Advanced Synthesis of Caronic Acid: A Safer, Scalable Route for HCV Drug Intermediates
The strategic landscape of antiviral drug manufacturing is constantly evolving, driven by the need for safer, more cost-effective synthetic routes for critical intermediates. A pivotal development in this domain is detailed in patent CN114456057A, which discloses a novel synthesis method for caronic acid, a vital building block for direct-acting antivirals (DAAs) such as boceprevir and telaprevir used in treating Hepatitis C. Unlike traditional methods that rely on hazardous solvent systems and scarce starting materials, this innovation leverages kungfu acid (cyhalothrin acid) as a robust precursor. By utilizing a water-based oxidative cleavage protocol, the process achieves exceptional purity profiles while mitigating significant industrial safety risks. This technical breakthrough represents a paradigm shift for reliable pharmaceutical intermediates suppliers aiming to optimize their supply chains for complex cyclopropane derivatives.
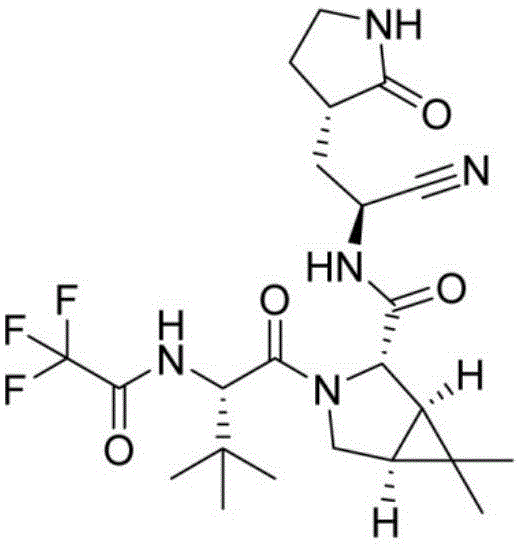
Caronic acid serves as a cornerstone scaffold in the construction of potent protease inhibitors, where stereochemical integrity and impurity control are paramount. The structural complexity of these final drug substances demands an intermediate synthesis that is not only chemically efficient but also commercially viable on a multi-ton scale. The methodology outlined in the patent addresses the historical bottlenecks of caronic acid production, specifically targeting the high costs and safety hazards associated with legacy routes. For procurement leaders and R&D directors, understanding this transition from niche fine chemicals to broadly available agrochemical intermediates is crucial for long-term cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
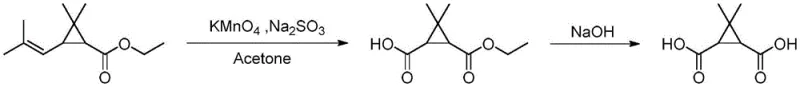
Historically, the industrial synthesis of caronic acid has relied heavily on ethyl chrysanthemate as the primary starting material. This conventional pathway, while chemically feasible, presents severe drawbacks that hinder modern pharmaceutical production standards. The oxidation step typically employs potassium permanganate in an acetone solvent system. This combination is notoriously dangerous because the reaction between permanganate and acetone can generate acetone peroxide, a highly unstable and explosive byproduct. This safety hazard imposes rigorous engineering controls and limits the maximum batch size, effectively capping production capacity and increasing insurance and operational overheads. Furthermore, ethyl chrysanthemate itself is a specialized fine chemical with a limited supplier base, leading to volatile pricing and potential supply chain disruptions. The reliance on acetone also complicates waste management, as solvent recovery is energy-intensive and the spent solvent often cannot be recycled indefinitely due to contamination, driving up both environmental compliance costs and raw material expenditure.
The Novel Approach
The innovative method described in patent CN114456057A fundamentally reengineers the synthesis by substituting the starting material with kungfu acid, a derivative widely produced for the agrochemical industry (specifically for pyrethroid insecticides). This switch immediately alleviates raw material scarcity, as kungfu acid is manufactured on a massive global scale, ensuring a stable and competitive supply. More critically, the new process replaces acetone with water as the reaction solvent. This substitution completely eliminates the risk of acetone peroxide formation, transforming a high-hazard operation into a benign aqueous oxidation. The reaction conditions are remarkably mild, proceeding efficiently at temperatures between 30-40°C, which reduces energy consumption for heating and cooling. The use of water also simplifies the workup procedure; inorganic salts and manganese byproducts are easily managed through filtration and aqueous washes, significantly reducing the environmental footprint and facilitating easier commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Permanganate-Mediated Oxidative Cleavage
The core chemical transformation in this novel route is the oxidative cleavage of the carbon-carbon double bond present in the side chain of kungfu acid. The mechanism initiates with the interaction of the alkene moiety with the permanganate ion (MnO4-) under alkaline conditions provided by potassium hydroxide. Initially, a cyclic manganate ester is formed via a syn-addition across the double bond. Under the controlled thermal conditions of 30-40°C, this intermediate undergoes hydrolysis and further oxidation, ultimately severing the carbon-carbon bond to generate two carboxylic acid functionalities. One end remains attached to the cyclopropane ring, while the other forms the acetic acid side chain characteristic of caronic acid. The presence of the electron-withdrawing trifluoromethyl and chloro groups on the leaving fragment facilitates the cleavage, driving the reaction to completion. The stoichiometry is carefully balanced, typically employing a molar ratio of potassium permanganate to kungfu acid of roughly 5:1 to ensure full conversion without excessive over-oxidation of the sensitive cyclopropane ring.
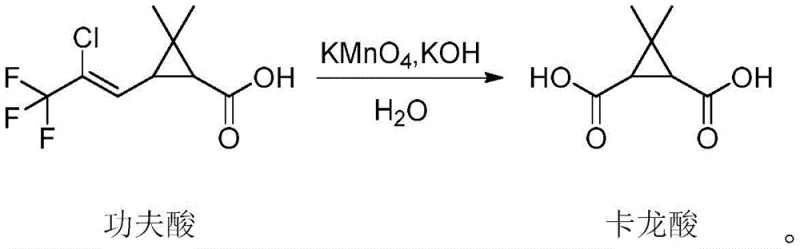
Impurity control is meticulously managed through the quenching and isolation phases. After the oxidation is complete, residual permanganate and manganese dioxide are handled by filtration and chemical reduction using sodium sulfite. This step is critical to prevent oxidative degradation of the product during the acidic workup. The subsequent acidification to pH 1-2 using hydrochloric acid protonates the carboxylate salts, precipitating the free acid. The addition of sodium chloride induces a salting-out effect, which enhances the partition coefficient of the organic product into the extraction solvent (ethyl acetate). Finally, the purification is achieved not through traditional recrystallization but via a pulping process in methyl tert-butyl ether at low temperatures (0-5°C). This technique effectively washes away soluble impurities while retaining the crystalline lattice of the high-purity caronic acid, consistently delivering material with purity exceeding 99% as confirmed by NMR analysis.
How to Synthesize Caronic Acid Efficiently
The operational protocol for this synthesis is designed for robustness and reproducibility in a GMP environment. The process begins with the dissolution of kungfu acid and potassium hydroxide in water, followed by the controlled, batch-wise addition of potassium permanganate to manage exotherms. Following the oxidation period, the reaction mixture is filtered to remove manganese solids, and the filtrate is quenched with sodium sulfite. The product is then liberated via acidification, extracted, and subjected to a specialized pulping crystallization to ensure pharmaceutical-grade quality. For the detailed standardized operating procedures, including specific addition rates, stirring speeds, and drying parameters, please refer to the technical guide below.
- Dissolve kungfu acid and solid potassium hydroxide in water, cooling the mixture to 10-20°C before batch addition of potassium permanganate.
- Heat the reaction mixture to 30-40°C to facilitate oxidation, monitoring until raw material consumption is complete, then filter off manganese dioxide byproducts.
- Quench the filtrate with sodium sulfite, adjust pH to 1-2 with hydrochloric acid, extract with ethyl acetate, and purify via pulping in methyl tert-butyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this kungfu acid-based route offers transformative advantages that extend beyond simple chemistry. The shift from a fine chemical starting material (ethyl chrysanthemate) to a commodity agrochemical intermediate (kungfu acid) fundamentally alters the cost structure of the supply chain. Because kungfu acid is produced in vast quantities for the pesticide industry, its price point is significantly lower and less susceptible to the volatility typical of low-volume pharmaceutical precursors. This abundance ensures that manufacturers can secure long-term contracts with stable pricing, mitigating the risk of production stoppages due to raw material shortages. Furthermore, the elimination of acetone removes a major regulatory and safety burden, streamlining the permitting process for new manufacturing lines and reducing the capital expenditure required for explosion-proof infrastructure.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by multiple factors inherent to the reaction design. By utilizing water as the primary solvent, the need for expensive, anhydrous organic solvents is eliminated, drastically cutting raw material costs. Additionally, the avoidance of acetone prevents the formation of hazardous peroxides, which in turn reduces the costs associated with specialized safety monitoring, waste disposal of hazardous peroxide-contaminated streams, and insurance premiums. The high yield and purity achieved reduce the need for extensive downstream purification steps, lowering labor and utility consumption per kilogram of product. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final API.
- Enhanced Supply Chain Reliability: Supply security is a critical metric for any pharmaceutical program, and this synthesis route significantly de-risks the supply of caronic acid. Kungfu acid is a mature industrial chemical with a well-established global supply base, unlike ethyl chrysanthemate which has a limited number of producers. This diversification allows buyers to qualify multiple vendors for the starting material, preventing single-source bottlenecks. Moreover, the simplified process technology—requiring only standard stainless steel reactors and filtration equipment rather than specialized hazard-contained units—means that more contract manufacturing organizations (CMOs) have the capability to produce this intermediate. This increased manufacturability expands the available capacity in the market, ensuring consistent delivery schedules and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this water-based oxidation is vastly superior to legacy methods. The absence of volatile organic compounds (VOCs) like acetone reduces atmospheric emissions and simplifies solvent recovery systems. The aqueous waste stream, primarily containing inorganic salts and manganese residues, is easier to treat and dispose of in compliance with stringent environmental regulations. The mild reaction temperatures (30-40°C) minimize energy usage for heating and cooling, contributing to a lower carbon footprint for the manufacturing process. These green chemistry attributes align perfectly with the sustainability goals of major pharmaceutical companies, facilitating smoother regulatory approvals and enhancing the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of caronic acid via this novel oxidative cleavage method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of the operational realities and benefits. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing drug substance workflows.
Q: Why is the new kungfu acid route superior to the traditional ethyl chrysanthemate method?
A: The new route eliminates the use of acetone, thereby removing the severe safety risk of explosive acetone peroxide formation. Additionally, kungfu acid is a widely available agrochemical intermediate, offering better supply stability and lower raw material costs compared to ethyl chrysanthemate.
Q: How does this process ensure high purity for pharmaceutical applications?
A: The process utilizes a rigorous workup involving pH-controlled acidification, salting-out effects with sodium chloride, and a specialized pulping crystallization step using methyl tert-butyl ether at low temperatures, consistently achieving purity levels of 99%.
Q: Is this oxidative cleavage method suitable for large-scale commercial production?
A: Yes, the use of water as the primary solvent significantly simplifies thermal management and waste treatment compared to organic solvents. The mild reaction temperatures (30-40°C) and standard filtration techniques make it highly amenable to multi-ton scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the future of antiviral drug manufacturing. As a premier CDMO partner, we possess the technical expertise to implement this advanced kungfu acid oxidation process at scale. Our facilities are equipped to handle complex oxidative chemistries with rigorous safety protocols, ensuring that every batch meets the highest standards of quality and consistency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to support your needs from clinical trial materials through to full commercial launch. Our commitment to excellence is backed by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify identity and assay.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and discover how our optimized manufacturing capabilities can drive value and security for your hepatitis C inhibitor programs.
