Advanced Stereoselective Synthesis of Beta-Hydroxy Artemisinyl Ether for Commercial Scale-Up
Advanced Stereoselective Synthesis of Beta-Hydroxy Artemisinyl Ether for Commercial Scale-Up
The global pharmaceutical landscape continues to demand higher purity and more efficient synthetic routes for critical antimalarial intermediates, specifically within the artemisinin derivative class. Patent CN103664985A introduces a groundbreaking stereoselective preparation method for beta-hydroxy artemisinyl ether, a pivotal intermediate in the synthesis of next-generation antimalarial and anticancer agents. This technology addresses the longstanding challenge of controlling stereochemistry at the C10 position of the artemisinin scaffold, a critical factor determining biological activity. By shifting away from traditional non-selective dehydration methods, this novel approach utilizes a strategic acylation-condensation-hydrolysis sequence that inherently favors the formation of the thermodynamically stable beta-isomer. For R&D directors and process chemists, this represents a significant leap forward in impurity profile management, while supply chain leaders will recognize the potential for drastic reductions in processing time and solvent consumption associated with chromatographic purification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxy artemisinyl ethers has relied heavily on the direct acid-catalyzed dehydration condensation of dihydroartemisinin with various diol compounds. While conceptually straightforward, this conventional pathway suffers from inherent thermodynamic limitations that result in poor stereoselectivity. The reaction typically produces a complex mixture of both alpha and beta isomers, often with the undesired alpha-isomer constituting a significant portion of the crude product. Separating these stereoisomers is notoriously difficult due to their similar physical properties, frequently necessitating the use of preparative column chromatography. For industrial-scale manufacturing, reliance on chromatography is economically prohibitive and operationally inefficient, leading to substantial losses in yield, excessive solvent waste, and extended production cycles. Furthermore, the harsh acidic conditions often required for direct dehydration can compromise the sensitive peroxide bridge of the artemisinin core, leading to degradation products that complicate downstream purification and jeopardize the safety profile of the final API.
The Novel Approach
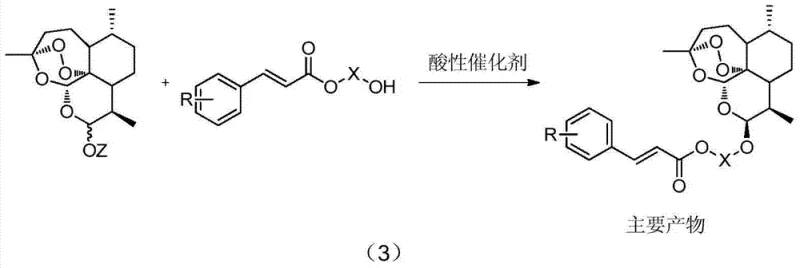
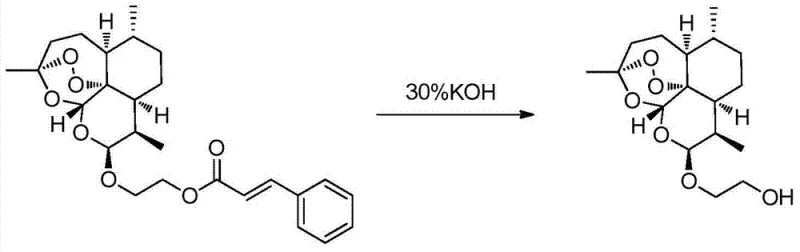
The methodology disclosed in CN103664985A circumvents these issues through a clever indirect strategy that leverages steric and electronic effects to drive stereoselectivity. Instead of reacting the diol directly with dihydroartemisinin, the process first converts the diol into a hydroxyl-substituted aryl acrylate via a mild acylation reaction. This functionalized intermediate then undergoes a condensation reaction with dihydroartemisinin (or its acetate/trifluoroacetate derivatives) under controlled acidic catalysis. Crucially, this specific reaction environment promotes the formation of the beta-isomer as the dominant product, with ratios often exceeding 90:10 in favor of the beta-form. The presence of the bulky aryl acrylate moiety appears to facilitate a highly efficient recrystallization step, allowing for the physical separation of the minor alpha-isomer to achieve optical purity greater than 99.9% without chromatography. Finally, a mild alkaline hydrolysis cleaves the ester linkage to release the target beta-hydroxy artemisinyl ether.
Mechanistic Insights into Acid-Catalyzed Stereoselective Etherification
The core of this technological advancement lies in the precise manipulation of the glycosidic bond formation at the C10 position of the artemisinin skeleton. The reaction mechanism involves the activation of the hemiacetal hydroxyl group of dihydroartemisinin by a Lewis or Brønsted acid catalyst, such as boron trifluoride ethyl ether complex or trifluoroacetic acid. This activation generates an oxocarbenium ion intermediate, which is then attacked by the hydroxyl group of the aryl acrylate derivative. The stereoselectivity is governed by the anomeric effect and the steric bulk of the incoming nucleophile. In this specific system, the aryl acrylate group likely stabilizes the transition state leading to the beta-anomer, or alternatively, the beta-anomer crystallizes preferentially from the reaction mixture due to favorable packing interactions introduced by the aromatic ring. The patent data indicates that varying the electronic nature of the aryl ring (e.g., introducing nitro or methoxy groups) allows for fine-tuning of the reaction kinetics and solubility profiles, providing process chemists with valuable handles for optimization. This level of control is absent in simple aliphatic diol reactions, where the lack of differentiating steric bulk leads to statistical mixtures.
Impurity control is further enhanced by the robustness of the aryl acrylate ester linkage, which remains stable during the acidic condensation but is readily cleaved under mild basic conditions in the final step. This orthogonality ensures that the sensitive endoperoxide bridge is never exposed to prolonged harsh alkaline conditions, which are known to degrade artemisinin derivatives. The recrystallization step serves as a powerful purification gate; because the alpha and beta isomers of the aryl acrylate intermediate have significantly different solubilities in solvents like petroleum ether or hexane, simple filtration can remove the alpha-impurity. This mechanistic understanding allows for the design of a 'quality by design' process where purity is built into the synthesis rather than inspected in at the end, a critical consideration for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Beta-Hydroxy Artemisinyl Ether Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and temperature control across four distinct operational units. The process begins with the acylation of a diol (such as ethylene glycol or propylene glycol) with an aryl acryloyl chloride in the presence of a base like triethylamine or sodium carbonate, typically conducted at temperatures between -10°C and 50°C to minimize side reactions. The resulting hydroxyl ester is then coupled with dihydroartemisinin using a catalytic amount of acid (e.g., 0.01 to 1 equivalent) in a non-protic solvent like dichloromethane or chloroform. Following the condensation, the crude solid is subjected to recrystallization from a mixed solvent system, which is the critical step for achieving high stereochemical purity. The final deprotection is achieved using aqueous alkali, yielding the free alcohol. For a detailed breakdown of the specific molar ratios, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized protocol below.
- Perform acylation of a diol compound with aryl acryloyl chloride under basic conditions to form a hydroxyl-substituted aryl acrylate.
- React the hydroxyl-substituted aryl acrylate with dihydroartemisinin (or its esters) using an acidic catalyst to stereoselectively generate a beta-dominant mixture.
- Purify the mixture via recrystallization to isolate optically pure beta-isomer, followed by alkaline hydrolysis to yield the final beta-hydroxy artemisinyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this stereoselective route offers transformative benefits for the procurement of high-purity pharmaceutical intermediates. The most significant advantage is the complete elimination of column chromatography, a unit operation that is notoriously difficult to scale and incredibly costly in terms of silica gel, solvents, and labor. By replacing chromatography with a simple crystallization and filtration step, manufacturers can achieve substantial cost savings in both raw materials and waste disposal. This shift not only lowers the cost of goods sold (COGS) but also drastically reduces the environmental footprint of the manufacturing process, aligning with modern green chemistry principles and regulatory expectations for sustainable production. Furthermore, the ability to produce optically pure material directly from the reactor train simplifies the supply chain, reducing the number of processing stages and the associated risk of batch failure or contamination.
- Cost Reduction in Manufacturing: The economic impact of removing chromatographic purification cannot be overstated. Traditional methods require vast quantities of organic solvents for elution and significant amounts of stationary phase, both of which represent major variable costs. By utilizing a crystallization-driven purification strategy, this process minimizes solvent usage and eliminates the need for expensive silica gel entirely. Additionally, the high stereoselectivity means that less starting material is wasted on forming the unwanted alpha-isomer, thereby improving the overall atom economy and effective yield of the valuable dihydroartemisinin starting material. These factors combine to create a leaner, more cost-effective manufacturing model that enhances margin potential for downstream API producers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable reagents. The aryl acryloyl chlorides and diols required for the initial acylation are commodity chemicals with robust global supply networks, reducing the risk of raw material shortages. Moreover, the reaction conditions are relatively mild and tolerant, operating effectively at temperatures ranging from -20°C to 50°C, which reduces the energy burden on manufacturing facilities compared to processes requiring cryogenic cooling or high-temperature reflux. The robustness of the recrystallization step ensures consistent product quality across different batches and scales, mitigating the risk of supply disruptions caused by failed purification runs. This reliability is crucial for maintaining continuous production schedules for essential antimalarial medications.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is straightforward due to the absence of complex separation technologies. Crystallization and filtration are unit operations that scale linearly and predictably, unlike chromatography which faces significant engineering challenges at large volumes. The process also generates less hazardous waste, as the primary byproducts are salts and water from the neutralization and hydrolysis steps, which are easier to treat than the complex organic waste streams generated by column chromatography. This simplified waste profile facilitates compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste disposal, ensuring long-term operational viability in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective synthesis technology. These insights are derived directly from the experimental data and claims within Patent CN103664985A, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement officers assessing supplier capabilities.
Q: Why is the stereoselectivity of beta-hydroxy artemisinyl ether difficult to control in traditional methods?
A: Traditional direct dehydration condensation of dihydroartemisinin with diols typically yields a mixture of alpha and beta isomers with poor selectivity, making it difficult to obtain the beta-isomer without expensive column chromatography.
Q: What represents the key innovation in Patent CN103664985A regarding impurity control?
A: The key innovation is the introduction of an aryl acrylate protecting group which facilitates highly efficient recrystallization, allowing for the removal of the alpha-isomer to achieve >99.9% optical purity without chromatography.
Q: Which acidic catalysts are suitable for this stereoselective condensation reaction?
A: The patent specifies several effective acidic catalysts including boron trifluoride ethyl ether complex, trifluoroacetic acid, tosic acid, and tin tetrachloride, providing flexibility for process optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Hydroxy Artemisinyl Ether Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic and patent research into commercially viable manufacturing processes. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate stereoselective requirements of artemisinin derivatives are met with precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical alpha/beta isomer ratios and peroxide content essential for antimalarial efficacy. We understand that the transition from bench-scale synthesis to multi-ton production requires not just chemical expertise but also deep engineering knowledge to manage heat transfer, mixing, and crystallization dynamics effectively.
We invite pharmaceutical companies and agrochemical innovators to collaborate with us to leverage this patented technology for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and comprehensive route feasibility assessments. By partnering with us, you secure a supply of high-purity intermediates that meets global regulatory standards while benefiting from a greener, more efficient manufacturing process.
