Advanced Chiral Sulfonamide Organocatalysts for Scalable Asymmetric Synthesis
Advanced Chiral Sulfonamide Organocatalysts for Scalable Asymmetric Synthesis
The landscape of asymmetric synthesis is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and cost-effective catalytic systems. Patent CN101569864B introduces a groundbreaking class of chiral sulfonamide organic small molecule catalysts that address critical limitations found in traditional metal-catalyzed processes. These novel catalysts, characterized by a unique sulfonamide group acting as a dual hydrogen bond donor, offer a robust solution for the asymmetric conjugate addition of aldehydes and ketones to nitroolefins. By leveraging specific chiral structures, this technology achieves exceptional enantioselectivity and chemical yields, positioning it as a vital tool for the synthesis of complex chiral medicaments and natural products. For R&D directors and procurement specialists alike, this innovation represents a strategic opportunity to streamline synthetic routes while adhering to increasingly stringent environmental and safety regulations.
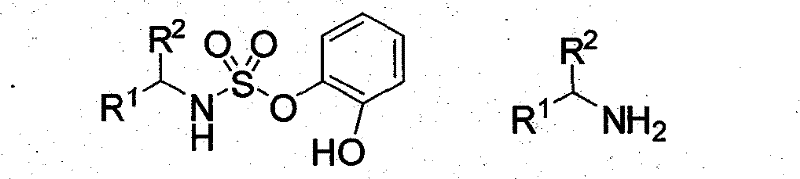
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral carbon centers in pharmaceutical intermediates has relied heavily on transition metal catalysis or biocatalysis, both of which present distinct operational challenges for large-scale manufacturing. Metal-catalyzed reactions often necessitate the use of expensive precious metals such as palladium, rhodium, or ruthenium, which not only inflate raw material costs but also introduce significant toxicity concerns that require rigorous downstream purification to meet regulatory limits. Furthermore, many metal-catalyzed processes demand strictly anhydrous and oxygen-free conditions, imposing heavy burdens on facility infrastructure and operational complexity. On the other hand, while biocatalysis offers high selectivity, it frequently suffers from a narrow substrate scope and sensitivity to reaction conditions, limiting its versatility across diverse chemical libraries. These factors collectively contribute to extended lead times and elevated production costs, creating bottlenecks in the supply chain for high-value chiral compounds.
The Novel Approach
The chiral sulfonamide catalyst described in this patent offers a paradigm shift by utilizing organic small molecule catalysis, which effectively bypasses the drawbacks associated with metal and enzymatic systems. This approach employs a sulfonamide moiety as a new type of double hydrogen bond donor, which activates substrates through non-covalent interactions rather than coordination with metal centers. The synthesis of these catalysts is straightforward, involving the reaction of readily available amines with pyrocatechol sulfonic acid esters, followed by coupling with chiral diamines. This methodology does not require inert atmospheres or specialized equipment, significantly simplifying the operational protocol. Consequently, manufacturers can achieve high enantioselectivity and excellent chemical yields under mild conditions, thereby reducing the overall environmental footprint and enhancing the economic viability of producing complex chiral intermediates.
Mechanistic Insights into Sulfonamide-Catalyzed Conjugate Addition
The efficacy of the chiral sulfonamide catalyst stems from its ability to function as a potent dual hydrogen bond donor, a mechanism that mimics the active sites of many enzymes. In the catalytic cycle, the sulfonamide group forms strong hydrogen bonds with the nitro group of the nitroolefin substrate, thereby lowering the energy of the lowest unoccupied molecular orbital (LUMO) and activating the electrophile for nucleophilic attack. Simultaneously, the chiral backbone of the catalyst, often derived from cyclohexanediamine derivatives, creates a well-defined steric environment that directs the approach of the nucleophile, such as an enamine formed from an aldehyde. This precise spatial arrangement ensures that the reaction proceeds through a highly organized transition state, resulting in the preferential formation of one enantiomer over the other. The result is a reaction profile that combines the broad substrate tolerance of small molecules with the high stereocontrol typically associated with biological systems.
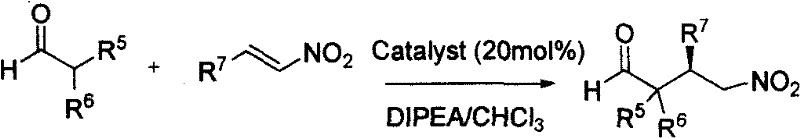
Furthermore, the impurity profile generated by this organocatalytic process is markedly cleaner compared to metal-mediated alternatives. Since no transition metals are involved, there is no risk of metal leaching or the formation of metal-containing byproducts that are notoriously difficult to remove. The primary impurities tend to be unreacted starting materials or diastereomers, which can often be separated through standard crystallization or chromatographic techniques. This inherent purity advantage is crucial for pharmaceutical applications, where residual metal limits are strictly enforced by agencies such as the FDA and EMA. By eliminating the need for scavengers or complex filtration steps to remove trace metals, the process not only improves the quality of the final API intermediate but also reduces the number of unit operations required, leading to a more streamlined and cost-efficient manufacturing workflow.
How to Synthesize Chiral Sulfonamide Catalyst Efficiently
The preparation of these high-performance catalysts follows a concise two-step synthetic route that is amenable to scale-up. The process begins with the sulfonylation of a primary or secondary amine using a catechol sulfonate ester in the presence of an organic base, yielding a stable sulfonamide intermediate. This intermediate is then subjected to a substitution reaction with a chiral diamine under reflux conditions to install the critical stereogenic elements. The simplicity of this route allows for the rapid generation of a library of catalyst variants by simply modifying the R groups on the starting amine. For detailed procedural specifics regarding stoichiometry, solvent choices, and purification methods, please refer to the standardized synthesis guide provided below.
- React an amine (Formula III) with catechol sulfonate in the presence of an organic base and DMF for 2-72 hours to obtain the intermediate compound (Formula II).
- Reflux the obtained intermediate (Formula II) with an equivalent amount of chiral diamine, such as (1R,2R)-cyclohexanediamine, in a solvent for 2-4 hours.
- Purify the resulting mixture via column chromatography to isolate the final chiral sulfonamide catalyst (Formula I) as a white solid.
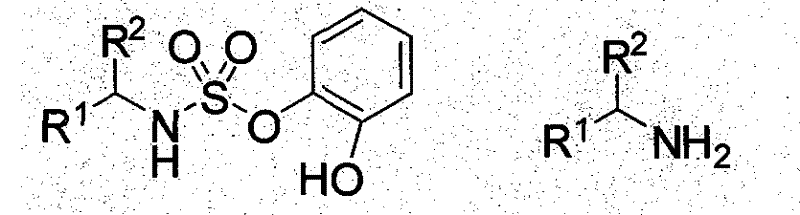
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this chiral sulfonamide catalyst technology offers substantial strategic benefits for procurement and supply chain management. The elimination of precious metal catalysts directly translates to a significant reduction in raw material volatility and cost, as the price of organic starting materials is generally more stable and predictable than that of rare earth metals. Additionally, the robustness of the reaction conditions means that production can be carried out in standard glass-lined or stainless steel reactors without the need for specialized inert gas blanketing, thereby maximizing asset utilization and reducing capital expenditure requirements. This operational flexibility enhances supply chain resilience, allowing for faster response times to market demands and reducing the risk of production delays caused by equipment constraints or catalyst shortages.
- Cost Reduction in Manufacturing: The transition to metal-free organocatalysis removes the necessity for expensive heavy metal scavengers and the associated validation testing for residual metals, which are costly and time-consuming processes. By simplifying the downstream purification train, manufacturers can achieve substantial cost savings in both consumables and labor. The high atom economy and yield of the reaction further contribute to lower cost of goods sold (COGS), making the final chiral intermediates more competitive in the global marketplace. Moreover, the catalyst loading can often be optimized to low levels without sacrificing performance, extending the value derived from each batch of catalyst produced.
- Enhanced Supply Chain Reliability: The starting materials for synthesizing these sulfonamide catalysts, such as substituted amines and catechol derivatives, are commodity chemicals with well-established global supply chains. This availability mitigates the risk of supply disruptions that are common with specialized ligands or bio-engineered enzymes. The stability of the catalyst itself allows for longer shelf life and easier storage and transportation logistics, reducing waste and inventory carrying costs. For supply chain heads, this reliability ensures consistent production schedules and the ability to secure long-term contracts with key customers who demand uninterrupted supply of critical pharmaceutical ingredients.
- Scalability and Environmental Compliance: The process operates under mild conditions and generates minimal hazardous waste, aligning perfectly with modern green chemistry principles and environmental regulations. The absence of toxic metals simplifies waste treatment protocols and reduces the environmental compliance burden on manufacturing sites. This eco-friendly profile is increasingly becoming a prerequisite for doing business with major multinational pharmaceutical companies that have aggressive sustainability goals. The scalability of the reaction from gram to multi-ton scale has been demonstrated, proving that the technology is ready for immediate commercial deployment without the need for extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral sulfonamide catalyst technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable foundation for decision-making. Understanding these aspects is essential for evaluating the feasibility of integrating this catalytic system into existing production workflows.
Q: What are the advantages of this sulfonamide catalyst over traditional metal catalysts?
A: This organocatalyst eliminates the need for expensive and toxic transition metals, avoiding complex heavy metal removal steps and ensuring higher purity profiles suitable for pharmaceutical applications.
Q: What types of reactions can this catalyst facilitate?
A: It is highly effective for asymmetric conjugate addition reactions between aldehydes or ketones and nitroolefins, delivering high enantioselectivity and chemical yields.
Q: Is this catalyst suitable for large-scale industrial production?
A: Yes, the synthesis involves simple operational conditions without stringent anhydrous or oxygen-free requirements, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Sulfonamide Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in modern drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of catalyst or intermediate meets the highest industry standards. We are committed to supporting your R&D efforts with custom synthesis services that leverage cutting-edge technologies like the chiral sulfonamide system to accelerate your timeline to market.
We invite you to collaborate with our technical team to explore how this metal-free catalytic solution can optimize your specific synthetic challenges. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current manufacturing processes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible value and performance capabilities of our catalyst offerings. Let us help you build a more sustainable and cost-effective supply chain for your chiral pharmaceutical intermediates.
