Advanced Synthesis and Purification of D-3-Thioacetyl-2-Methylpropionyl-L-Proline for High-Purity Captopril Production
Introduction to Advanced Intermediate Synthesis
The pharmaceutical industry continuously seeks robust synthetic routes for critical antihypertensive agents, particularly for the ACE inhibitor Captopril. A pivotal breakthrough in this domain is documented in patent CN101289417B, which discloses a novel process for preparing D-3-thioacetyl-2-methylpropionyl-L-proline. This compound serves as the chiral key intermediate in the synthesis of Captopril, and its stereochemical purity directly dictates the therapeutic efficacy and safety profile of the final drug substance. The patented methodology represents a significant departure from legacy resolution techniques, introducing a sophisticated pH-controlled extraction and crystallization protocol that simultaneously enhances both chemical yield and optical purity. By optimizing the partitioning behavior of diastereomers between aqueous and organic phases, this technology addresses long-standing bottlenecks in the manufacturing of sulfhydryl-containing proline derivatives.
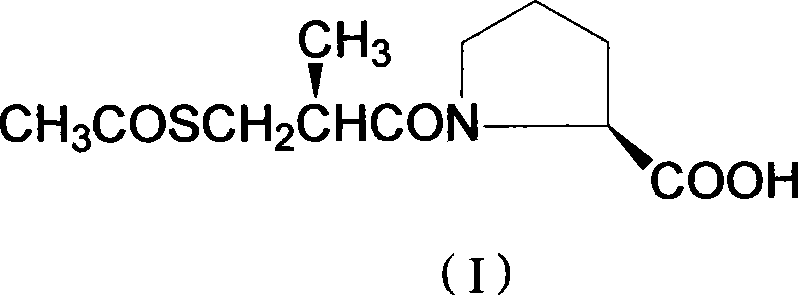
For procurement specialists and supply chain directors, the implications of this technological advancement are profound. The ability to consistently produce high-purity intermediates reduces the burden on downstream purification steps, thereby lowering the overall cost of goods sold (COGS) for the active pharmaceutical ingredient (API). As a reliable pharmaceutical intermediate supplier, understanding the nuances of such proprietary processes allows us to offer superior quality materials that meet the stringent regulatory requirements of global health authorities. The transition from older, less efficient resolution methods to this streamlined approach underscores a commitment to process intensification and green chemistry principles, minimizing waste generation while maximizing resource utilization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of racemic or diastereomeric mixtures of 3-acetylthio-2-methylpropionyl-L-proline has been fraught with inefficiencies. Early methodologies, such as those described in U.S. Patent 4,046,889, relied heavily on the formation of diastereomeric salts using resolving agents like dicyclohexylamine. While chemically effective, this approach introduces significant operational complexity, requiring additional salification and free-basing steps that increase solvent consumption and processing time. Furthermore, alternative methods utilizing simple solubility differences in water, as seen in Chinese Patent ZL89108856 (CN1021044C), often suffer from suboptimal recovery rates. In these conventional water-based crystallizations, the yield of the desired D-isomer typically hovers around 28% to 36%, with optical rotation values frequently falling short of the ideal specification, often ranging between -150° and -155°. These limitations not only inflate production costs but also compromise the consistency of the final API quality.
The Novel Approach
The innovative process outlined in CN101289417B circumvents these drawbacks through a clever manipulation of phase behavior. Instead of relying solely on precipitation from water, the new method involves adjusting the pH of the reaction mixture to a highly acidic range of 0.01 to 3.0. This acidification induces the separation of an oil reservoir rich in the target diastereomers, which is then physically separated from the bulk aqueous phase. Subsequently, the remaining aqueous layer is subjected to extraction with ethyl acetate, effectively scavenging any dissolved product that did not separate as oil. By merging the oil reservoir with the ethyl acetate extract and subjecting the combined organic phase to controlled cooling crystallization, the process achieves a remarkable enhancement in performance. Yields are consistently pushed to the 36.5% to 41% range, and optical purity is significantly elevated, with specific rotation values reaching -165° to -170°. This demonstrates a clear superiority over prior art in terms of both economic and quality metrics.
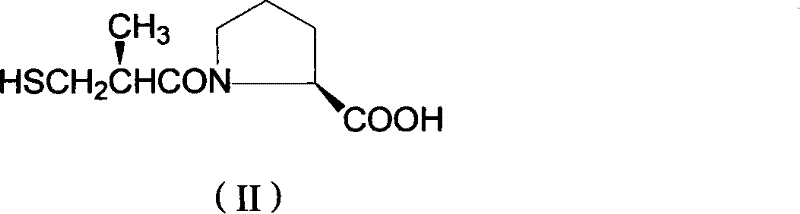
Mechanistic Insights into Diastereomeric Resolution and Crystallization
The core of this technological advancement lies in the precise control of the ionization state of the carboxylic acid moiety within the proline derivative. At neutral or alkaline pH, the molecule exists primarily as a carboxylate anion, which exhibits high solubility in water, making isolation difficult without the use of bulky counter-ions. By dropping the pH to the 0.01–3.0 range, the carboxyl group is fully protonated, drastically reducing the molecule's polarity and water solubility. This shift triggers the "oiling out" phenomenon, where the hydrophobic D-isomer preferentially separates from the aqueous matrix as a dense liquid phase. This physical separation acts as a primary enrichment step, removing a significant portion of water-soluble impurities and the more soluble L-isomer which remains in the mother liquor. The subsequent ethyl acetate extraction serves as a secondary polishing step, ensuring that no valuable product is lost to the aqueous waste stream.
Furthermore, the crystallization kinetics play a vital role in defining the final impurity profile. The patent specifies a cooling regime from 0°C down to -20°C, often aided by the introduction of seed crystals. This controlled supersaturation prevents the rapid, chaotic precipitation that can trap impurities within the crystal lattice. Instead, it promotes the orderly growth of the thermodynamically stable D-isomer crystals. The use of ethyl acetate as the crystallization solvent is particularly advantageous due to its favorable boiling point and solubility characteristics, which facilitate easy removal and recycling. This mechanistic understanding allows for cost reduction in pharmaceutical intermediate manufacturing by minimizing the need for repetitive recrystallizations, which are traditionally the most yield-eroding steps in chiral synthesis.
How to Synthesize D-3-Thioacetyl-2-Methylpropionyl-L-Proline Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and phase separations to replicate the high yields reported in the patent literature. The process begins with the acylation of L-proline, followed by the critical pH adjustment and extraction sequence that defines the invention. Operators must ensure precise temperature control during the exothermic acylation step and maintain strict pH monitoring during the resolution phase to maximize the partitioning coefficient. The following guide outlines the standardized operational framework derived from the patent examples, serving as a baseline for process development and scale-up activities.
- Condense 3-acetylthio-2-methyl-propionyl chloride with L-proline in aqueous alkali at -5 to 20°C to form the diastereomeric mixture.
- Adjust the pH of the aqueous solution to 0.01-3.0 to precipitate an oil reservoir containing the target diastereomers.
- Extract the remaining aqueous layer with ethyl acetate, merge with the oil reservoir, and crystallize at -5 to -20°C to isolate the pure D-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages beyond mere technical specifications. The elimination of expensive chiral resolving agents like dicyclohexylamine removes a significant cost driver from the bill of materials. Additionally, the simplified workup procedure reduces the consumption of auxiliary chemicals and minimizes the volume of wastewater generated, aligning with increasingly stringent environmental regulations. This streamlined workflow enhances the overall agility of the supply chain, allowing for faster batch turnover and more reliable delivery schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing costly resolving agents with commodity solvents like ethyl acetate and hydrochloric acid. By avoiding the formation and subsequent breaking of amine salts, the number of unit operations is reduced, leading to lower energy consumption and labor costs. The improved yield directly lowers the cost per kilogram of the intermediate, providing a competitive edge in pricing negotiations for bulk contracts.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-hazardous raw materials ensures a robust supply chain that is less susceptible to market volatility. Unlike specialized chiral auxiliaries that may have limited suppliers, ethyl acetate and mineral acids are globally sourced commodities. This availability mitigates the risk of production stoppages due to raw material shortages, ensuring continuous availability of high-purity intermediates for critical hypertension medications.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively across various batch sizes without loss of efficiency. The reduction in organic waste and the avoidance of heavy metal catalysts or complex amines simplify waste treatment protocols. This environmental compatibility facilitates easier regulatory approval and permits for manufacturing sites, supporting the long-term sustainability of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of D-3-thioacetyl-2-methylpropionyl-L-proline. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the process capabilities and quality attributes.
Q: How does the new purification method improve upon traditional solubility-based separation?
A: Unlike traditional methods relying solely on water solubility differences which often result in lower yields (around 28-36%) and inferior optical purity, the patented process utilizes a specific pH range (0.01-3.0) combined with ethyl acetate extraction. This dual-phase approach effectively removes water-soluble impurities and enriches the D-isomer, achieving optical rotations of -165° to -170° and yields up to 41%.
Q: What are the critical process parameters for maximizing optical purity?
A: Critical parameters include strict pH control between 1.0 and 2.0 during the oil separation phase, the use of ethyl acetate for both washing and extraction to remove non-polar impurities, and controlled crystallization temperatures between -5°C and -20°C. The addition of seed crystals during the cooling phase is also recommended to ensure consistent polymorphic form and high enantiomeric excess.
Q: Is this process scalable for industrial captopril intermediate production?
A: Yes, the process is highly scalable as it avoids the use of expensive and difficult-to-remove resolving agents like dicyclohexylamine. The reliance on common solvents like ethyl acetate and simple pH adjustments makes it ideal for large-scale manufacturing, ensuring supply chain continuity and cost efficiency for API producers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-3-Thioacetyl-2-Methylpropionyl-L-Proline Supplier
At NINGBO INNO PHARMCHEM, we leverage deep technical expertise to bring advanced synthetic routes like CN101289417B to commercial reality. Our R&D team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in the lab are faithfully reproduced at an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art chiral HPLC and polarimetry instruments to guarantee that every batch meets the exacting standards required for Captopril synthesis.
We invite global partners to collaborate with us to optimize their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and reliability for your pharmaceutical projects.
