Scalable Synthesis of 5 Alpha-Pregnane-3 Beta Impurity for Advanced Pharmaceutical Quality Control
Scalable Synthesis of 5 Alpha-Pregnane-3 Beta Impurity for Advanced Pharmaceutical Quality Control
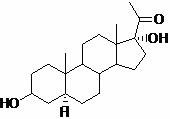
The pharmaceutical industry's relentless pursuit of high-purity active pharmaceutical ingredients (APIs) necessitates the development of robust reference standards and impurity profiles, particularly for complex steroid therapeutics like Ganaxolone. Patent CN116041409A introduces a groundbreaking preparation method for 5 alpha-pregnane-3 beta, 17 alpha-dihydroxyl-20-ketone, a critical impurity used to ensure the quality and safety of 5 alpha-pregnane-3, 20-dione synthesis. This technical breakthrough addresses the historical scarcity of reliable literature regarding the specific preparation of this stereoisomer, offering a reproducible pathway that leverages mono-enolone acetate as a cost-effective starting material. By establishing a clear five-step reaction sequence culminating in a refined product with verified structural integrity, this innovation provides essential support for regulatory compliance and quality assurance protocols in neuroactive steroid manufacturing.
For procurement specialists and supply chain managers, the availability of such well-defined synthetic routes translates directly into supply chain resilience. The ability to synthesize specific impurities in-house or source them from capable partners reduces dependency on unpredictable external vendors who may struggle with the nuanced stereochemistry of pregnane derivatives. This patent outlines a method that not only fills a gap in the scientific literature but also establishes a foundation for commercial-scale production of reference materials, ensuring that pharmaceutical manufacturers can maintain rigorous quality control standards without facing bottlenecks in their analytical validation processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of specific steroid impurities has been fraught with challenges related to stereochemical control and hazardous reaction conditions. Conventional methods for modifying the D-ring of pregnane steroids often rely on high-pressure hydrogenation or harsh acidic conditions that can lead to epimerization, resulting in mixtures of isomers that are difficult to separate. Furthermore, many existing protocols lack detailed optimization for the specific 17-alpha-hydroxyl configuration required for this impurity, leading to low yields and inconsistent purity profiles. The reliance on gaseous hydrogen in reduction steps introduces significant safety risks and requires specialized high-pressure reactors, which increases capital expenditure and complicates the scale-up process for fine chemical manufacturers aiming to produce reference standards efficiently.
The Novel Approach
The methodology disclosed in CN116041409A represents a paradigm shift by introducing a safer and more controllable debromination strategy that eliminates the need for external hydrogen gas. By utilizing Raney nickel in an acetone solvent system under reflux conditions, the process achieves the necessary reduction through a mechanism that likely involves hydrogen transfer from the solvent or surface-adsorbed species, thereby avoiding the complexities of high-pressure infrastructure. This approach not only enhances operational safety but also improves the selectivity of the reaction, preserving the delicate 3-beta and 17-alpha stereochemistry essential for the molecule's function as a reference standard. The integration of this unique step into a streamlined five-step sequence demonstrates a sophisticated understanding of steroid chemistry, offering a viable alternative to legacy methods that are often too cumbersome for modern GMP environments.
Mechanistic Insights into Raney Nickel-Mediated Debromination
The core innovation of this synthesis lies in the mechanistic nuances of the debromination step, which dictates the final stereochemical outcome of the D-ring modification. In this protocol, the brominated intermediate undergoes reductive debromination in the presence of Raney nickel, a highly active catalyst known for its ability to facilitate hydrogenolysis. Unlike traditional catalytic hydrogenation which requires pressurized H2, this system operates under atmospheric pressure in refluxing acetone, suggesting a transfer hydrogenation mechanism or a surface-mediated reduction where the catalyst activates C-H bonds from the solvent or residual hydrides. This mild yet effective reduction environment prevents the epimerization of the adjacent chiral centers, ensuring that the 17-alpha-hydroxyl group remains intact while the bromine atom is cleanly removed to restore the ketone functionality at the C20 position.
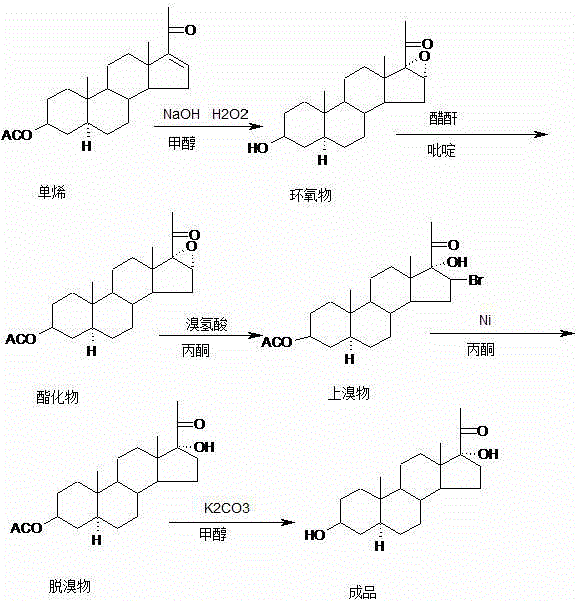
Furthermore, the preceding epoxidation and bromination steps are meticulously designed to set up the correct geometry for this final reduction. The initial epoxidation of the delta-16 double bond using alkaline hydrogen peroxide creates a strained oxirane ring that is subsequently opened and functionalized to introduce the bromine atom with high regioselectivity. This precise positioning of the leaving group is crucial, as it allows the Raney nickel to access the carbon-bromine bond without disturbing the rest of the steroid skeleton. The subsequent hydrolysis step using potassium carbonate in methanol then removes the acetate protecting group under mild basic conditions, revealing the free 3-beta-hydroxyl group without inducing elimination side reactions, thereby securing the high purity profile observed in the final crystalline product.
How to Synthesize 5 Alpha-Pregnane-3 Beta Efficiently
The synthesis of this high-value impurity requires strict adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process begins with the careful control of temperature during the epoxidation phase, followed by precise stoichiometric addition of reagents in the esterification and bromination stages. The critical debromination step demands attention to catalyst loading and reflux duration to achieve complete conversion without over-reduction. For detailed operational procedures, including specific solvent volumes, addition rates, and workup protocols, please refer to the standardized synthesis guide below which breaks down the laboratory-to-pilot scale translation.
- Epoxidation of mono-enolone acetate using sodium hydroxide and hydrogen peroxide in methanol at 30-50°C.
- Esterification of the epoxy intermediate with acetic anhydride and pyridine, followed by bromination using hydrobromic acid in acetone.
- Critical debromination step utilizing Raney nickel in acetone under reflux without hydrogen gas, followed by hydrolysis and ethanol refining.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for organizations managing the supply chain of neuroactive steroid intermediates. The elimination of high-pressure hydrogenation equipment significantly lowers the barrier to entry for manufacturing this compound, allowing a broader range of chemical suppliers to produce it reliably. This increased manufacturability fosters a more competitive market environment, which typically drives down costs and improves availability for downstream pharmaceutical users. Additionally, the use of common industrial solvents like methanol, acetone, and ethanol simplifies the procurement of raw materials, reducing the risk of supply disruptions associated with exotic or highly regulated reagents.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive high-pressure reactors and the associated safety infrastructure required for gaseous hydrogen handling. By utilizing Raney nickel in a refluxing solvent system, the method reduces energy consumption and capital investment, translating into lower production costs per kilogram. Furthermore, the high overall yield reported in the examples minimizes raw material waste, ensuring that the cost of goods sold remains competitive even when producing small batches of high-purity reference standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like mono-enolone acetate and common reagents ensures a stable supply chain that is less susceptible to geopolitical or logistical fluctuations. The robustness of the five-step sequence means that production can be scaled up or down rapidly in response to market demand without requiring complex process re-validation. This flexibility is crucial for pharmaceutical companies that need to secure long-term supplies of impurity standards for ongoing stability testing and regulatory filings.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram commercial production without fundamental changes to the chemistry. The avoidance of heavy metal catalysts other than the recoverable Raney nickel, combined with the use of relatively benign solvents, simplifies waste treatment and environmental compliance. This aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing process and facilitating easier permitting for production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific steroid impurity. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy for R&D and procurement decision-making. Understanding these details is essential for integrating this material into your quality control workflows effectively.
Q: What is the primary advantage of the debromination step in this patent?
A: The process utilizes Raney nickel in acetone under reflux conditions without the need for external hydrogen gas, significantly enhancing operational safety and reducing equipment complexity compared to traditional hydrogenation.
Q: Why is 5 alpha-pregnane-3 beta, 17 alpha-dihydroxyl-20-ketone important?
A: It serves as a critical reference substance and impurity standard for controlling the quality of 5 alpha-pregnane-3, 20-dione, a key intermediate in the synthesis of the anti-epileptic drug Ganaxolone.
Q: What is the reported purity and yield of this method?
A: According to the provided data, the method achieves a total yield of approximately 71.9% with an HPLC purity of 96.5% after final refining in ethanol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5 Alpha-Pregnane-3 Beta Supplier
As the demand for high-purity reference standards in the neuroactive steroid sector continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to adapt complex laboratory routes like the one described in CN116041409A to industrial manufacturing environments. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with advanced analytical instrumentation, guaranteeing that every batch of 5 alpha-pregnane-3 beta, 17 alpha-dihydroxyl-20-ketone meets the exacting standards required for pharmaceutical regulatory submissions.
We invite global pharmaceutical partners to collaborate with us to secure a stable supply of this critical intermediate. By leveraging our technical expertise, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain for Ganaxolone-related intermediates remains robust, compliant, and cost-efficient.
