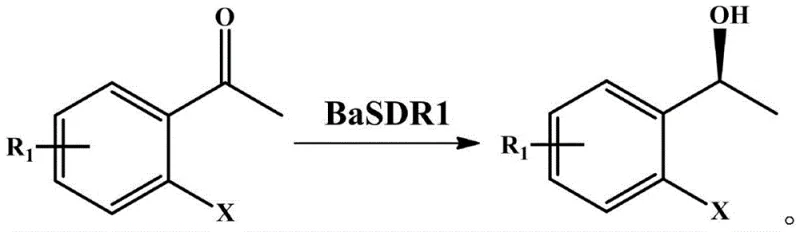
Furthermore, the impurity control mechanism inherent in this biocatalytic system is superior to chemical alternatives due to the enzyme's high chemoselectivity. In chemical reductions, the reactive metal hydrides or hydrogen gas can often reduce other functional groups present on the aromatic ring or interact with the halogen substituent, leading to a complex impurity profile that complicates purification. The BaSDR1 enzyme, however, targets exclusively the ketone functionality, leaving the ortho-halogen (fluorine, chlorine, or bromine) and other potential substituents on the phenyl ring intact. This specificity ensures that the crude reaction mixture contains primarily the desired chiral alcohol and unreacted starting material, significantly reducing the burden on purification units. The stability of the enzyme under the specified reaction conditions (pH 5.5-10.5) also prevents protein denaturation that could release intracellular proteases or other contaminants into the product stream. This clean reaction profile translates directly into higher overall process yields and reduced solvent consumption during workup, which are key metrics for sustainable manufacturing.
How to Synthesize Chiral Ortho-Halogenated-α-Phenethyl Alcohol Efficiently
Implementing this biocatalytic route in a production environment involves a streamlined sequence of operations that leverages the robustness of the engineered biocatalyst. The process begins with the preparation of a buffered aqueous reaction system, where the pro-chiral ortho-halogenated acetophenone substrate is introduced at concentrations ranging from 5 mmol/L to 300 mmol/L, demonstrating the system's capacity for high substrate loading. The biocatalyst, either as a purified enzyme or as wet whole cells of the engineered E. coli BL21(DE3)/pET30a-Basdr1 strain, is added to the mixture along with a co-substrate such as glucose to drive the cofactor regeneration cycle. Detailed standardized synthesis steps see below guide.
- Prepare the reaction system using a phosphate buffer (pH 5.5-10.5) containing the pro-chiral ortho-halogenated acetophenone substrate.
- Introduce the biocatalyst, either purified BaSDR1 enzyme or engineered E. coli whole cells, along with a co-substrate like glucose for cofactor regeneration.
- Maintain the reaction at mild temperatures (20-55°C) until completion, followed by extraction and purification to isolate the high-optical-purity chiral alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the BaSDR1 biocatalytic process represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with chiral intermediate manufacturing. The transition from chemical to biological catalysis eliminates the dependency on precious transition metals, the prices of which are subject to volatile geopolitical market fluctuations. By removing the need for expensive metal scavengers and the associated validation testing for residual metals, the overall cost of goods sold (COGS) is significantly reduced. Additionally, the mild reaction conditions allow for the use of standard stainless steel reactors without the need for specialized high-pressure hydrogenation equipment, lowering capital expenditure requirements for scale-up. The high conversion rates and optical purity achieved in a single step minimize the need for multiple recrystallizations or chiral separations, which are traditionally the most yield-loss-intensive stages of chiral synthesis. This efficiency gain results in a substantial reduction in raw material consumption and waste disposal costs, contributing to a more sustainable and economically resilient supply chain.
- Cost Reduction in Manufacturing: The elimination of toxic transition metal catalysts removes the necessity for costly metal removal resins and extensive analytical testing for heavy metal residues, leading to direct savings in both materials and quality control overhead. The use of glucose as a cheap and renewable co-substrate for cofactor regeneration further drives down the variable costs of the reaction compared to stoichiometric chemical reducing agents. The high stereoselectivity ensures that nearly all converted substrate becomes the desired product, maximizing atom economy and reducing the financial loss associated with discarding unwanted enantiomers. Consequently, the overall manufacturing process becomes leaner and more cost-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The biocatalyst is produced via fermentation of a stable engineered E. coli strain, which can be stored frozen for extended periods without significant loss of activity, ensuring a consistent and reliable supply of the catalyst for long-term production campaigns. The broad substrate tolerance of the BaSDR1 enzyme allows a single biocatalytic platform to be used for generating a family of related ortho-halogenated intermediates, simplifying inventory management and reducing the need for multiple specialized catalysts. The robustness of the whole-cell system against varying substrate concentrations provides flexibility in batch sizing, allowing manufacturers to respond agilely to fluctuating demand without compromising process performance or product quality.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium significantly reduces the volume of organic solvents required compared to traditional chemical synthesis, aligning with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The mild operating conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing process. The absence of hazardous reagents simplifies waste treatment protocols, as the effluent is primarily composed of biodegradable organic matter rather than toxic heavy metals or persistent chemical byproducts. This environmental compatibility facilitates smoother regulatory approvals and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic technology, providing clarity for stakeholders evaluating its integration into their supply chains. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and reliability for decision-making purposes. Understanding these details is essential for assessing the feasibility of adopting this green synthesis route for large-scale production of high-value chiral intermediates.
Q: What is the optical purity achievable with the BaSDR1 catalyzed process?
A: The patented process utilizing carbonyl reductase BaSDR1 consistently achieves an enantiomeric excess (ee) of up to 99% for the resulting chiral ortho-halogenated-α-phenethyl alcohols, ensuring high stereochemical integrity required for pharmaceutical applications.
Q: Does this biocatalytic method support diverse halogen substituents?
A: Yes, the BaSDR1 enzyme demonstrates broad substrate adaptability, effectively catalyzing the asymmetric reduction of ortho-fluoro, ortho-chloro, and ortho-bromo acetophenone derivatives, making it versatile for various API intermediate syntheses.
Q: How does this biological route compare to traditional chemical reduction regarding safety?
A: Unlike traditional chemical methods that often require toxic transition metal catalysts and harsh reaction conditions, this biocatalytic approach operates under mild aqueous conditions, eliminating heavy metal contamination risks and significantly reducing hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ortho-Halogenated-α-Phenethyl Alcohol Supplier
As the pharmaceutical industry continues to demand higher purity and more sustainable manufacturing practices, NINGBO INNO PHARMCHEM stands ready to leverage advanced technologies like the BaSDR1 biocatalytic system to meet your complex synthesis needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art chiral analysis capabilities to guarantee that every batch of chiral ortho-halogenated-α-phenethyl alcohol meets the exacting standards required for API synthesis. Our commitment to quality and innovation makes us a trusted partner for navigating the challenges of modern chiral chemistry.
We invite you to engage with our technical procurement team to discuss how this biocatalytic route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this enzymatic process for your specific molecule. We encourage you to contact us to obtain specific COA data for similar compounds and to receive comprehensive route feasibility assessments that will empower your R&D and supply chain teams to make informed strategic decisions. Let us collaborate to build a more efficient and sustainable future for your pharmaceutical manufacturing operations.
